Rheumatoid arthritis (RA) is a type of chronic systemic autoimmune disease that due to persistent synovial inflammation, resulting in progressive joint damage, pain, stiffness and functional limitation. While the disease predominantly affects joints, its inflammatory toll may have a greater systemic activities that are best viewed as comorbidities associated with greater cardiovascular and pulmonary activity. Consequently, rheumatoid arthritis is no longer simply a musculoskeletal disorder but rather a complex immune-mediated health condition that necessitates long-term multidisciplinary management.
The management of many patients has been dramatically better with current therapeutic options. Traditional synthetic DMARDs, biological agents, targeted synthetic therapies, corticosteroids and supportive rehabilitation continue to be the cornerstones of modern RA management. However, despite these advances, a subset of patients still has persistent low response rates, intolerance to medication and repeated flares or functional decline. Individualized treatment decisions remain emphasized in clinical guidelines, integrating disease control, safety, comorbidities and patient-specific risk factors.
In this context, regenerative medicine has received more and more academic interest as an additional research field. Umbilicle cord-derived mesenchymal stem cells (MSCs), such as umbilical cord-derived ones, known collectively as UC-MSCs, are also being extensively studied for their putative immunomodulatory and tissue-supportive properties against transplant rejection and a variety of autoimmune and inflammatory diseases. Their suggested role in rheumatoid arthritis is not to supersede established rheumatologic therapy, but rather to determine if they may qualify for consideration as cell-based interventions that can intervene and help stabilize or modulate immune activity; assist with limiting the generation of endogenous or exogenously applied pro-inflammatory signals; and/or enable a safer/healthier biologic milieu for tissue repair.
Autoimmune Joint Destruction: Pathophysiological Mechanisms and Clinical Manifestations
The breakdown of immune tolerance and subsequent activation of both innate and adaptive immune pathways is the process called rheumatoid arthritis pathogenesis. Immune cells including T and B cells as well as macrophages, dendritic cells, and fibroblast-like synoviocytes penetrate the synovium. These cells secrete pro-inflammatory mediators including tumor necrosis factor-alpha (TNF-α), interleukin-1 (IL-1), interleukin-6 (IL-6) and interleukin-17(IL-17), which maintains chronic inflammation in the joint microenvironment.
Chronically, sustained inflammation fosters synovial hyperplasia and fibrovascular tissue formation (pannus). Such invasive inflammatory tissue mediates cartilage destruction and induces osteoclast-mediated pathological bone erosion. Clinically, the patients usually present with symmetrical joints pain, swelling, morning stiffness, fatigue and decreased ability to grip or walk. In advanced disease, progressively destructive joints can deform and disable the patient, leaving them increasingly dependent on others for everyday activities.
Regenerative capacities and immunomodulatory mechanisms of UC-MSCs
The expanded use of UC-MSCs in the field of regenerative medicine has stemmed from their high proliferation rate, immunomodulatory capabilities, and their ability to create an ideal microenvironment through paracrine signalling activity by releasing soluble factors that can modify immune-cell behaviour. In contrast to drugs that usually hit a single well-defined pathway, mesenchymal stem cells seem to work through multiple approaches including secretion of cytokines, growth factors, extracellular vesicles and other bioactive molecules. These in turn can have paracrine effects that provide autocrine control of inflammation and facilitate tissue repair processes.
As regard to studies on rheumatoid arthritis, you might have come across that UC-MSCs easily restore the immune imbalance. These proposed mechanisms are based on the inhibition of overstimulation of T-cell activity, with modulation of Th1 and Th17 inflammatory responses, enhancement of regulation by regulatory T-cells, suppression of dendritic-cell maturation and downregulation of pro-inflammatory cytokine signaling. Such consequences are particularly relevant, since rheumatoid arthritis is characterized by increased immune activation and loss of immune tolerance.
Available preclinical data and early-phase clinical studies suggest promise for MSC-based strategies in some populations of RA patients with an acceptable safety profile. However, at this moment the evidence is still evolving. A systematic review and meta-analysis yielded promising safety results and a non-significant trend towards clinical improvement, though underscored the necessity for larger well-designed randomized studies to elucidate on efficacy, including optimum dose-response relationships, method of delivery route, durability of response over time and patient selection criteria.
Thus, UC-MSCs should be considered an investigational or adjunctive regenerative therapy and not a replacement for rheumatologist-guided standard of care. ConclusionIn conclusion, in an academic and clinical environment the best positioning would be of a supportive therapy for helping characterization of immune modulation and tissue repair signaling by UC-MSCs as other studies look to define their role in rheumatology.
Clinical Positioning at a Thai Stem Cell Clinic
A role for a targeted approach is exemplified by the use of an integrated clinical pathway by a specialized stem cell clinic in Thailand-based on regenerative possibilities for patients with rheumatoid arthritis. An academic (appropriate) protocol would start with an extensive medical oncologic review including duration of the disease, medication history, previous response to biologics or DMARDs, pattern of flares, presence of comorbidities and infections risk factors, imaging history and baseline markers of inflammation such as ESR or CRP. Other measures for patient outcomes such as scoring of disease activity, functional assessment and collaboration from rheumatology play also critical role to monitor the patients more objectively.
UC-MSC effects in RA: future directions
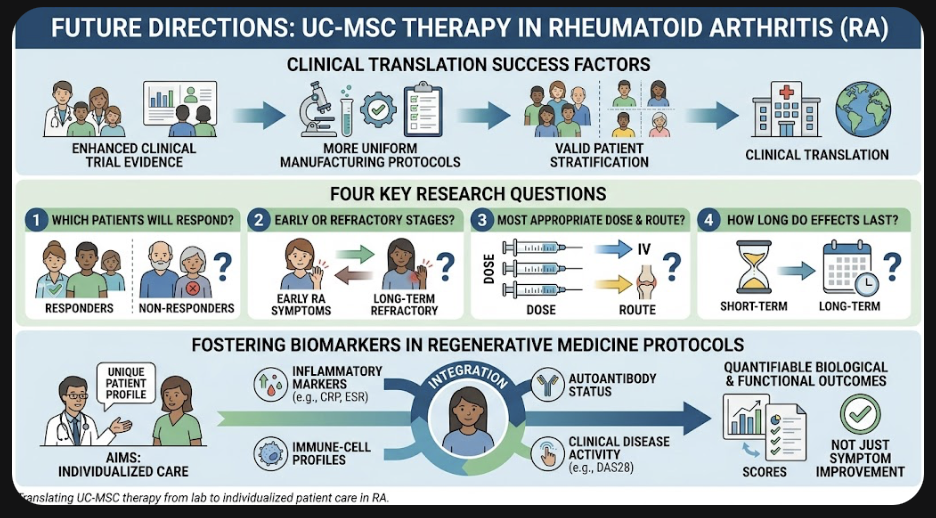
Figure A: Toward Personalized Regenerative Medicine for Rheumatoid Arthritis
The translation of UC-MSC-based therapies for rheumatoid arthritis will be contingent on an enhanced clinical trial evidence, more uniform manufacturing protocols and valid patient stratification. There are four key research questions: (1) which patients will respond, (2) whether early or more refractory stages of disease will yield the greatest response, (3) what is the most appropriate cell dose and route of administration, and (4) how long do the effects last?
Fostering Biomarkers in Regenerative Medicine Protocols Another key avenue is the inclusion of biomarkers into regenerative medicine protocols. Measurements of inflammatory markers, immune-cell profiles, and autoantibody status, as well as clinical disease activity may provide insight into treatment response over time among clinicians. This model promotes a more individualized type of care because regenerative interventions are assessed not just based on symptom improvement, but on quantifiable biological and functional outcomes.