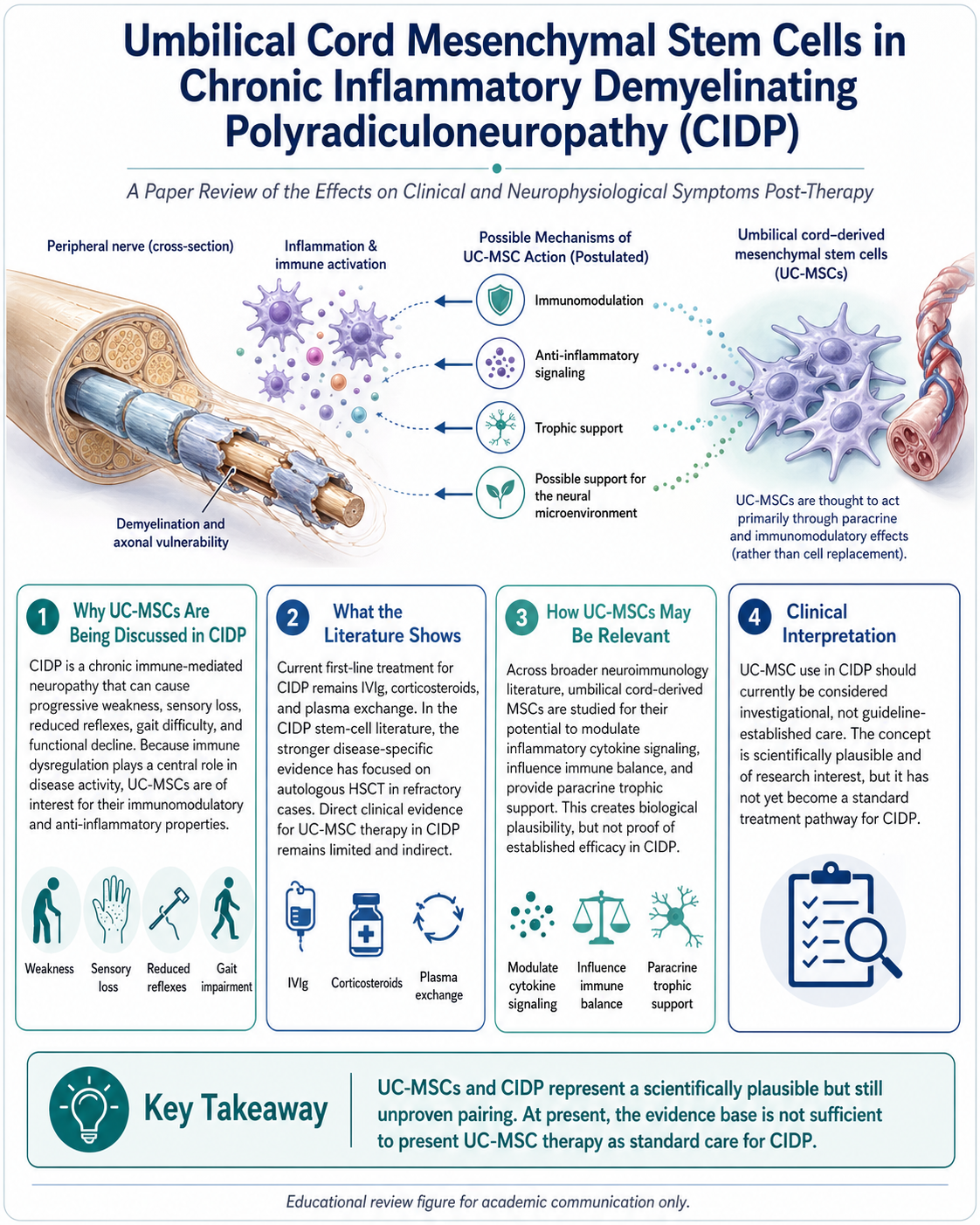
Why UC-MSCs Are in The News In CIDP
Chronic Inflammatory Demyelinating Polyradiculoneuropathy (CIDP) is chronic immune-mediated neuropathy characterized by inflammation and dysregulated immune activity damaging the myelin sheath of peripheral nerves and sometimes the axon. The onset is clinically characterized by progressive weakness and may also feature sensory loss, decreased reflexes, gait impairment and functional decline over time. The first-line treatment continues to consist of conventional immunotherapy mainly IVIg, corticosteroids, and plasma exchange. That point is relevant because any conversation about UCMSCs in the context of CIDP has to start with the reality that cell-based therapy isn’t a first-line treatment in major guidelines from a clinical evidence standpoint.
Biological plausibility is the reason UC-MSCs garner interest. Mesenchymal stromal cells (MSCs), including those obtained from umbilical cord tissue, are investigated for immunomodulatory, anti-inflammatory and trophic signaling activity. In neuroimmunological and more broadly neurological literature, this is described in terms of how MSCs may alter T-cell balance, inflammatory cytokine signaling or wound healing pathways as opposed to direct replacement of damaged nerves. Its theoretical profile makes them of general relevance to CIDP, which is increasing viewed not just as a structural neuropathy but rather a complex disorder of the immune system itself.
Paper Literature vs The Reality
An extensive literature review reveals that there has been a growing focus in CIDP stem-cell literature over the past two decades, especially in more recent years, and this investigation appears to be directed mainly by clinical trials based on hematopoietic stem cell transplantation versus UC-MSC. The most distinctive record of CIDP-specific interventional data found among the retrieved sources is ClinicalTrials. Presented is a gov study that assesses hematopoietic stem-cell transplantation in CIDP. Similarly, CIDP studies and reviews available from the search results are concentrated on autologous HSCT for resistant disease including long-term follow-up plus outpatient protocols in carefully chosen patients.
This doesn’t mean that UC-MSCs are without merit. This means that the evidence base continues to be indirect. Reviews of stem cell therapy in neuroimmunological diseases and neurological disorders mention MSCs, including umbilical cord-derived types, as highly remarkable research tools. But those reviews do not create an established UC-MSC protocol for CIDP. It’s an interesting concept scientifically, but it still lacks CIDP specific clinical evidence.
The UC-MSC Angle Is Still Something Researchers Are Considering
The argument for UC-MSCs as a mechamism-agnostic adjunctive therapy is understandable, even with the absence of direct high-level CIDP trial evidence. In CIDP, there is immune-cell activation and inflammatory signaling in the context of chronic injury in the peripheral nervous system microenvironment. Content on the pathogenesis of CIDP discusses evidence for a central role for immune dysregulation in disease activity; this compares with MSC reviews that refer to these cells as potential modulators of inflammation or immune balance. This will be the principle bridge from UC-MSC interest to CIDP in a paper-review perspective: clinically unproven efficacy mediated through at least biologically plausible immunoregulatory rational.
MSC derived from the umbilical cord are especially promising in the wider regenerative literature because they are more readily available than some adult tissues, and this has been increasingly coupled with discussion of vigorous paracrine signaling and immunomodulatory activity. But plausibility is not the same as evidence. Such a literature-based review must therefore make clear that attractive biology does not in itself constitute an assured role for any agent in routine clinical use in CIDP.
The benefits of reviews are limits on what they recommend
The most compelling supportive evidence in the CIDP stem-cell literature is found in reports of refractory cases treated with autologous HSCT that have demonstrated sustained improvement in subsets of patients and a decreased reliance on long-term immunotherapy. These findings are important because they are in line with the general notion that immune reset using cell-based strategies can play a role (strictly chosen) in severe cases of immune-mediated neuropathy. HSCT is still a very distinct intervention in comparison to UC-MSC therapy and has its own risk-benefit profile. This should not be taken as an evidence that UC-MSC therapy has already been proven for CIDP.
Clinical Interpretation: Investigational, Not Established
The best conclusion for clinicians, researchers, and medically informed readers is therefore that the use of UC-MSC with CIDP continues to be investigational. The guidance on CIDP available one prefers conventional immunotherapies. The CIDP-relevant stem-cell literature primarily refers to studies concerning hematopoietic transplantation in refractory disease, whereas MSC literature provides a much more speculative and disease-adjacent rather than an evidence-based CIDP-specific clinical proof. Which means that UC-MSCs are an open question (read: it has not yet passed a guideline backed standard of care treatment pathway).
Final Paper-Review Takeaway
As an evidence-based paper-review subject, it makes the most sense to regard UC-MSCs and CIDP as discussable scientifically-plausible but not yet warranted pairings. CIDP is an immune-mediated neuropathy with well-defined standards for conventional treatment. There is definite interest in stem-cell for this disease space, but the existing clinical literature specific to CIDP and HSCT currently favours autologous HSCT in treatment-refractoy cases vs an established UC-MSC protocol.