Why does this topic sound more certain than it really is
More than likely, if someone is searching for Plant-Based Stem Cell Therapy for baldness, they are seeking a more optimistic answer besides shampoo, as well as something further along the spectrum of technology and efficacy than basic hair serums. That makes sense. It’s so visible, emotional, and marketable—hair loss. But this is precisely the sort of subject that can see language get ahead of science. But so far, the evidence is compelling enough to take seriously and messy enough not to be able to flatten out into a straightforward cure narrative. In 2025 and 2026, reviews described regenerative hair medication as a “fast-moving area” with promise, yet always underlying the point that standardization and longitudinal validation are generally not where patients think they are.
What “baldness” usually means in real clinical terms
Most of the time, people mean androgenetic alopecia
They are probably looking for a more optimistic answer than basic shampoo, something at the far end of the technology and efficacy spectrum from hair serums or what passes for therapy in corporate salons. That makes sense. Hair loss: so visible, emotionally-charged, and marketable. However, this is exactly the kind of topic that lets language outrun science. Yet thus far the evidence is intriguing enough to warrant serious consideration and inconsistently messy adequate not be flattened out into an unambiguous remedy narrative. Even in 2025 and ’26, reviews of regenerative hair medicine described it as “a fast-moving area” which had potential but invariably seemed to come back around to the idea that standardizations and longitudinal validation are rarely where people wish them to be.
What Plant-Based Stem Cell Therapy usually means
In most cases, this is closer to cosmetic biotechnology than clinical stem cell medicine
The phrase Plant-Based Stem Cell Therapy sounds medical, but in published literature, it sits much closer to the cosmetic and personal-care world than to established clinical hair-loss therapy. A 2025 review on plant stem cells in the cosmetic industry describes this area as a source of active ingredients for cosmetic use, not as a standard medical treatment class for baldness. The same review also notes that some plant-cell-derived extracts have shown anti-hair-loss qualities in laboratory or product-development settings, which helps explain why the phrase has such strong marketing momentum. Still, that is very different from saying plant stem cells themselves are being implanted into the scalp as a proven baldness treatment.
A good example of Plant-Based Stem Cell Therapy
Plant-Based Stem Cell Therapy is perhaps one of the clearest examples Plant-Based Stem Cell Therapy Malus Domestica is an ingredient from Mibelle Biochemistry that targets hair, and the company says it helps to preserve characteristics of human stem cells in situ; prolongs follicular life span and reduces loss while promoting greater visible hair density. That is informative for categorization: these products are simply marketed as bioactive cosmetic ingredients and may aid in extending follicle lifespan or overall scalp biology, not to be confused with human stem cell transplantation or regulated regenerative medicine.
Where stem cell exosomes for hair loss enter the picture
This is a different conversation from plant stem cells
Stem cell exosomes for hair loss is a different layer of the field. Related Exosomes are extracellular vesicles secreted by cells, and usually their cellular use is acknowledged as cell-free messengers for therapeutic opportunities rather than living stem cells on themselves. Publications from 2025 and 2026 state that exosome-based hair strategies are beneficial since they can activate Wnt/β-catenin signaling, promote dermal papilla survival, enhance angiogenesis (the development of new blood vessels), and fine-tune inflammation in the vicinity of the follicle. One of the reasons why exosomes are so buzzworthy in hair restoration is that they sound more high-tech than topical agents but less invasive than cell transplantation.
The early evidence is promising, but still uneven
The current best evidence summary is stem cell exosomes for hair loss (most advanced, androgenetic alopecia), which shows promise, but the studies are still heterogeneous. BACKGROUND A 2025 systematic review on exosomes and hair regeneration calls mesenchymal stem cell-derived exosomes a new promising option of cell-free therapy for alopecia, while also determining that with their regenerative properties, there is hope in targeting them via treatment modalities, including Exosome Therapy, according to the conclusion, which supports testing some very preliminary studies along results however they are small & heterogeneous making larger calibrated clinical trials necessary. Similarly, there was a clinical publication for increased hair density and continued patient satisfaction after exosome injection in androgenetic alopecia published in 2024, which is not the same as fully mature guideline-level evidence.
What the strongest current evidence still says
The field is stronger on “improvement” than on “cure.”
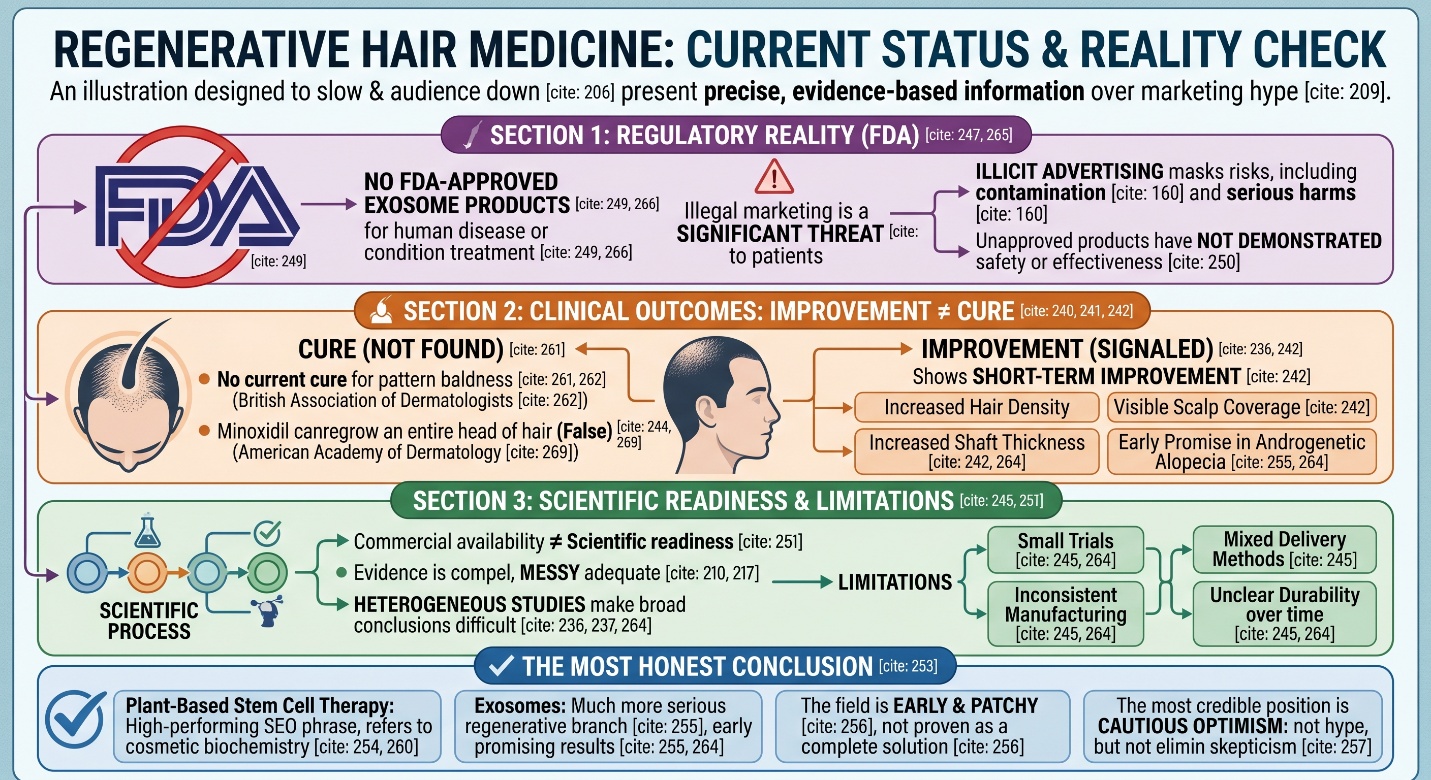
This is where the article needs its most important layer. Regenerative hair medicine is increasingly able to show short-term improvements in hair density, shaft thickness, or visible scalp coverage. But those outcomes are not the same thing as a permanent reversal of baldness. The BAD says there is no cure for male-pattern hair loss, and the AAD says minoxidil cannot regrow an entire head of hair. Even the more optimistic regenerative reviews keep returning to the same limitations: small trials, inconsistent manufacturing, mixed delivery methods, and unclear durability over time. That is why Plant-Based Stem Cell Therapy and stem cell exosomes for hair loss should be discussed as emerging approaches, not as settled answers.
Why regulation still matters
Exosomes are heavily marketed, but they are not FDA-approved hair-loss products
A significant reality check comes from the FDA. There are no FDA-approved exosome products at this time, which the agency takes pains to point out, and adds that any product intended for human disease or condition treatment must be approved by the FDA. The FDA states that patients might also be deceived by illicitly advertised regenerative products, which are significant threats because they do not demonstrate safety or effectiveness. This does not mean that the science is false. And so, commercial availability and scientific readiness are not one and the same. That distinction, in hair loss particularly, helps a lot.
The most honest conclusion
The best you could describe it as: Plant-Based Stem Cell Therapy is a high-performing SEO phrase, but in most circumstances still refers more to plant-based cosmetic biochemistry rather than tested medicine for baldness. Stem cell exosomes for hair loss are a much more serious branch of regenerative research, and early results have been promising — especially in androgenetic alopecia. But even then, the field is early and patchy, not proven enough to be described as more than a near-solution. The most credible position at present is cautious optimism: better science than just hype, but not yet evidence that should eliminate skepticism.
Frequently Asked Questions
1) What is Plant-Based Stem Cell Therapy for baldness?
Usually, it refers to plant-cell-derived cosmetic or scalp-care ingredients rather than the use of living human stem cells as a regulated medical treatment.
2) Is there a real cure for pattern baldness?
Not at this point. The British Association of Dermatologists states that there is no cure for male-pattern hair loss, although treatments can help slow progression and improve hair growth in some people.
3) Do stem cell exosomes for hair loss actually work?
Early studies and recent reviews suggest they may improve hair density and thickness, especially in androgenetic alopecia, but the evidence is still heterogeneous and not yet fully standardized.
4) Are exosome hair-loss treatments officially approved?
No FDA-approved exosome products currently exist, and the FDA says exosome products intended to treat disease or conditions in humans require approval.
5) What still works best in routine care for hair loss?
Established options such as minoxidil and other diagnosis-specific treatments remain the most grounded starting point. The AAD notes that minoxidil can help with early hair loss, even though it cannot regrow an entire head of hair.