Degenerative disc disease (DDD) is a widespread spinal condition characterised by chronic back pain, decreased mobility, stiffness, nerve-root irritation and overall diminished health-related quality of life. Despite the use of the word “disease”, disc degeneration is frequently more of an incremental biological and structural process than a one-time event. It is characterized by stepwise alterations in the intervertebral disc including dehydration, aberrant extracellular matrix composition, loss of disc height, annular degeneration, inflammatory cellular infiltration and loss of mechanical function.
UC-MSCs in degenerative disc disease have become an emerging area of scientific interest in modern regenerative medicine. Historically, umbilical cord-derived mesenchymal stem/stromal cells (UC-MSCs) have been studied regarding their immunomodulatory, anti-inflammatory, and paracrine signaling properties. They are unlikely to be a panacea for disc degeneration, but rather provide a more favourable biological environment for tissue repair, inflammation regulation and functional improvements.
Biological Basis of Degenerative Disc Disease
Intervertebral disc tissue is primarily formed by the nucleus pulposus, annulus fibrosus and cartilaginous endplates. When it comes to a healthy spine the disc is structurally designed to absorb mechanical load and preserve motion. The reason for reduction of the proteoglycans in nucleus pulposus is complex, but with aging, repetitive stress, injury/ inflammation and genetic–metabolic imbalance, a low quantity of water content in addition to loss of proteoglycan concentration may take place. This depletes its capacity for shock absorption and could lead to pain, stiffness, and mechanical instability.
A hostile microenvironment also influences disc degeneration. The intervertebral disc has a low blood supply, low oxygen tension, acidic pH and poor nutrient diffusion. These factors compete against natural repair, which is already challenged and may hinder the survival or viability of exogenously added therapeutic cells once injected. A systematic review in 2025 stated that stem-cell-based approaches to intervertebral-disc degeneration remain promising but fraught with immense translational challenges, particularly because clinical improvement is often not well correlated with strong imaging evidence of true structural disc regeneration.
Limitations of Conventional Management
What Is the Conventional Treatment for Degenerative Disc Disease? Pain medication, anti-inflammatory drugs, physiotherapy, lifestyle modification and also agencies like epidural steroid injections nerve obstructs and sometimes surgery are common. These approaches may have clinical utility, particularly in the context of symptom management and functional support. They tend to somehow be more about reducing pain or mechanical compression than restoring the biological quality of a degenerating disc.
Although pain medication will eliminate pain, it does not help rebuild the disc matrix. Corticosteroids injections can reduce inflammation in the short-term, but do nothing to reverse age-related changes associated with disc degeneration. When nerve compression, instability and neurological deficit are severe, surgery becomes imperative; however, it is an invasive procedure with recovery time. Hence, regenerative medicine has emerged as a potential adjunct for patients looking for a more biologically based treatment approach to spinal care.
It is also essential to make regulatory restrictions clear. From the U.S. FDA: Regenerative medicine products, including stem cells and exosomes, are not approved in the United States to treat orthopedic conditions (such as disc disease, back pain, neck pain or treatment for other musculoskeletal diseases). This is not the regulatory approach in ever country, but it does emphasize the need for patient education, safety monitoring and responsible clinical claims.
UC-MSCs and Regenerative Mechanisms
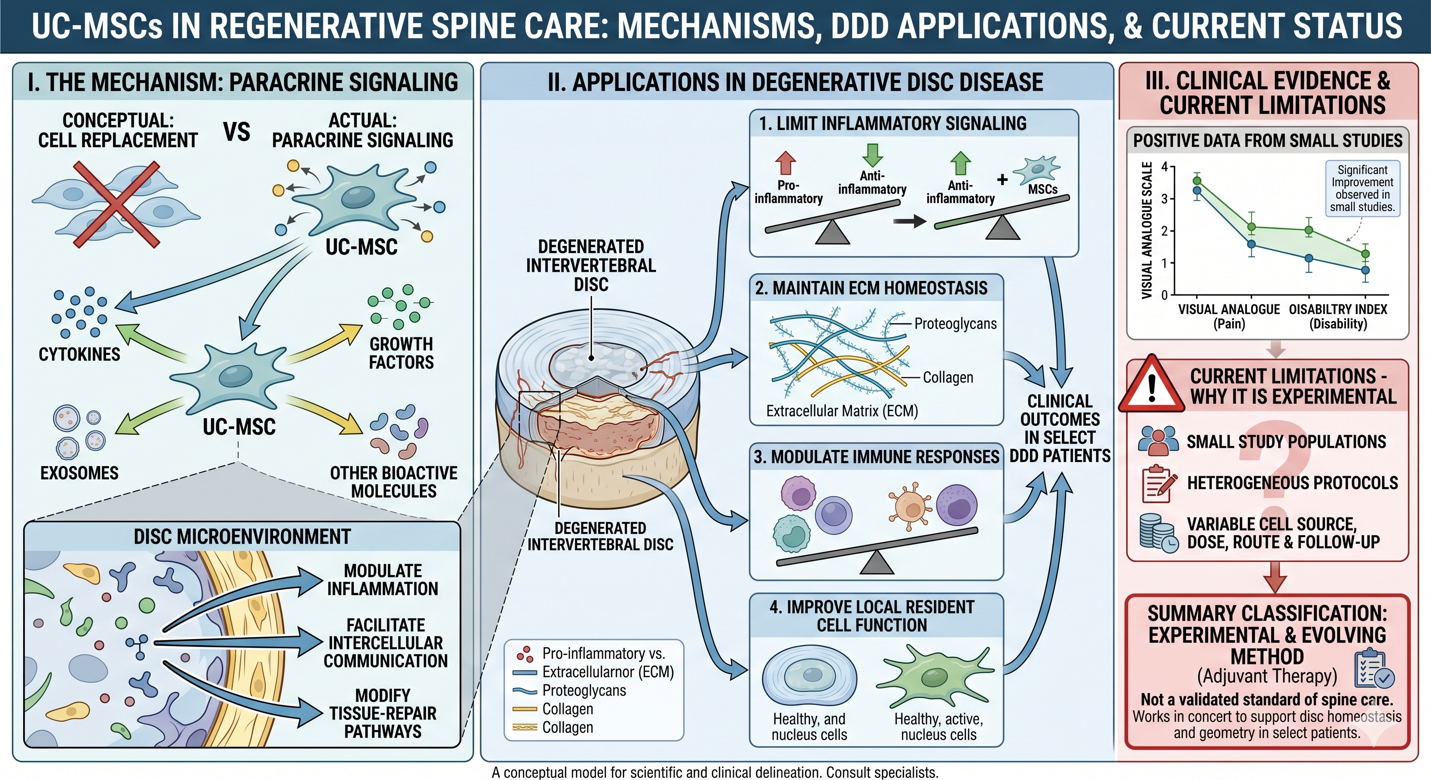
In regenerative ways, UC-MSCs are being studied as they have unique biological mechanisms. Instead of providing new cells as a replacement, MSCs seem to do their work mainly through paracrine signaling. This means that they secrete cytokines, growth factors, exosomes and other bioactive molecules that may modulate inflammation, facilitate intercellular communication and modify tissue-repair pathways.
In degenerative disc disease, UC-MSCs could contribute by limiting inflammatory signaling within the disc microenvironment, maintaining extracellular matrix homeostasis, modulating immune responses, and improving local resident disc cell function. There were significant improvement in pain and disability scores, within human mesenchymal stem cells studies like reduction of Visual Analogue Scale and Oswestry Disability Index outcomes for degenerative disc disease. But the current evidence is still limited by small study populations, heterogeneous protocols and variability in cell source, dose, route of administration and duration of follow-up.
Due to these uncertainties UC-MSCs should be considered more as an experimental and evolving regenerative method rather than a validated alternative standard of spine care. In the most scientifically correct framing, UC-MSCs may serve as adjuvant therapy that works in concert with or to support disc microgeometry and inflammatory homeostasis in select patients while additional clinical studies are performed to better delineate efficacy and long-term safety.
Figure 1: Conceptual Framework of UC-MSCs in Degenerative Disc Disease (DDD).
Individual: Recovery Time After UC-MSCs for Degenerative Disc Disease
The recovery time following UC-MSCs for degenerative disc disease patients can differ from one person to another. It is based by on the severity of disc degeneration, number of affected levels, whether there is nerve compression or not, baseline pain level, age and general health, quality of rehabilitation done and injection route. Patients sometimes report transient soreness, stiffness or mild discomfort for a few days after a spinal-related injection.
Changes may take place a few weeks into treatment, usually in spite of variations between individuals; with discomfort sensitivity, rigidity or movement confidence are normally some of the first changes during this stage. Functional changes that are inductively meaningful are usually evaluated over a range of 2 to 6 months based on the gradual process of biological signaling, and tissue-environment alterations. In chronic degenerative disc disease, follow-up durable to response, physical function (6 to 12 months where appropriate) and imaging should be performed at longer-term.
A responsible recovery framework might include:
| Timeframe | Clinical Focus |
| First 24–72 hours | Monitoring for soreness, headache, fever, neurological change, or injection-site reaction |
| First 1–2 weeks | Rest, light movement, avoidance of heavy spinal loading |
| Weeks 3–8 | Gentle rehabilitation, pain tracking, mobility observation |
| Months 2–6 | Main period for evaluating functional improvement |
| Months 6–12 | Longer-term monitoring and outcome assessment |
Rehabilitation and Patient Selection
But UC-MSC-based care doesn’t need to be the only solution. Prehabilitation still works wonders for core stability, postural issues, maintaining flexibility and normal gait mechanics as well as spacing loads away from the spine. Medical Management: Programs should include MRI review, neurological assessment, pain scoring and functional evaluation, medication history or contraindications;
The ideal candidate is that one with well-diagnosed degenerative disc disease, chronic clinical signs resistant to conservative treatment, and not imminently in need of surgery. Those with severe spinal instability, major neurological deficit, active infection, tumor or advanced structural collapse may be treated using different medical or surgical pathways.
Conclusion
Real-time Analysis of Soft Tissue Regeneration UC-MSCs for Degenerative Disk Disease UC-MSCs have been on the rise as a regulated regenerative medicine option, targeting inflammatory modulation in paracrine cell signaling and providing disc microenvironment support. Some early clinical research implies there could be pain and function improvement for some patients, however, much stronger evidence is needed to prove long-term efficacy, structural disc repair are achievable end-points along with the most appropriate dose and selection of patients.