Alzheimer’s disease (AD) is a progressive neurological disorder that gradually impairs memory, thinking ability, and everyday functioning. While conventional medications may temporarily ease symptoms, they do not address the underlying mechanisms driving the disease. This limitation has encouraged the exploration of regenerative medicine approaches, including mesenchymal stem cell (MSC) therapy, which aims to repair damaged brain tissue and slow or potentially alter disease progression.
The Importance of Paracrine Effects
Contrary to the common assumption that stem cells directly replace damaged neurons, much of the benefit from stem cell therapy arises from their paracrine activity. This refers to the release of bioactive molecules that influence surrounding cells. Stem cells secrete a variety of growth factors and neurotrophic substances that play essential roles in maintaining and restoring brain health.
These include brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), glial cell line-derived neurotrophic factor (GDNF), vascular endothelial growth factor (VEGF), and insulin-like growth factor (IGF). Together, these compounds help protect existing neurons, encourage the formation of new synaptic connections, and enhance the brain’s ability to adapt and reorganize—processes that are critical for learning and memory.
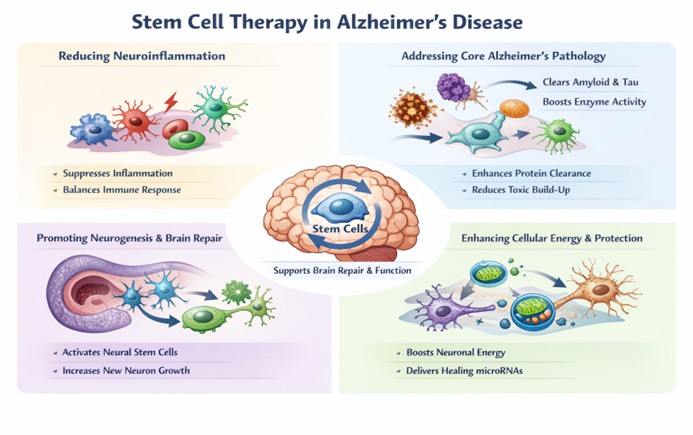
Reducing Neuroinflammation
Chronic inflammation in the brain is a key contributor to the progression of Alzheimer’s disease. Immune cells such as microglia and astrocytes can become overactivated, releasing substances that damage neurons rather than protect them. Stem cells have the ability to counteract this harmful process by releasing anti-inflammatory cytokines.
Substances like interleukin-10 (IL-10) and transforming growth factor-beta (TGF-β) help suppress pro-inflammatory molecules, including IL-1β and tumor necrosis factor-alpha (TNF-α). In addition, stem cells can influence immune cell behavior, encouraging a shift from harmful inflammatory states to more protective and restorative ones. This rebalancing of the brain’s immune environment helps limit further damage and supports recovery.
Addressing Core Alzheimer’s Pathology
One of the most promising aspects of stem cell therapy is its potential to directly impact the defining features of Alzheimer’s disease—amyloid-beta accumulation and tau pathology. Stem cells contribute to the reduction of these toxic proteins through several complementary mechanisms.
They can stimulate microglia to actively identify and remove amyloid deposits. They also promote the production of enzymes such as neprilysin and insulin-degrading enzyme (IDE), which break down amyloid-beta. Additionally, stem cells enhance cellular processes like autophagy and lysosomal activity, which are responsible for clearing damaged or unnecessary proteins within cells.
Beyond these actions, stem cells release regulatory molecules that support cellular cleanup and improve neuron survival. By reducing the burden of toxic proteins, these therapies may help restore healthier brain function.
Promoting Neurogenesis and Brain Repair
Alzheimer’s disease disrupts the brain’s natural ability to regenerate and maintain neural networks. Stem cell therapy offers a way to stimulate these regenerative processes. Research suggests that stem cells can activate dormant neural stem cells, encouraging them to produce new neurons.
This effect is particularly important in the hippocampus, a brain region essential for memory formation and learning. By increasing neurogenesis and enhancing synaptic density, stem cells help rebuild communication pathways between neurons. Strengthening these connections is vital for preserving cognitive abilities and may even contribute to partial recovery of lost functions.
Enhancing Cellular Energy and Protection
In addition to structural repair, stem cells provide significant support at the cellular level. One notable mechanism is their ability to transfer mitochondria to damaged neurons. Mitochondria generate cellular energy, and when they function improperly, it is often linked to the development of neurodegenerative disorders. By supplying healthy mitochondria, stem cells can improve energy production and help restore neuronal activity.
Stem cell-derived extracellular vesicles (EVs) also play an important role. These microscopic particles carry proteins, lipids, and genetic material such as microRNAs. Specific microRNAs, including miR-21 and miR-124, are known to reduce inflammation, protect neurons, and promote synaptic function. Through this form of intercellular communication, stem cells deliver targeted instructions that enhance the brain’s ability to repair itself.
A Multifaceted Therapeutic Approach
Stem cell therapy stands out because it targets Alzheimer’s disease through multiple pathways simultaneously. Rather than focusing on a single aspect of the disease, it combines anti-inflammatory effects, protein clearance, neuroprotection, and regenerative stimulation. This comprehensive approach offers a distinct advantage over traditional treatments that primarily address symptoms.
By supporting neuron survival, enhancing brain plasticity, and improving cellular function, stem cells create an environment that is more conducive to healing. This integrated strategy may ultimately lead to more meaningful and sustained outcomes for patients.
Looking Toward the Future
The development of stem cell-based therapies represents a significant shift in how Alzheimer’s disease may be treated in the future. As research continues, these therapies could evolve from experimental interventions into widely accepted clinical options. Advances in cell processing, delivery methods, and patient selection are expected to further improve their effectiveness and safety.
While more large-scale studies are needed to confirm long-term benefits, the current evidence suggests that stem cell therapy holds considerable promise. By addressing the root causes of Alzheimer’s rather than merely alleviating symptoms, it offers new hope for slowing disease progression and potentially restoring aspects of brain function.
In the years ahead, regenerative medicine may play a central role in transforming the management of neurodegenerative disorders, providing patients and families with more effective and meaningful treatment possibilities.

