Spinal disorders are common and among the most frequent causes of chronic pain, reduced activity, neurological impairment and long-term disability around the globe. Degenerative disc disease, chronic low back pain, spinal inflammation as well as spinal cord injury can put a major hindrance on movement and activities of daily living. Spinal disease is rarely a singular structural problem in many patients It frequently receives stronger interest due to inflammation, nerve irritation, disc degeneration, less muscle strength and circulation reduction with specially situational changes at local tissue microenvironment.
Recently, UC-MSCs injection into the spinal region as adjunctive therapy for certain spinal diseases has gained attention in contemporary regenerative medicine. Umbilical cord-derived mesenchymal stem/stromal cells (UC-MSCs) have been proposed as an attractive and powerful cell source for various clinical applications due to their immunomodulatory, anti-inflammatory, paracrine and tissue-supportive characteristics. They are not intended to be a replacement for standard spine care, surgery, rehabilitation, or neurology management. Instead of spinal UC-MSCs injection itself, it might be a therapeutic part of medically oriented regenerative regimen which possibly assist with cellular signalling, inflammation modification and augmentation/repairing function.
Patient education requires an understanding of recovery time after spinal stem cell injection. Although recovery should occur, it is not immediate and outcomes relate to each individual diagnosis, degree of tissue injury, method of delivery[to a muscle], rehabilitation regime and the age/health/duration of disease. As with clinical expectations, the current evidence continues to evolve, and ought to be tempered with caution.
Neurophysiological Basis of Spinal Degeneration and Injury
They may occur due to mechanical, inflammatory, neurological and age-related biological mechanisms. Degenerative disc disease is a process whereby the intervertebral disc gradually becomes dehydrated, and less able to maintain its extracellular matrix architecture as well as its shock-absorbing characteristics. It contributes to the reduction of disc height, annular tears, nerve irritation, stiffening of your spine and ultimately leads to chronic back pain. Methods: A recent systematic review highlighted that degenerative disc disease continues to be one of the most common diseases leading to chronic low back pain and global disability; however, is hampered by a methodologically heterogeneous clinical evidence base for stem-cell-based disc regeneration (SCDR).
In the spinal cord injury, mechanical trauma is followed by a secondary injury cascade characterized by inflammation; ischemia; oxidative stress; excitotoxicity; and axonal degeneration, as well as disruption of the microenvironment in the injured spinal cord. These secondary processes may go on for months after the initial trauma and world then combine to cause permanent functional is a biomechanics. The biological properties of MSCs in SCI, including anti-inflammatory, immunoregulatory, pro-angiogenic and tissue-supportive effects are reviewed; the mechanisms underlying these properties and their clinical application remain inconclusive.
That is why spinal regenerative medicine has been trending towards targeting the tissue environment, not just the structural defect that is visible. Factors Maybe Affecting Pain, Stiffness, Weakness and Reduced Function (Inflammation, Neural Sensitization, Cellular Stress, Impaired Blood Supply by Ischemia or Infarction which Limits Repair Signaling. So a biologically focused approach seeks to enhance the setting in which recovery may take place.
Limitations of Conventional Spinal Treatments
However conventional spinal therapy is still critical and may involve pain medication anti inflammatory medications physiotherapy steroid injections nerve blocks decompression surgery or other interventions, spinal fusion, disc replacements, neurological rehabilitation. These options can be appropriate and effective depending on the diagnosis and severity of the sickness.
There are some inherent limitations to traditional spinal therapy. Medications for pain may relieve symptoms but do not necessarily halt progression of genital degeneration. Although steroid injections might reduce inflammation temporarily, they do nothing to restore disc structure and spinal cord tissue. Operative intervention is reserved for the more severe cases in which there is a significant level of compression, instability, deformity or progressive neurological deficit; however surgery always comes with significant risks due to its invasive nature and requires careful consideration regarding rehabilitation post-op.
Clinical studies of stem-cell-based approaches for degenerative disc disease indicate modest improvements in pain and disability, but imaging evidence of true biological repair remains limited. The disc is also difficult to target, as it is avascular and biologically hostile, which may decrease cell survival and regenerative activity.
Regulatory caution is also important. According to the U.S. FDA, regenerative medicine products such as stem cells and exosomes are not approved for orthopedic conditions (disc disease, back pain, neck pain, or shoulder pain), neurological disorders (stroke, Parkinsons disease, ALS / Lou Gehrings disease or multiple sclerosis). This is not true for every countries regulatory pathway but reflects the key features that underpin an informed consent process, oversight by a physician, and realistic clinical communication.
The Injection of UC-MSCs and their Regenerative Mechanisms
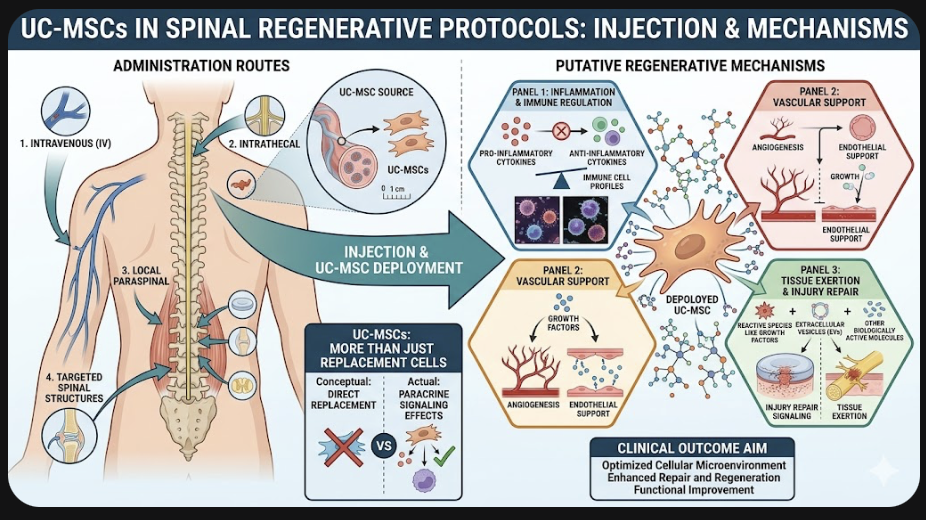
Spinal UC-MSCs injection — clinical use of umbilical cord (UC) derived mesenchymal stem/stromal cells in a spinal-related regenerative protocol. Administration can be intravenously (IV), intrathecally, through local paraspinal injection or targeting injections around selected spinal structures depending on the particular condition and clinical judgment of provider.
UC-MSCs are not mere “replacement cells.” Their putative paracrine signaling effects lie at the root of many regenerative medicine models. This has been suggestive that the cells will release pro- inflammatory cytokines, reactive species like growth factors and extracellular vesicles as well as other biologically active molecules for regulation of inflammation and immune activity such as tissue exertion, vascular support, and injury repair signaling.
Figure 1: Administration Routes and Putative Regenerative Mechanisms of UC-MSCs in Spinal Care
A medically responsible recovery timeline may be described as follows:
| Timeframe | Possible Recovery Focus |
| First 24–72 hours | Monitoring for procedural discomfort, headache, fever, injection-site reaction, fatigue, or neurological changes |
| First 1–2 weeks | Rest, inflammation monitoring, gradual return to light activity, and avoidance of excessive spinal loading |
| Weeks 3–8 | Early functional observation, gentle rehabilitation, pain and mobility tracking |
| Months 2–6 | Main period for evaluating changes in pain, stiffness, function, walking tolerance, or neurological signs |
| Months 6–12 | Longer-term monitoring, especially for spinal cord injury or chronic neurological conditions |
This timeline should not be interpreted as a guarantee. Some patients may experience earlier improvement, while others may require more time or may not respond significantly. Recovery depends on disease severity, chronicity, baseline function, imaging findings, inflammation level, rehabilitation quality, and individual biological response.
Reviews of spinal cord injury research that utilize MSC detail combination approaches as rehabilitation, physical stimulus, biomaterial scaffolds and other means to aid the effects. The implications resonate with a key clinical principle: the clinical relevance of regenerative medicine may be potentiated by concomitant delivery of appropriately structured functional recovery programs rather than longevity-enhancing cellular therapies alone.
Hence one should clarify recovery time as a biological response, medical monitoring and rehabilitation process in clinical communication in practice. The injection may provide cellular milieu support but the movement retraining plus functional therapy converts biology to functionality.
Organisational Safety, Patient Selection and Clinical Supervision
Spinal procedures are always requiring the highest level of safety standards and patient selection because of the sensitive anatomical structures that may be involved. Patients should be carefully evaluated before spinal stem cell UC-MSCs injection, which may include medical history, neurological examination, medication review, infection-risk screening and imaging (MRI or others), baseline pain scoring, functional study and especially discussions about previous treatments.
Realistic outcome measures should also be part of clinical monitoring. Including in some instances, pain score, ambulation, range of motion, Oswestry Disability Index, neurological grading (examination), sensory testing, muscle strength assessment and quality-of-life scoring and followup imaging when relevant.
Possible adverse effects include but are not limited to: infection, bleeding, post-injection pain, headache (which can occur because of needle-related effects such as the local hematoma formation), immune system reaction, a temporary increase in symptoms and some neurological complication related to the procedure. Risk of contamination, unwanted inflammatory response, product inconsistency, or unsupported treatment claims may be broader safety concerns associated with the unproven nature of many regenerative medicine products. The FDA cautions specifically that unapproved regenerative products can pose serious risks including infections, tumor formation, neurological complications and immune system reactions.
From Epigenetics Toward a Personalized Spinal Regenerative Program
This could mean a more personalized future for spinal regenerative medicine. Instead of grouping every patient into one protocol, advanced programs might consider diagnosis type, MRI results, inflammatory markers, neurological status, pain pattern, age, metabolic health physical function and potential for rehabilitation.
Future strategies for degenerative disc disease might include enhanced cell delivery methods, biomaterial scaffolds, extracellular vesicles and approaches specifically targeted to modulate the microenvironment of the disc. A recent systematic review suggested that future advances may require disc niche engineering or support, as opposed to direct cell replacement.
Questions for future research on spinal cord injury include the optimal time window after injury, acute as opposed to chronic disease response, cell dose, route of administration (e.g., topical vs. systemic), use of repeated doses, combination with rehabilitation and ability to track objective neurological outcomes. The tailored model could help pinpoint which patients are optimal targets for this intervention and the length of time before monitoring recovery is appropriate.
Conclusion
The injection of spinal UC-MSCs is a new frontier in regenerative medicine for diseases involving chronic inflammation, disc degeneration, neurological injury and disrupted tissue repair signaling. The scientific rationale is based primarily on the immunomodulatory, anti-inflammatory, paracrine and tissue microenvironment-supporting functions of UC-MSCs.
On the other hand, injection of spinal stem cell UC-MSCs should be deliberated with caution. Do not refer to it as a certain science-based solution for back pain, disc degeneration, paralysis or spinal cord injury. Current evidence from various therapies offers potential clinical promise in selected areas but larger, more randomized studies are still needed for confirmation of efficacy, determination of timing and duration of recovery, and standardization of treatment protocols.
It is important to note that these gains were gradual, in particular the overseeding of the spinal cord with UC-MSCs. The monitoring of musculoskeletal spine patients may last weeks to months, while neurological recovery may need much longer with structured rehabilitation. The best model is that of a medically supervised personalized regenerative program that covers all aspects from accurate diagnosis, cell grade preparation, safe injection technique and rehabilitation planning to objective outcome monitoring.