UC-MSCs and DFPP in Liver Treatment: Why Reducing LDL and the Inflammatory Burden First May Matter
UC-MSCs could originally be beneficial because of their capacity to delay pro-inflammatory responses induced by pathological conditions and, secondly, for the role as potential carriers able to give support for achieving anti-inflammatory functions. In particular, when therapeutic benefits must target a liver disease where both LDL reduction and lowering immune burden are essential for ameliorating overall prognosis.
There is not one pathway that drives chronic liver disease. In the majority of patients, particularly those with metabolic-associated steatotic liver disease or mixed metabolic-inflammatory injury, there is integrated exposure of the liver to lipid overload, oxidative stress, immune activation and fibrotic signaling. This explains why treatment of liver disease in clinical practice has become more and more directed not just towards the liver itself, but also toward the systemic environment that facilitates progression of hepatic injury. Dyslipidemia and inflammatory signaling, both of which are closely associated with metabolic liver disease, can all drive more steatosis, inflammation and fibrosis over time.
Based on this framework, it may be possible to conceptually present DFPP and UC-MSCs in a sequential manner as an investigational pathway rather than as standard protocol. The logic is simple: the initial role of DFPP is to wash away circulating atherogenic and inflammatory plasma components, while antecedent stimulation of downstream immunomodulatory, anti-fibrotic, and regenerative signaling come from UC-MSCs. While this is a nice biologically plausible concept, it is more appropriately characterized as investigational than routine clinical care of the liver. Clinical data for the exact sequence i.e., DFPP first, UC-MSC second in liver fibrosis remains limited.
Reasons For Electrolyte Drain First Before UC-MSCs
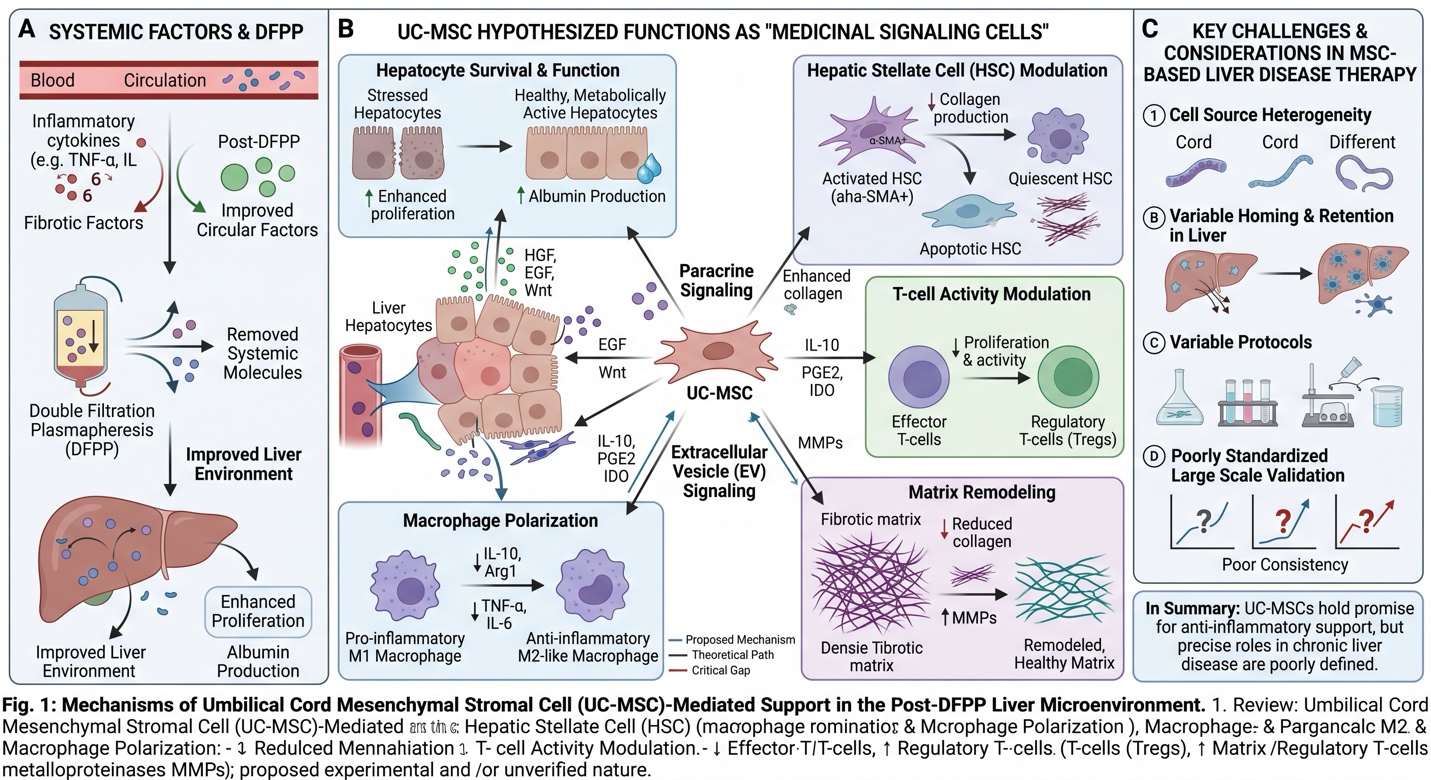
DFPP provides a distinct advantage in the form of selectively isolated large components from circulated plasma. DFPP is well characterized in the lipid-apheresis literature, where it has been described to lower both LDL cholesterol and lipoprotein(a), although a recent review notes wider “pleiotropic” activities including removal of substances involved in lipid metabolism and major reductions in inflammatory markers (e.g., C-reactive protein). That matters since a liver already under the duress of chronic inflammatory and lipid stress may not be as happy a landing for any subsequent regenerative approach. Simply put, DFPP could offer to remove some of the circulating burden before a cell-based therapy is implemented.
This is particularly pertinent for liver patients with pronounced metabolic traits. Atherogenic lipoproteins and systemic metabolic dysfunction do not represent isolated cardiovascular problems but are deeply interwoven with hepatic steatosis and inflammatory progression of metabolic liver disease. DFPP is not a treatment for liver fibrosis, but may represent an opportunity to reduce adverse circulating factors (e.g., LDL-related burden) prior to the consideration of a second-stage regenerative intervention.
What UC-MSCs might contribute after DFPP
Alternatively, if DFPP is thought of as a way to improve the factors in circulation, UC-MSCs may be viewed as tools through which to influence what goes on next at hepatocyte level in the liver microenvironment. Recent liver MSC reviews describe mesenchymal stromal cells as functioning more closely to: ‘medicinal signaling cells’, rather than direct replacement cells. They proposed roles for inhibiting hepatic stellate cell activation, guiding macrophages toward an anti-inflammatory state, as well as modulating T-cell activity, maintaining hepatocytic survival and enhancing matrix remodeling by paracrine and extracellular vesicle-mediated signaling.
Recent reviews on MSC therapy for liver disease also describe the preliminary clinical data indicating beneficial effects of MSC-based therapy on liver-related measures in selected conditions, but consistently highlight the same cautions: heterogeneous cell sources, varying homing; variable protocols and poor standardized large scale validation. In summary, UC-MSCs are appealing based on their anti-inflammatory and supportive biology, but the exact role of UC-MSCs in chronic liver disease remains poorly defined.
Why the Sequence Matters Conceptually
Mechanistically, we believe the alternating DFPP + UC-MSCs schedule is appealing as these two methods act on non-overlapping disease biology layers. DFPP exerts hypothetic mechanism on the systemic plasma level, where it may affect atherogenic lipoproteins and some inflammatory mediators. In contrast, UC-MSCs tend to mediate their effects at the level of cell-signalling and tissue-response: modulating inflammation, reducing fibrogenic signalling promote endogenous repair pathways. While this does not demonstrate clinical synergy, it provides a rationale for the rationale etc for why some clinicians and researchers might view these combinations worthwhile trialing in liver patients with ongoing inflammatory–metabolic burden.
Although not in direct relationship with DFPP specifically, there is some liver-specific precedent to use plasma purification for combination with UC-MSC therapy, but most published experience relates more broadly to plasma exchange. Clinical trials and published clinical experience in hepatitis B-related liver failure describing plasma exchange as a method to improve the micro-environment by removal of adverse circulating factors pre- or concomitantly with UC-MSC therapy. Although that evidence does not directly prove DFPP equals the same term, it lends credence to the greater concept that conditioning the internal milieu prior to performance of a cell therapy is not just speculation.
Potential Benefits for Liver Patients
The promises of potential benefits if it is carefully crafted might be as follows:
Right before UC-MSCs: DFPP might serve to lower LDL and other negative plasma ingredients which could assist diminish section of the systemic inflammatory and metabolic burden.
Post-UC-MSCs: UC-MSCs could regulate inflammatory signaling and thus stellate-cell activation, promote hepatocyte recovery, or remodel the hepatic microenvironment via paracrine mechanisms.
Combined: sequence may be useful as a research method in select liver patients, particularly those with overlapping metabolic injury, inflammation and fibrosis. But this is still a biological-null model, it isn’t an ACL-education imperative.
A Balanced Clinical Interpretation
The most prudent conclusion is therefore that DFPP in conjunction with UC-MSCs should currently be considered an investigational liver-treatment approach. The rationale for DFPP and UC-MSCs for their acute removal of larger plasma factors associated with autoimmune diseases, while DFPP is more clearly demonstrated for its support in these paradigms. UC-MSCs have a growing but still incomplete evidence basis in liver regeneration and fibrosis modulation. Doing DFPP first and UC-MSCs second is a scientifically coherent idea, especially in the setting of metabolically inflamed liver disease; larger, controlled studies are required to demonstrate which patients fare best with this approach, at what point treatment should be timed, and whether the sequence improves on standard care.

