Why this topic needs a slower, more careful conversation
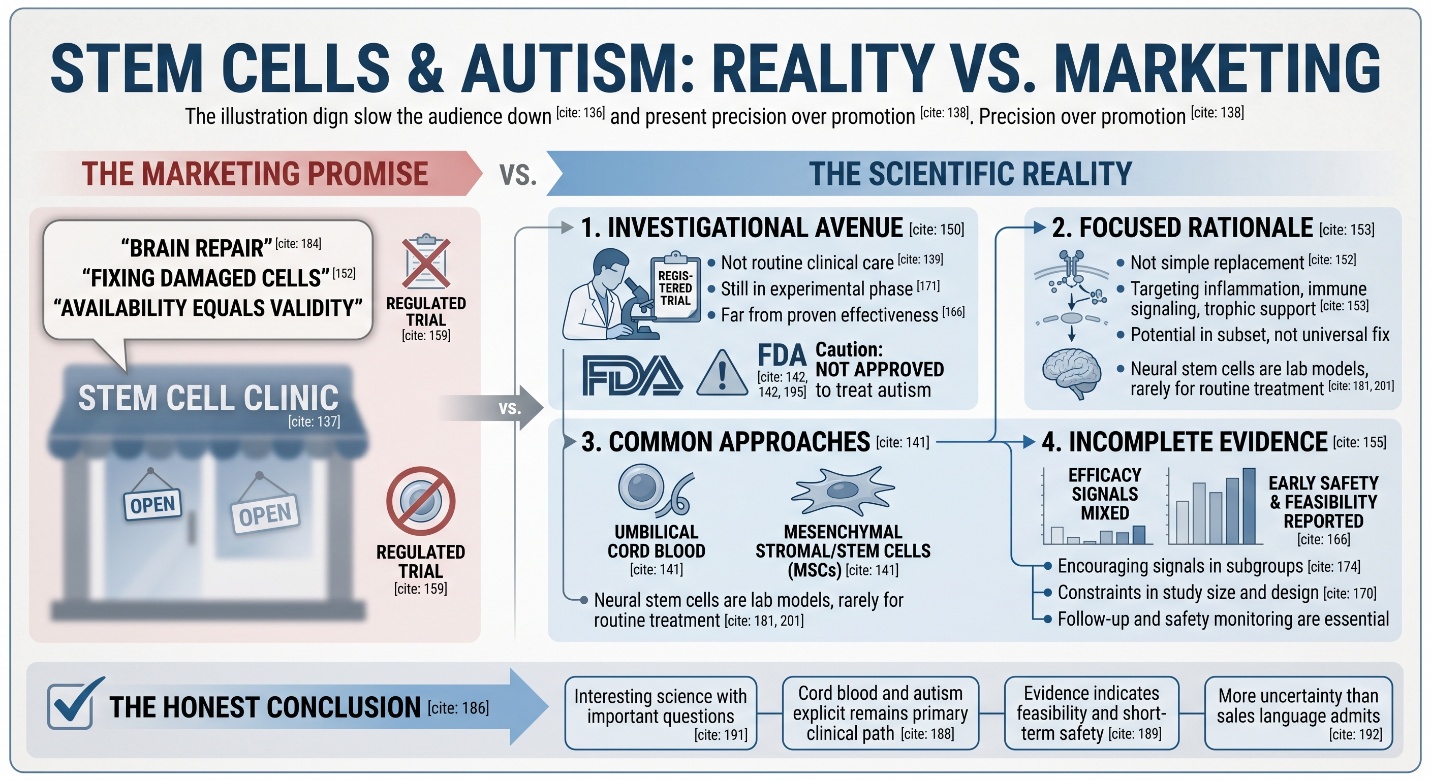
When families search stem cell clinic and autism, they usually are not looking for abstract science. They are looking for possibilities. Autism spectrum disorder is a neurodevelopmental condition with a very wide range of presentations, supports, and outcomes, which is exactly why this topic needs more precision than clinic marketing often gives it. At the moment, stem cell research in autism is real enough to discuss seriously, but it is still far from routine clinical care.
The distinction is important because the words “stem cell treatment” like so many health buzzwords here and elsewhere, can sully honest discussions around medical science, making very different things sound interchangeable. In autism research, the majority of investigated approaches have included either umbilical cord blood or mesenchymal stromal/stem cells (MSCs), not a ubiquitous therapy that every clinic provides in an identical way. Regenerative medicine therapies have not been approved to treat autism, according to the FDA; meanwhile, the ISSCR urges patients and families to closely examine evidence of safety or efficacy for any stem cell intervention being promoted.
What “cord blood and autism” actually means
Cord blood is one of the main clinical research paths
Cord blood and autism is one of those phrases that we see repeated over, and over again in this field. This is partly because some of the most well-known research trials have examined umbilical cord blood transfusions in autistic kids. While subsequent discussion about that program emphasized that any positive effects were still restricted to specific segments rather than providing a general benefit for all children with autism, a randomized phase II Duke trial focused on whether cord blood infusion was safe and if it correlated with improvements in social communication.
Moreover, smaller studies outside that trial program exist. A 2024 study, for instance, on the safety and efficacy of autologous umbilical cord blood infusion in 20 autistic children was only intended to show that this question —as it concerns all forms of autism— has not been answered. The important caveat here is that this remains an investigational avenue as opposed to a treatment silo which has already made the way into established autism care.
Why researchers were interested in cord blood in the first place
The scientific rationale has never really been about “replacing damaged brain cells” in a simple way. The more common research idea is that cord blood cells or MSCs might influence inflammation, immune signaling, trophic support, or neurodevelopment-related pathways in a subset of autistic children. That is a much narrower and more defensible claim than saying stem cells “fix” autism. Reviews of autism stem cell studies keep returning to this same point: the rationale is biologically interesting, but the mechanisms are still being worked out and the clinical evidence is still incomplete.
Where stem cell clinic language starts to blur the science
A clinic offer is not the same thing as a validated treatment
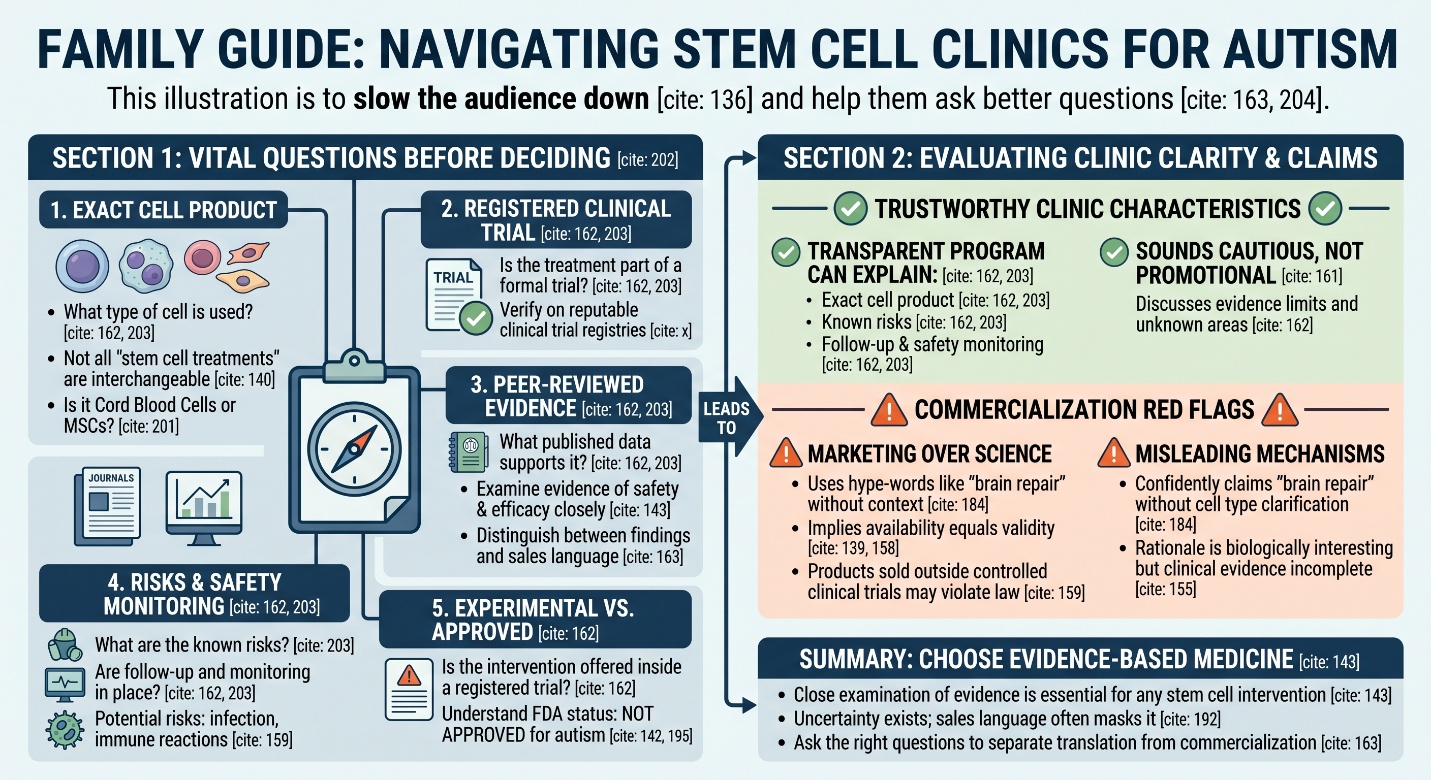
This is where stem cell clinic comes into play. A clinic can say they have availability, but that doesn’t mean they’re validating. First, the FDA cautions that regenerative medicine products sold outside of well-controlled clinical trials may violate Federal law and it specifically says: Regenerative Medicine Therapies Have Not Been Approved to Treat Autism When the products are marketed with little evidence or oversight, their risks include infection and unwanted immune reactions to them; contamination of tissue from intended injuries caused by derived cells, as well as other serious harms.
A trustworthy clinic conversation should therefore sound more cautious than promotional. It should explain what exact cell product is being discussed, whether the intervention is being offered inside a registered clinical trial, what the published evidence actually shows, what remains unknown, and what follow-up and safety monitoring are in place. That is also the spirit of the ISSCR patient guidance, which is designed to help families separate legitimate clinical translation from premature commercialization.
What the studies have actually shown so far
Safety signals are stronger than efficacy signals
Across the published literature, the most consistent finding is not “proven effectiveness.” It is early safety and feasibility. A 2020 phase I study of intravenous human umbilical cord tissue-derived mesenchymal stromal cells in children with autism reported that treatment in 12 children was safe and feasible. Reviews and commentaries built around those data continue to describe MSC administration in autism as promising enough to justify further trials, but still too early for routine clinical use.
This same pattern holds true for bigger assessments as well. In 2022, a group of meta-analysis authors concluded autologous stem cell therapy is similarly safe and potentially efficacious in children with autism spectrum disorder but also concentrated on the constraints to available evidence. But a 2023 review of 10 clinical studies noted that “nearly all studies report positive outcomes and no apparent serious adverse events,” yet also showed whatever findings they made remain too small in both study size and experimental designs to draw broad conclusions about the area. Two other reviews from 2025 and 2026 also labeled autism stem cell therapy an experimental field with potential for clinical utility, but still not evidence based.
The efficacy story is still mixed
This is the part families most need stated plainly. There are signals of possible benefit in some studies, and some trial programs have reported more encouraging results in certain subgroups. But the overall evidence is still mixed because studies vary in cell source, dosing, age range, outcome measures, trial design, and follow-up duration. That is why recent reviews keep using phrases like emerging evidence, clinical prospects, or promising, rather than treating efficacy as a proven fact.
Where neural stem cells fit — and where they do not
Neural stem cells matter more in the lab than in current autism treatment
Stem cell treatment for autism must mean neural stem cells, right? Many clinicians who might treat Autism using an autologous HSCT readily assume that what they use is a form of neural stem. Neural stem cells are considerably more influential in laboratory disease models and mechanistic studies than they are for routine patient treatment at the moment, according to current autism research. Stem cells are reported in reviews to be an important tool for autism modeling of neurodevelopment and potential future therapeutics, but clinical trials remain skewed heavily towards cord blood-based or MSC-type approaches.
That difference matters because it prevents the conversation from becoming more futuristic than it really is. A clinic that talks loosely about “brain repair” without clarifying the actual cell type, rationale, and evidence is usually sounding more confident than the literature does. The real science is narrower, more careful, and much more investigational.
The most honest conclusion
The most reasonable reading of the evidence is not scornful or exuberant. Research in stem cell focused to Autism is active and cord blood and autism & explicit remains one of the most often studied clinical pathways. Early studies indicate feasibility and at least some short-term safety in carefully selected settings, with potential benefit reported among certain groups. However, the field remains investigational and although there is signaling of clinical efficacy no signal has been definitive and stem cell therapy by itself to treat autism is not an FDA-approved treatment. Which means that today, if someone were to ask what a stem cell clinic and autism should mean, the most honest answer would be: interesting science; important questions — but much more uncertainty than the sales language usually admits.
Frequently Asked Questions
1) Is stem cell therapy for autism FDA-approved?
No. The FDA states that regenerative medicine therapies have not been approved to treat autism.
2) What does “cord blood and autism” usually refer to?
Usually it refers to clinical studies using umbilical cord blood infusions in children with autism, including randomized and early-phase trials that have examined safety and possible behavioral outcomes.
3) Have autism stem cell studies shown any benefit at all?
Some studies and reviews suggest possible benefits in selected measures or subgroups, but the evidence is still mixed and limited by small samples, heterogeneous protocols, and incomplete long-term data.
4) Are neural stem cells the same thing as the stem cells used in most autism clinics or trials?
Usually not. Most clinical autism studies have involved cord blood cells or MSC-type approaches, while neural stem cells are more prominent in research models and preclinical work.
5) What should families ask a stem cell clinic before taking the next step?
They should ask what exact cell product is being used, whether the treatment is part of a registered clinical trial, what peer-reviewed evidence supports it, what the known risks are, and what follow-up care is included. The ISSCR patient guide is specifically meant to help families ask those questions.