Why this topic needs a slower, more honest explanation
If someone searches Plant-Based Stem Cell Therapy for Back Pain, they are usually hoping for something more biological than painkillers and more restorative than temporary relief. That hope is understandable. Low back pain is extremely common and remains a leading cause of disability worldwide, which is exactly why regenerative ideas attract so much attention.
I’m reading this topic mainly through the lens of chronic low back pain, because your secondary keyword is stem cell injections for low back pain. That matters because “back pain” is not one diagnosis. Chronic primary low back pain, discogenic pain from degenerative disc disease, facet-related pain, nerve-related pain, and muscular pain are not interchangeable problems, and they should not be discussed as though one regenerative approach applies equally to all of them.
What “Plant-Based Stem Cell Therapy” usually means
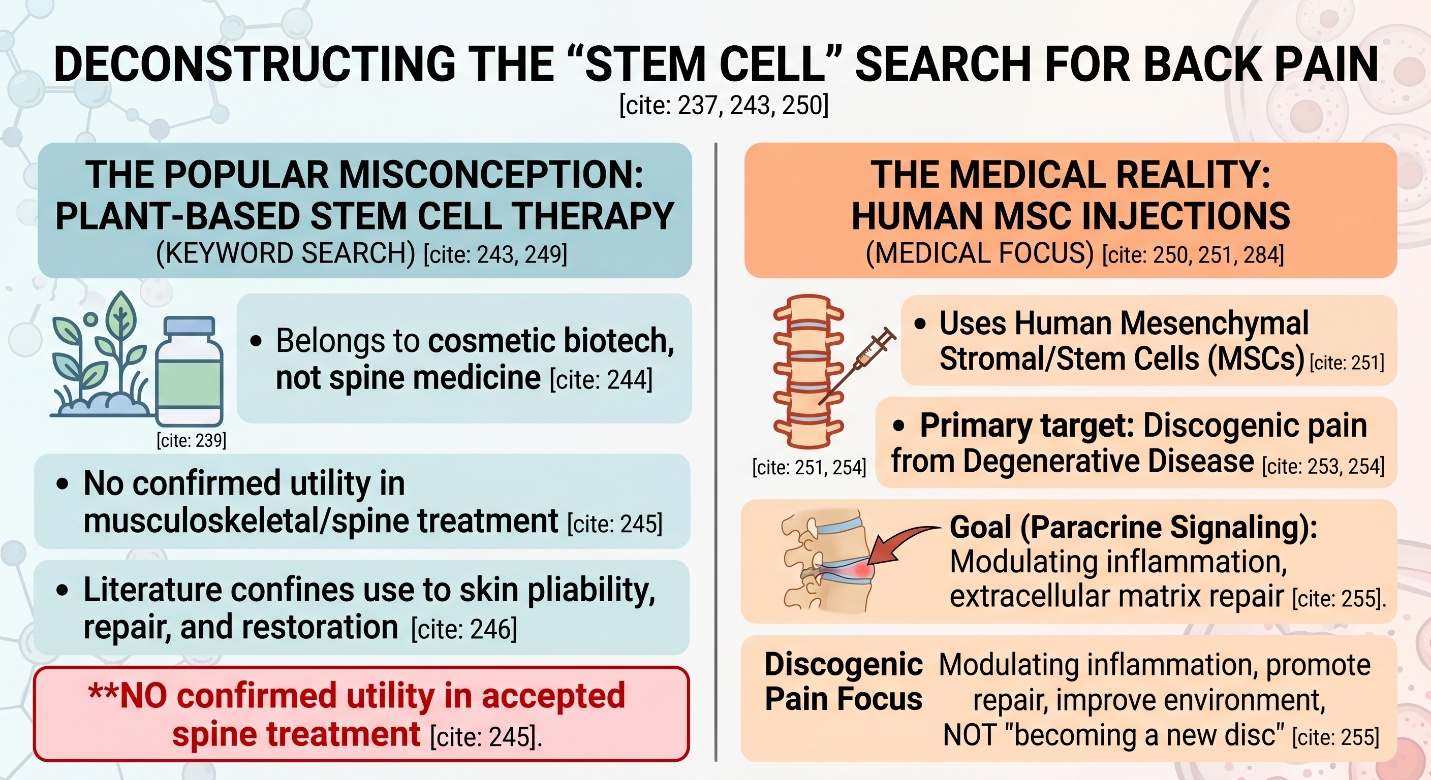
In most literature, it belongs more to cosmetic biotech than spine medicine.
While Plant-Based Stem Cell Therapy suggests a medical orientation, the published literature confines plant stem cells to predominantly cosmetic and personal-care biotechnology applications with no confirmed utility in any accepted musculoskeletal or spine treatment. An overview of plant stem cells in cosmetics published in 2025 details various applications for plant stem cells related to skin pliability, repair, and restoration, and antioxidant protection, as well as the development of new ingredients [62]. This is a field of research, but it does not equate to guideline-based management for chronic low back pain.
That does not make the phrase meaningless, but it does narrow what it can honestly mean.
So the first layer of honesty here is simple: Plant-Based Stem Cell Therapy is a much stronger keyword than it is a recognized back-pain treatment category. In back pain medicine, the real regenerative discussion is not about plant stem cells. It is about human mesenchymal stromal/stem cells, usually in the context of disc degeneration and intradiscal injection research.
What stem cell injections for low back pain are actually studying
The main target is usually discogenic pain from degenerative disc disease
When discussions of stem cells for low back pain arise, it has typically been the topic of intradiscal mesenchymal stromal/stem cell injections as a treatment for degenerative disc disease. The biologic rationale is that their paracrine signaling may have a role in modulating inflammation, promoting extracellular matrix repair, and improving the disc environment rather than just “becoming a new disc.” This is one of the principal regenerative pathways currently under investigation in spine care, found in both recent reviews and guideline updates.
This is a much narrower and more specific question than most marketing suggests
That distinction matters because many people read “back pain” and imagine a universal spine solution. The current evidence does not support that. Even the more optimistic papers are generally focused on selected adults with lumbar degenerative disc disease and discogenic low back pain, not on every person with back pain, scoliosis, instability, spinal stenosis, or mixed mechanical pain.
What the evidence actually shows right now
There are encouraging signals, but they are still modest and still incomplete.
A recent systematic review and meta-analysis of 12 randomized trials found that intradiscal MSC injection is “safe with a small but marked improvement in pain and function” for degenerative disc disease, while reiterating the need for larger and higher-quality studies. In a more general 2025 evidence-based guideline update from the American Society of Interventional Pain Physicians, Regenerative therapies for chronic low back pain were also categorized as active areas rather than settled routine care. Which is likely the most just interpretation of the area: alluring enough to trail genuinely, however not fully developed enough to feverishly sell.
Standard care still has the stronger everyday evidence base.
This is the second layer that often gets lost. WHO’s guideline for chronic primary low back pain recommends non-surgical approaches such as education, exercise programs, some physical therapies, psychological therapies, and NSAIDs in appropriate settings. That does not mean regenerative medicine is irrelevant. It means the best current evidence for most people with chronic low back pain still begins with structured, non-surgical care rather than jumping straight to an injection.
Where regulation and clinic claims start to matter
A stem cell product being sold is not the same thing as a treatment being established
The FDA says regenerative medicine therapies have not been approved for any orthopedic condition, including disc disease and back pain, and its consumer alert also warns patients about products marketed as stem cells or exosomes before adequate proof of safety and effectiveness exists. That is a very important line in this whole conversation. It does not mean all stem cell research for low back pain is useless. It means the field is still investigational, and commercial availability should not be mistaken for proof.
The best clinic conversation should sound more precise than promotional.
A credible discussion about stem cell injections for low back pain should explain the diagnosis clearly, describe why discogenic pain is being suspected, specify what cell product is being used, discuss what level of evidence exists, and acknowledge that current outcomes are usually described as modest improvements, not guaranteed correction of chronic back pain. If the conversation sounds more certain than the trials do, that is usually a sign that the marketing is ahead of the science.
The most honest conclusion
A systematic review and meta-analysis of 12 randomized studies reported intradiscal MSC injection is “safe with a small to moderate improvement in pain and function” for degenerative disc disease, but emphasized the need for larger and better-quality studies. An additional, overarching 2025 evidence-based guideline update from the American Society of Interventional Pain Physicians also classified Regenerative therapies as active areas for chronic low back pain rather than settled routine care. Which is probably the fairest take on the place: too attractive to actually walk there, but not developed enough to justify a purchasing frenzy.
Frequently Asked Questions
1) Is Plant-Based Stem Cell Therapy a real treatment for back pain?
Not as a recognized spine-treatment category. In the literature, plant stem cells are mainly discussed in cosmetics and personal-care biotechnology, not as standard therapy for low back pain.
2) What do “stem cell injections for low back pain” usually mean?
Usually, they refer to intradiscal mesenchymal stromal/stem cell injections being studied for degenerative disc disease and discogenic low back pain.
3) Do stem cell injections for low back pain work?
Current randomized-trial evidence suggests they may offer modest improvements in pain and function in selected patients, but larger and more standardized trials are still needed.
4) Are stem cell injections for back pain FDA-approved?
No. The FDA says regenerative medicine therapies have not been approved for orthopedic conditions such as disc disease and back pain.
5) What still matters most for chronic low back pain?
For most people, WHO still recommends a non-surgical foundation that includes education, exercise, some physical therapies, psychological approaches, and appropriate medication use.