Amyotrophic Lateral Sclerosis (ALS) is a progressive neurodegenerative disease characterized by the selective loss of motor neurons, leading to muscle weakness, paralysis, and eventual respiratory failure. Current therapies provide limited disease-modifying effects, prompting increasing interest in regenerative approaches. Among these, umbilical cord-derived mesenchymal stromal/stem cells (UC-MSCs) and double-filtration plasmapheresis (DFPP) have emerged as potential strategies targeting neuroinflammation and systemic immune dysregulation. This article reviews the biological rationale, current evidence, and limitations of combining UC-MSCs with DFPP in ALS. While preliminary data suggest safety and theoretical synergy, robust clinical evidence remains limited, and this approach should be considered investigational.
Introduction
Amyotrophic Lateral Sclerosis (ALS) is a fatal neurodegenerative disorder marked by progressive degeneration of upper and lower motor neurons. The disease leads to declining motor function, respiratory compromise, and ultimately death, typically within 3–5 years of diagnosis. Despite advances in supportive care, therapeutic options remain limited, with approved agents such as riluzole and edaravone offering only modest benefits in disease progression.
Given the multifactorial pathophysiology of ALS including neuroinflammation, oxidative stress, mitochondrial dysfunction, and immune dysregulation there is increasing interest in therapies that target the broader disease environment rather than a single pathway. In this context, mesenchymal stem cells (MSCs) and extracorporeal blood purification techniques such as DFPP are being explored as complementary approaches.
Pathophysiology of ALS and Therapeutic Targets
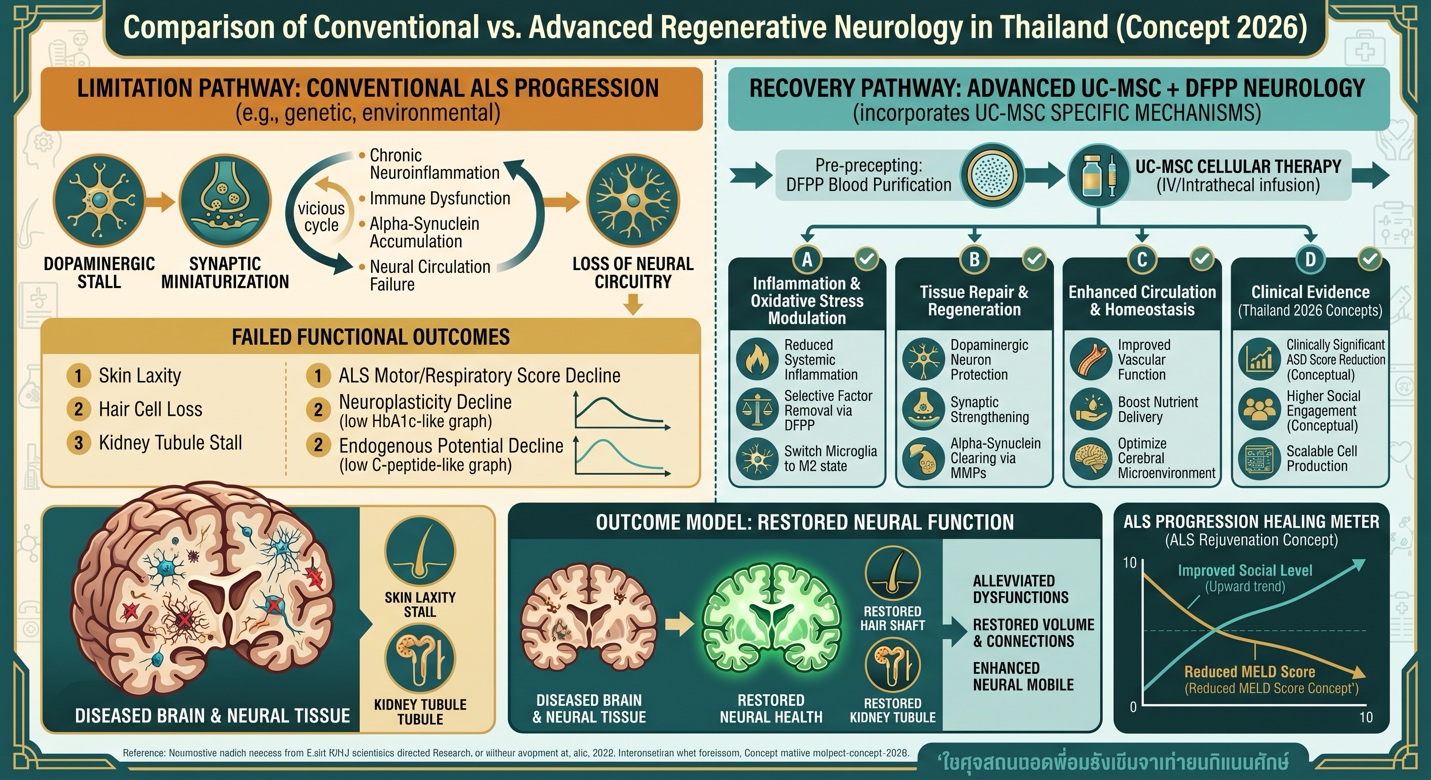
ALS is increasingly understood as a multisystem disorder involving not only neuronal degeneration but also glial activation, systemic inflammation, and immune dysregulation. Microglial activation and astrocyte dysfunction contribute to neurotoxicity through the release of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6 [4].
Additionally, circulating immune mediators and peripheral inflammation have been implicated in disease progression, suggesting that ALS may involve both central and systemic pathological processes. This dual-component pathology supports the exploration of combined therapies targeting both the central nervous system and peripheral immune environment.
UC-MSCs: Biological Mechanisms in ALS
Umbilical cord-derived mesenchymal stem cells (UC-MSCs) have gained attention due to their low immunogenicity, high proliferative capacity, and strong paracrine activity. Unlike neural stem cells, MSCs are not primarily used for direct neuronal replacement. Instead, their therapeutic potential lies in their ability to modulate the disease environment.
Key mechanisms of UC-MSCs include:
Immunomodulation: MSCs suppress pro-inflammatory cytokines and promote regulatory immune responses
Paracrine signaling: Secretion of growth factors such as BDNF, GDNF, and VEGF
Neuroprotection: Reduction of apoptosis and oxidative stress
Microglial modulation: Shifting microglia from pro-inflammatory (M1) to anti-inflammatory (M2) states
Preclinical studies in ALS models have demonstrated delayed disease progression and improved survival following MSC administration. Early-phase clinical trials have also reported favorable safety profiles, although efficacy outcomes remain variable.
Double-Filtration Plasmapheresis (DFPP): Mechanistic Rationale
DFPP is an extracorporeal blood purification technique that separates plasma and selectively removes high-molecular-weight substances, including immune complexes, autoantibodies, and inflammatory mediators.
Unlike conventional plasma exchange, DFPP allows for selective filtration, reducing the need for plasma replacement and minimizing associated risks. DFPP has been used in conditions such as autoimmune diseases, hyperlipidemia, and transplant-related complications.
In ALS, DFPP is hypothesized to:
Reduce circulating inflammatory cytokines
Remove pathogenic plasma components
Modulate systemic immune activation
Improve the biological environment for cellular therapies
However, its application in ALS remains experimental, with limited direct clinical data.
Rationale for Combining UC-MSCs with DFPP
The combination of UC-MSCs and DFPP is based on a complementary therapeutic hypothesis:
DFPP may reduce systemic inflammatory burden, potentially improving the host environment
UC-MSCs may provide regenerative signaling, supporting neuronal survival and repair
This approach reflects a broader shift in regenerative medicine toward multi-modality strategies that address both systemic and local disease mechanisms.
Theoretically, preconditioning the immune environment via DFPP may enhance the effectiveness of MSC-based therapy. However, this synergy remains hypothetical and requires validation through controlled studies.
Clinical Evidence and Current Limitations
MSC-Based Therapy in ALS
Several clinical trials have investigated MSC therapy in ALS. A randomized controlled trial of intrathecal MSC administration demonstrated safety and suggested potential slowing of disease progression in selected patients. Other studies have explored repeated dosing and different delivery routes, including intravenous and intrathecal approaches.
Despite these findings, major limitations remain:
Small sample sizes
Heterogeneous study designs
Lack of long-term efficacy data
Variability in cell source and preparation
DFPP in Neurological Disease
DFPP has shown efficacy in immune-mediated neurological disorders such as myasthenia gravis and Guillain–Barré syndrome. However, its application in ALS is not well established, and evidence remains limited to theoretical and exploratory use.
Combination Therapy
At present, no large-scale clinical trials have specifically evaluated the combination of UC-MSCs with DFPP in ALS. As such, this approach should be considered investigational, and conclusions regarding efficacy cannot yet be drawn.
Safety and Regulatory Considerations
Stem cell therapies are subject to regulatory oversight to ensure safety, purity, and potency. While MSC-based therapies have demonstrated favorable safety profiles in early trials, concerns remain regarding:
Standardization of cell preparation
Long-term safety
Risk of unregulated clinical use
Similarly, DFPP is generally considered safe when performed in controlled medical settings, but it carries risks associated with extracorporeal circulation, including hypotension and coagulation disturbances.
Clinical Implications and Patient Selection
Given the current evidence, the use of UC-MSCs with DFPP in ALS should be limited to:
Carefully selected patients
Structured clinical programs or research settings
Multidisciplinary medical supervision
It should not replace standard ALS care, which remains centered on neurological management, respiratory support, and symptom control.
Conclusion
The combination of UC-MSCs and DFPP in ALS represents an emerging and biologically plausible approach within regenerative medicine. By targeting both systemic inflammation and local neurodegeneration, this strategy aligns with current understanding of ALS as a multifactorial disease.
However, despite promising theoretical mechanisms and early safety data, robust clinical evidence remains insufficient. Future research should focus on well-designed randomized trials to evaluate efficacy, optimize protocols, and define patient populations most likely to benefit.
Until then, this approach should be regarded as investigational, with cautious interpretation and responsible clinical application.

