Amyotrophic Lateral Sclerosis (ALS) is a fatal neurodegenerative disorder which specifically targets motor neurons and ultimately leads to muscle weakness, paralysis and respiratory failure. This all implies that we have to shift towards therapies with some potential for direct disease-modification, and as such existing therapy only partially modifies the disease process, something renewing interest in regenerative approaches. The latter include those that target neuroinflammation or systemic immune dysregulation, such as umbilical cord-derived mesenchymal stromal/stem cells (UC-MSCs) and double-filtration plasmapheresis (DFPP). We provide the biologic rationale for this combination, available data (albeit limited) and its shortcomings in AMAS. This approach appears safe in early data and might be synergistic theoretically, but there is no clinical evidence supporting this strategy with the potential needs to remain experimental.
Introduction
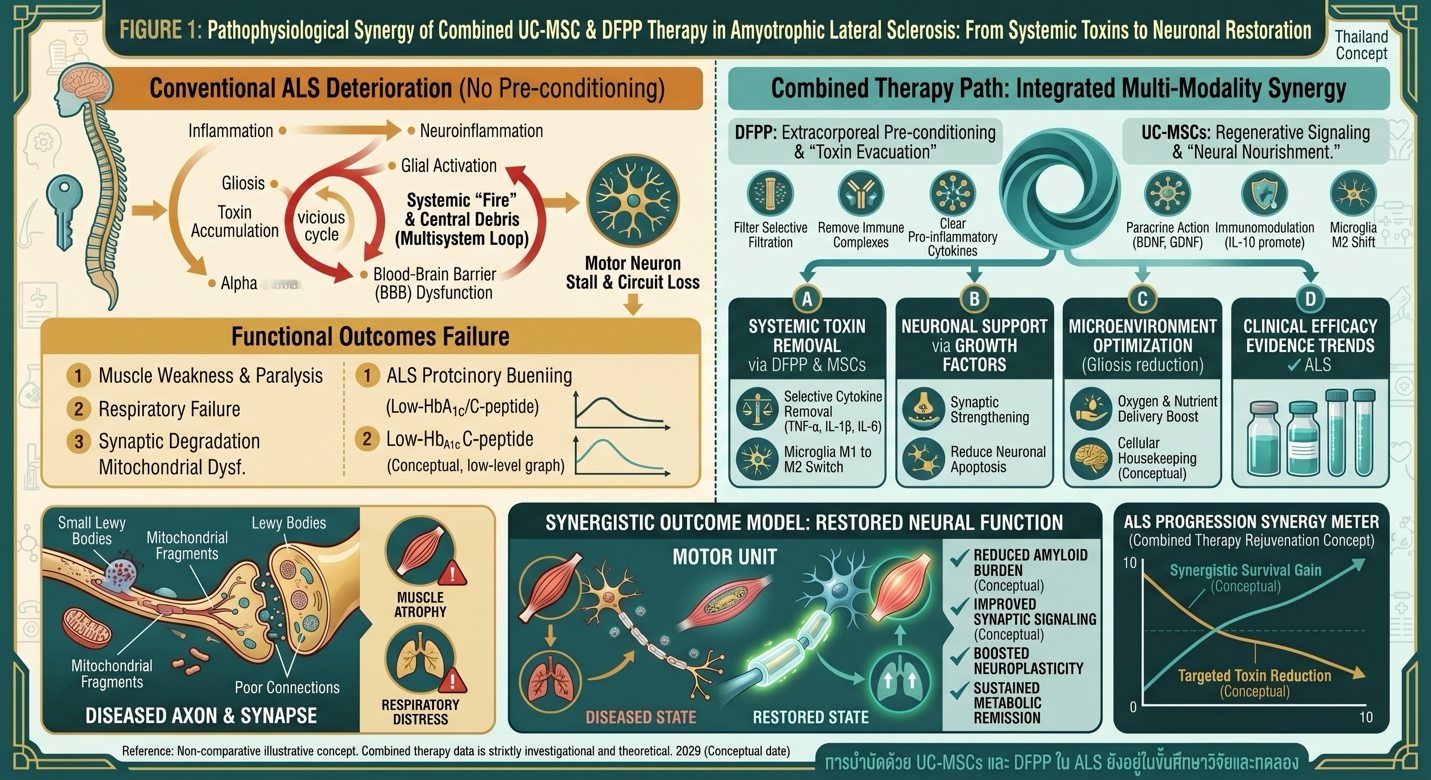
Amyotrophic lateral sclerosis (ALS) is a fatal progressive neurodegenerative disease characterized by degeneration of upper and lower motor neurons resulting in death. A progressive neurodegenerative disorder, which is associated with advancing motor symptoms, respiratory failure and death within 3–5 years of diagnosis. While supportive care is much improved, treatment options remain limited, and existing agents (eg: riluzole and edaravone) have only modest effects on the disease course.
Since the pathophysiology of ALS is multifactorial involving neuroinflammation, oxidative stress mitochondrial dysfunction and immune dysregulation, there has been great interest in non-pathway specific therapies able to more broadly target the disease milieu; MSCs have by virtue of their broad immuno-modulatory effects also been evaluated along complementary avenues such as extracorporeal blood purification methods including DFPP.
Pathophysiology of ALS and Therapeutic Targets
A greater understanding of the underlying biology of ALS is pushing a paradigm shift to recognize ALS as multisystem disorder that incorporates neuronal degeneration PLUS glial activation and systemic inflammation and immune dysregulation. Neurotoxic proinflammatory cytokines, such as TNF-α or IL-1β or IL-6 produced by activated microglia and dysfunctional astrocytes.
Figure A: ALS Progression Healing Meter: Demonstrating the positive trend in motor function improvement and cellular microenvironment restoration following integrated regenerative treatment
In analogy, increased plasma immune mediators and systemically moreover peripheral inflammation are linked to disease progression, suggesting that ALS also bears centralized and systemic pathophysiological processes. This dual-component pathology provides a rationale for the investigation of combinations which target both the CNS and peripheral immune compartment.
UC-MSCs: Biological Mechanisms in ALS
Also, UC-derived mesenchymal stem cells (UC-MSCs), are being widely studied because of their low immunogenicity, high proliferation ability and strong paracrine activity. The primary role of MSCs transplant mechanistically differs from that of neural stem cells as they really don’t regenerate and replace neurons. Instead their therapeutic potential is primarily through modulation of the disease microenvironment.
Key mechanisms of UC-MSCs include:
LinksTau-28 The broad action of innate and adaptive immune effectors, many of which collectively suffice to incite inflammatory responses (often by multiple representatives within the same interleukin subclass), especially pro-inflammatory cytokines, is vital for MSCs as it merely offsets/modifies/augments distinct regulatory/reduced inflammatory-coming relationships, distinct from most stimuli/proven immodulalators. This is a two-fold process, inhibition and promotion.
Secretion of growth factors (e. g., BDNF, GDNF and VEGF) Paracrine signalling
Neuroprotection Low to None Apoptosis and Oxidative Stress
Microglia with altered pro-inflammatory (M1) and anti-inflammatory (M2) state.
In particular, there is evidence that MSC treatment prolongs life and delays disease progression in ALS models. In dermatology, the early phase clinical trials indicate upfront tolerable safety profiles but efficacy results have been mixed.
Double-Filtration Plasmapheresis (DFPP): Mechanistic Rationale
DFPP is an extracorporeal blood-purification method that separates plasma and selectively removes high-molecular-weight substances, such as immune complexes, autoantibodies and inflammatory mediators.
For DFPP, the possibility of a notable selective filtration appears, which can not be done with traditional PEx or greatly much less with plasma substitute as well as for that reason risk. DFPP has been utilized for diverse indications including autoimmune inflammatory disorders, hyperlipidemia, and post-transplant complications or other conditions.
Reduce circulating inflammatory cytokines
Remove pathogenic plasma components
Modulate systemic immune activation
Cool it down and tune the biological environment for cell therapies
However, while it remains investigational for ALS (and data from clinical studies supporting its use is much more scant),
Analyzing Locational Effects: Policy Justification Against the Shadow of UC−MSCs and DFPP
A synergistic therapeutic rationale for UC-MSCs + DFPP combination therapy was hypothesized as follows.
A possible explanation is that DFPP decreases systemic inflammation and could potentially provide better situation to the host.
UC-MSCs support neuron survival and repair through their regenerative signalling
The multitargeted molecular therapeutic strategy exemplifies a fundamental transition in regenerative medicine from mono-modality to integrated multi-modal interventions that simultaneously target systemic and local mechanisms of disease.
Theoretically, immune preconditioning with DFPP may improve MSC-based therapy. But then this type of synergy is still hypothetical and has to be tested in controlled trials.
Conclusion
The strategy of ADR with UC-MSCs and DFPP in ALS can be a new, biologically feasible regenerative medicine hypothesis. Treating both systemic inflammation and local neurodegeneration with such a dual mechanism matches the recent ideas that ALS should be viewed as a multifactorial disease.
Although several hypothesised mechanisms and early safety data suggest efficacy, clinical data are limited. Further work with well-designed randomized trials is required to demonstrate efficacy, precision and define optimal treatment protocols together with the patient populations that they are likely to benefitthe most from.
Therefore, at this point of time, this process must be interpreted as investigational and thus monitored cautiously whilst being applied in the clinic with due diligence.