Stem Cell Clinic and Parkinson: What Parkinsons Stem Cell Trial Research Really Shows
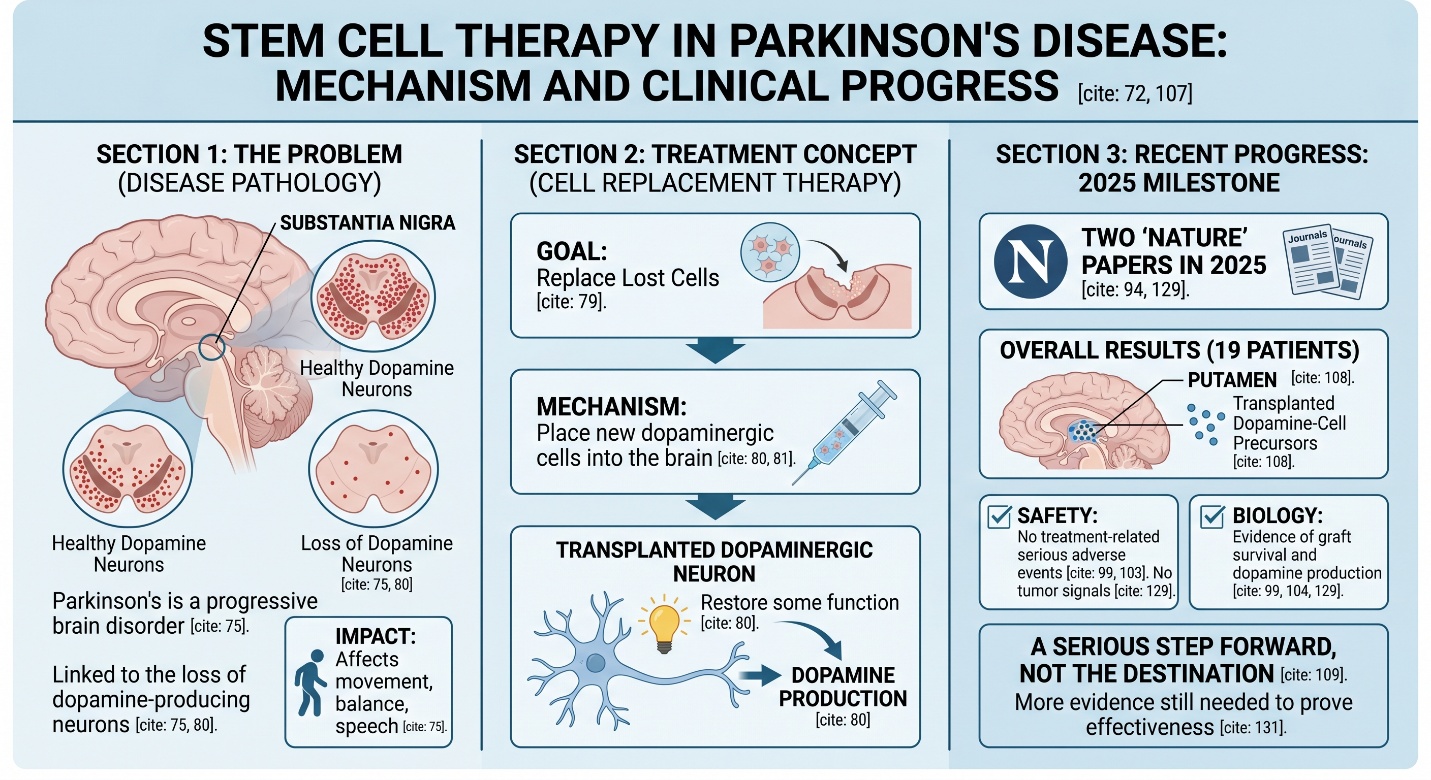
If someone is searching for a stem cell clinic and Parkinson, they are usually not looking for abstract lab science. They are looking for a real answer to a hard question: can anything do more than manage symptoms? That question makes sense. Parkinson’s disease is a progressive brain disorder linked to the loss of dopamine-producing neurons, and over time it can affect movement, balance, speech, sleep, mood, and daily independence. Current standard treatments can help a great deal, but they do not stop the disease itself.
Why stem cells became such a major Parkinson topic
The logic is biologically strong
Among neurodegenerative diseases, Parkinson’s has always been one of the clearest candidates for cell replacement. The reason is straightforward: a relatively defined population of dopamine-producing neurons is lost, so researchers have long asked whether new dopaminergic cells could be placed into the brain to restore some function. That is the core scientific idea behind the modern parkinsons stem cell trial landscape.
But clinical reality is slower than clinic marketing
This is where the conversation needs more discipline. FDA says stem cell products generally require approval, and it explicitly warns that regenerative products have not been approved to treat neurological disorders such as Parkinson’s disease. FDA also says many patients may be misled by products that are illegally marketed and have not been shown to be safe or effective. So when a stem cell clinic presents Parkinson’s treatment as if it were already routine medicine, that should immediately trigger harder questions.
What standard Parkinson care still does better than hype
Established therapy is still symptom-focused, but it matters
One reason people keep looking beyond standard care is that Parkinson treatment is still largely symptomatic. NINDS notes that medicines and deep brain stimulation can reduce tremor, rigidity, stiffness, and some motor complications, but they are not a cure. That gap is exactly what makes regenerative medicine so compelling. At the same time, it also explains why people can become vulnerable to overpromising language from clinics that sound more certain than the evidence really is.
Where the real Parkinson stem cell trial evidence stands now
2025 was a turning point, but not the finish line
The most important recent development is that two independent Nature papers in 2025 reported early human trial data for Parkinson’s cell therapy. Nature’s accompanying analysis summarized the message clearly: transplanting dopamine-releasing neurons into the brain is promising, the two trials supported safety, but more evidence is still needed to prove effectiveness. That is probably the single best one-line summary of the field right now.
Trial 1: iPS-cell-derived dopaminergic progenitors
Seven patients received bilateral transplantation of dopaminergic progenitors from induced pluripotent stem cells (iPSCs) in a phase I/II trial at Kyoto University. There were no serious adverse events on patients, proof of graft survival and dopamine production through imaging or tumorigenic overgrowth at 24 months from the Nature paper. The authors concluded that the findings were consistent with safety and potential clinical benefit, which is good news but remains a long way from robust evidence of efficacy.
Trial 2: hES-cell-derived dopaminergic neurons
A second Nature study in 2025 led by Dr. Dinsmore had bemdaneprocel, a human embryonic stem-cell-derived dopaminergic progenitor product, tested on 12 patients also published at the same time as Dr. Birsofer’s findings (3). No treatment-related serious adverse events occurred, MRI showed neither tumors nor putaminal overgrowth; PET supported the survival of dopaminergic grafts and exploratory motor outcomes suggested a possible benefit particularly in high-dose group through 18 months. However, the paper also makes it clear that this was a small early-phase safety trial and did not have power or design to determine efficacy.
Why these studies matter even though they are still early
These trials matter because they move Parkinson’s stem cell research out of the realm of theory and into real human evidence. The Parkinson’s Foundation highlighted the same point: the two Nature studies involved 19 people in total, with transplanted dopamine-cell precursors placed into the putamen, and the results were reassuring on safety with no tumor signals in either study. That is a serious step forward. It is also still a step, not the destination.
What this means for a stem cell clinic conversation
A credible clinic should sound more careful than promotional
This is where the keyword stem cell clinic becomes useful in a practical way. A credible clinic conversation about Parkinson’s should sound cautious, specific, and trial-aware. It should be clear about whether the treatment is part of a registered clinical trial, what cell type is being used, how the cells are delivered, what surgery and immunosuppression may be involved, what outcomes are being measured, and what long-term follow-up exists. ISSCR’s patient guide exists for exactly this reason: to help patients make informed decisions about stem cell treatments and clinical trials rather than relying on broad promotional claims.
The biggest red flag is false certainty
The real Parkinson trial literature does not sound like a miracle. It sounds careful. The strongest papers are saying “safe so far,” “dopamine production seen,” “possible clinical benefit,” and “larger efficacy studies still needed.” If a clinic skips that nuance and jumps straight to claims that Parkinson’s can already be reversed, repaired, or routinely restored, it is moving beyond what the best current evidence supports.
The honest conclusion
Stem cell therapy for Parkinson’s is no longer science fiction. The 2025 parkinsons stem cell trial results are a genuine milestone, because they show that dopaminergic cell replacement can be delivered in humans with reassuring early safety signals and biologic evidence of graft survival. But the field is still early, still surgical, still highly specialized, and still not approved as routine Parkinson’s treatment. So the most responsible way to talk about a stem cell clinic and Parkinson today is this: the science is real, the momentum is real, and the hope is justified — but the proof is still being built.
Frequently Asked Questions
1) Is there already an approved stem cell treatment for Parkinson’s disease?
No. FDA says regenerative products such as stem cell products have not been approved to treat neurological disorders including Parkinson’s disease.
2) Why are Parkinson’s stem cell trials getting so much attention now?
Because two 2025 Nature studies showed that dopaminergic cell replacement in Parkinson’s patients had reassuring early safety findings, no tumor signal in those early follow-up windows, and biologic signs that the grafted cells survived and produced dopamine.
3) Did those trials prove that stem cells cure Parkinson’s?
No. Nature’s own News & Views said the trials showed safety, but more evidence is needed to prove effectiveness. The hES-cell trial also stated that it was not designed or powered to determine efficacy.
4) What should I ask a stem cell clinic about Parkinson’s?
Ask whether the treatment is part of a registered clinical trial, what exact cell product is being used, what surgery and immunosuppression are involved, what outcomes are tracked, and what long-term safety follow-up exists. ISSCR specifically provides patient guidance to help people ask those questions before agreeing to treatment.
5) Are standard Parkinson treatments still important if stem cell therapy is advancing?
Yes. Current treatments such as medication and deep brain stimulation remain central because they can meaningfully improve symptoms, even though they do not stop the disease. Stem cell therapy is still investigational, so standard care remains essential.