Rheumatoid arthritis (RA) is a chronic systemic autoimmune disease that manifests as long-term synovial inflammation, progressive articular destruction and loss of function. By contrast with other inflammatory arthritis, rheumatoid arthritis causes dysregulated immune responses that abnormally target synovial tissues rather than simply affecting joint discomfort, but the resulting inflammation may drive the destruction of cartilage and erode bone leading to pain, stiffness and impaired mobility. In more advanced or inadequately controlled cases, systemic inflammation may be associated with some extra-articular complications including cardiovascular and pulmonary involvement.
Although symptoms control has become a reality with the advent of standard pharmacological therapies, a subset of patients continues to have an insufficient response or intolerance to treatment, frequent flares or long-term functional impairment. Such clinical restrictions have promoted greater interest in regenerative medicine, including studying umbilical cord-derived mesenchymal stem/stromal cells, called UC-MSCs.
In the stem cell therapy Thailand perspective, UC-MSCs are already being studied for their possible immunomodulatory and tissue-supportive effects. They should be viewed as an evolving adjunctive treatment option, and not a substitute for standard rheumatologic care. Your main scientific interest is whether UC-MSCs may regulate excess immune activity and inflammatory signaling — generating a better biological environment for normal joint function.
Pathophysiological Mechanisms and Clinical Manifestations
Rheumatoid arthritis involves a complicated interplay of genetic predisposition, environmental initiation, aberrant immune regulation and chronic inflammation in the joint synovium. Impairment of immune tolerance with activation and expansion of autoreactive immune cells to attack healthy joint structures is a key feature of the disease.
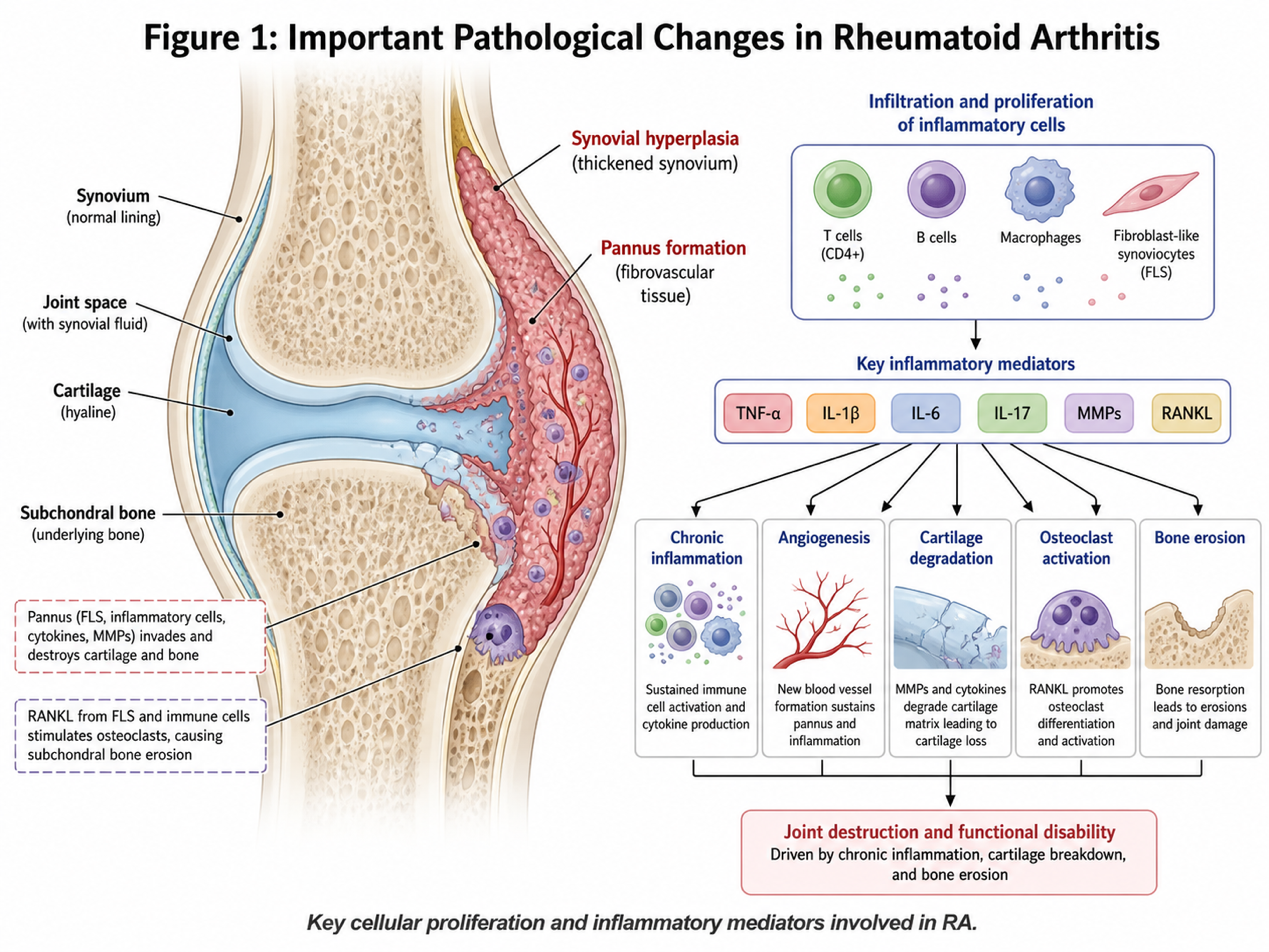
At the molecular level, activated T cells, B cells, macrophages, dendritic cells and fibroblast-like synoviocytes accumulate in the synovial membrane. These cells release proinflammatory cytokines, including tumor necrosis factor-alpha (TNF-α), interleukin 6 (IL-6), IL-1β, and IL-17. Persistent cytokine action results in synovial hyperplasia and development of an invasive inflammatory tissue called pannus, that gradually erodes cartilage and bone.
The growth of the pannus involves cartilage matrix degradation by the action of matrix metalloproteinases and focal bone resorption due to activation of osteoclasts. Clinically, a patient can present with symetric swelling of joints, prolonged morning stiffness, tenderness (tender hand/wrist), decreased grip strength, fatigue and decreased range of motion. Long-term, uncontrolled or inadequate controlled disease may cause joint deformity, disablement and less independence in activities of daily living.
RA is a systemic disease, however, that inflammation may not be limited to the joints. Fatigue, anemia, and vascular inflammation lead to higher cardiovascular risk in patients. These findings underscore the need for disease-modifying treatments targeting local joint inflammation as well as systemic immune activation.
Rheumatoid arthritis (RA) is an autoimmune, chronic inflammatory disorder that mainly affects the joints. It is associated with several clinical features due to systemic involvement and pathophysiological mechanisms.
Figure 1: shows a simple depiction of some of the important changes that occur in RA leading to proliferation of cells and inflammatory mediators involved in RA.
Conventional Treatment Modalities and Limitations
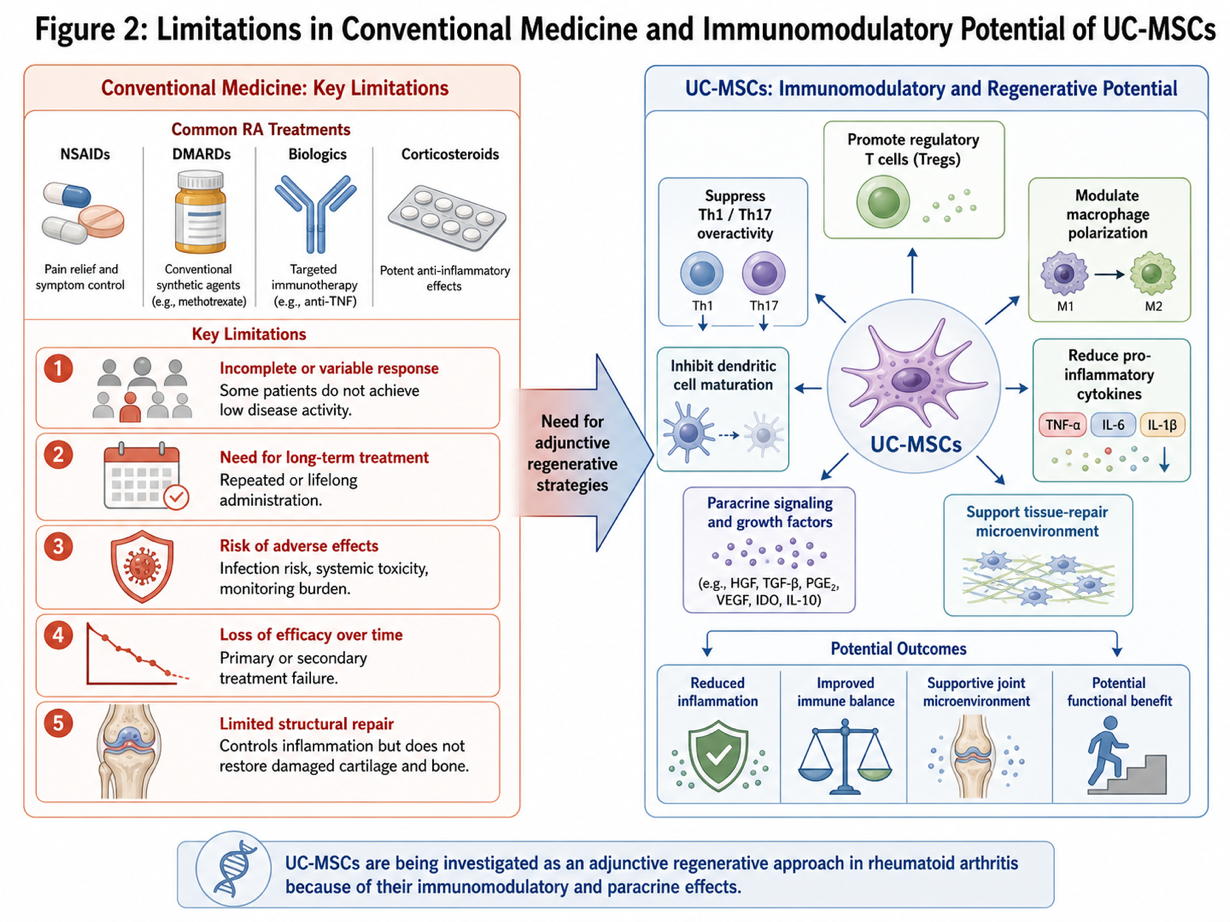
Rheumatoid arthritis is currently managed with a stepwise and tailored approach. Management may include non-steroidal anti-inflammatory drugs, corticosteroids, conventional synthetic disease-modifying antirheumatic drugs such as methotrexate, biologic agents and targeted synthetic therapies. These interventions remain the corner stone of evidence‐based rheumatoid arthritis care and have dramatically improved long‐term outcomes for many patients.
However, several limitations remain. Some patients have a primary non-response pattern where adequate disease control is not reached following treatment initiation. Some develop secondary loss of response, where an initially effective therapy is rendered less effective over time. Immunosuppression over the longer-term may predispose to infectious diseases requiring close monitoring especially in older patients or those with multi-morbidity.
Moreover, one more limitation is that traditional therapies mainly focus on targeting immune pathways in order to stop the disease course. Although this can preserve function and alleviate symptoms, it may not restore cartilage, subchondral bone or soft tissue architecture once substantial structural compromise has occurred. In retrospective studies among patients with established joint destruction, residual mechanical pain and disability may still occur in the setting of controlled inflammatory biomarkers.
Such challenges have prompted researchers and clinicians to consider adjuctive biological approaches that could promote enteric immune regulation and tissue repair signaling. With this inMind, UC-MSCs have emerged as an important target for research in regenerative medicine.
UC-MSCs For Therapeutic Purposes and Immunomodulatory Role
Umbilical cord derived mesenchymal stem/stromal cells have drawn scientific interest because of their well-documented proliferative capacity, low immunogenicity, paracrine activity and ability to interact with immune cells. Compared with certain adult tissues tissues used to derive MSCs, UC-MSCs are more openly described for their availability, juvenile biological signature and potential fitness for clinical allogeneic applications.
The therapeutic potential of UC-MSCs in rheumatoid arthritis could integrate more than their immunomodulatory action on T cells. These cells can modulate inflammatory environments via direct cell–cell contact and also releasing bioactive molecules. Instead of primarily serving as progenitor or replacement cells, UC-MSCs are thought to act through paracrine signaling, by releasing cytokines, growth factors, extracellular vesicles and other mediators that could modulate the immune response.
An important mechanism that has well-defined immune checkpoints includes the enhancement of regulatory T cell function. They are responsible for maintaining immune tolerance and negatively regulating excessive inflammatory responses. UC-MSCs are also thought to contribute to inhibiting the activity of overactive Th1 and Th17 pathways that mediate pathogenesis in rheumatoid arthritis. UC-MSC-derived factors including indoleamine 2,3-dioxygenase and prostaglandin E2 have also been linked to suppression of dendritic-cell maturation and modulation of immune-cell activation.
Besides immune regulation, UC-MSCs may also participate in the tissue repair signaling pathways through specific growth factor secretions that directly alter chondrocytes, synovial cells and angiogenic activity as well as optimize extracellular matrix homeostasis. But that potential must be shown with care. There is little current evidence supporting claims that UC-MSCs can routinely regenerate damaged joints or cure rheumatoid arthritis. A more scholarly interpretation is that UC-MSCs may promote a more favorable inflammatory microenvironment that facilitates maintenance of tissue and function in selected patients.
Figure 2: Limitations in Conventional Medicine and Immunomodulatory Potential of UC-MSCs
Development of MSC Research and Clinical Application in Thailand
Thailand is gaining visibility in regenerative medicine, medical wellness and cell-based clinical services. Stem cell Therapy Thailand is a result of a growing investment into bio-technology infrastructure, specialized laboratory systems and physician-led regenerative medicine programs.
Future development of UC-MSC applications in RA will hinge on several major areas, including standardised cell culture and expansion in defined media conditions; consistency of quality-control measures, sterility analysis and potency recalculation is strategic for clinical coordination to be established; the testing will therefore emphasise selection criteria to optimise therapeutic potential within clinical trials as well as a systemic but structured follow-up upon treatment. These components are crucial to ensuring that regenerative medicine translates appropriately from basic sciences to clinical practice.
More personalized therapeutics protocols for UC-MSC research in Thailand In the future Advanced programs may use separate treatment models based on an assessment of disease duration and damage index, as well as medication history, inflammatory markers, autoantibodies status, imaging outcome (if necessary), functional Scores and comorbid Conditions instead of applying conventional management strategies for all individuals. This type of patient-specific evaluation may better delineate those patients who are ideal candidates for regenerative support and promote consistency for how to quantify their outcomes.
Other aspects of emerging research interest include MSC priming, extracellular vesicle-based approaches, repeated dosing and biomarker-guided monitoring. They may also help guide future applications, but large controlled clinical studies are still needed to prove safety and efficacy as well as optimal dosing, durability of response, and longer-term outcomes.
The responsible ime in Thailand should be the balance between progress and enlightening patients about transparency. Patients must be clearly informed that UC–MSCs remain an experimental treatment option and should not be presented as a potential solution of established rheumatology care.
Conclusion
Rheumatoid arthritis (RA) continues to be an intricate autoimmune disorder characterized by permanent synovial joint inflammation, immune system dysfunction, articular structures damage, and jaw bone erosion for the affected joints accompanied by systemic consequences. While conventional therapies have dramatically advanced the management of disease, some patients remain inadequately treated due to the limited efficacy and safety of these agents, prolonged drug exposure along with persistent damage to joints.
Due to their potential immunomodulatory and paracrine signaling properties, UC-MSCs embody an emerging area of regenerative medicine. Their potential roles may include regulation of inflammatory immune responses in RA, promotion of the activity of regulatory T cells (Tregs), suppression of exuberant cytokine signaling, and creation of an environment more amenable to physiological tissue repair.