Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative disorder which affects both the upper and lower motor neurons. Later in the disease, individuals are vulnerable to weakness of the muscles, muscle twitching, pain in speech or difficulty swallowing food and weight loss and breathing problems. Care for ALS today still revolves around early diagnosis driven by methods rooted in neuromuscular testing machines, symptom management via drugs and physical therapy as needed, respiration through mechanical aids, nutrition with supplements such as PEGs breathing support devices rather than the stem cells we were all promised would be a standard from day one of the disease. The only drugs now approved for the general ALS populations are riluzole and edaravone but tofersen has been approved for SOD1-associated ALS. Multidisciplinary care continues to be a cornerstone of optimal ALS management through preservation of function and quality of life as needs evolve over time.
Discussion of UC-MSCs in ALS
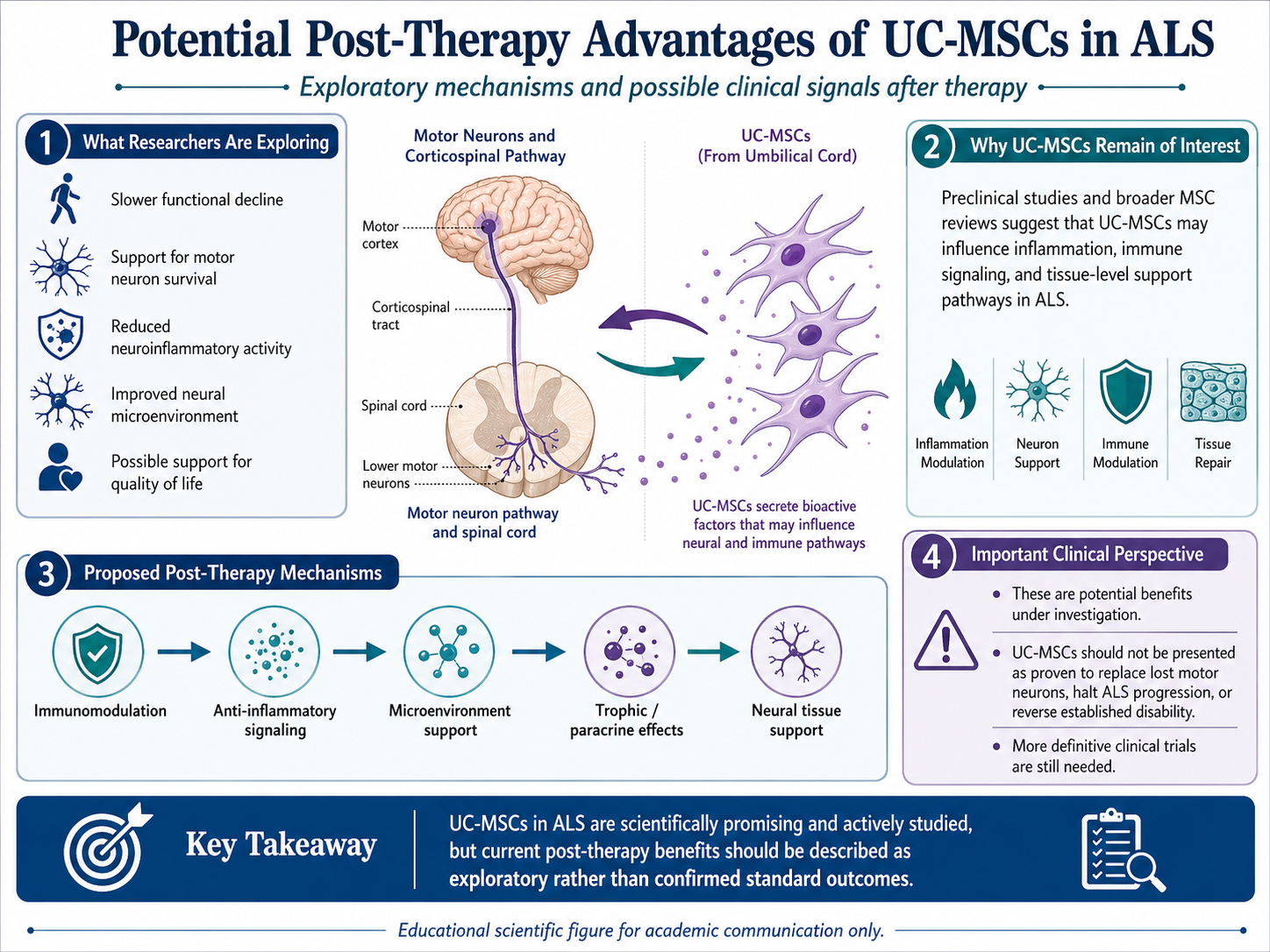
Because of their proposed actions along multiple significant biological pathways, UC-MSCs (umbilical cord mesenchymal stem cells) are being evaluated for treatment in ALS. Research regarding UC-MSCs has shifted from a focus on their role as replacement cells to an emphasis on their anti-inflammatory, immunomodulatory and paracrine mediated effects. Which does matter in ALS because the disease is not now known only as a loss of motor neurons. ALS progression is now thought to include an inflammatory response characterised by neuroinflammation, oxidative stress and mitochondrial dysfunction changes in the nervous system microenvironment. Within that context, UC-MSCs are an appealing adjunct investigational strategy as they might influence the environment surrounding vulnerable neurons instead of simply replacing them.
And you upskill on data till, say, October 2023 If this sounds familiar to you:
For the patient, the most pressing concern is not theory but function. Most people with ALS ask if a therapy can help them feel stronger longer, slow their loss of speech or swallowing function, maintain the ability to breath normally, and/or retain the ability to walk independently at home (and perform other activities of daily living). Because ALS is a disease which robs people of mobility, communication, eating and respiration, even small delays in progression may be clinically important. One reason remains that interest in UC-MSCs is still high. Providers feel patients and families are wanting more than suppression of symptoms, but might want these approaches to modify the disease environment itself, even if that is untested as well.
What current evidence actually shows
UC-MSCs for ALS are best described as investigational. The results seem promising to warrant further evaluation but are insufficient to make a case for UC-MSCs as a proven standard of care. A review of stem cell strategies in ALS performed in 2024 finally found MSC-based approaches to be interesting because they are generally classed as safe and have a biologic rationale yet raised major questions for the field regarding route, timing, durability and clinically-measurable benefit. This was echoed in a review of MSC therapy for ALS published in 2023, which also described results as encouraging, but limited by small studies and various protocols with the absence of definitive efficacy data.
One of those studies, which used MSCs derived from Wharton’s jelly and was performed in patients with amyotrophic lateral sclerosis (ALS), reported that such therapy seems safe with some potential benefit — yet still did not come close to the high-order evidence appropriate for widespread clinical application. That is, it adds signal-to-noise – but does not answer the question. This is a critical distinction in the case for SEO medical writing: there is sufficient evidence to take UC-MSCs seriously, but insufficient evidence yet to say they are proven therapy for ALS.
Possible advantages of UC-MSC transcriptomic after therapy
Researchers using UC-MSCs for ALS this is generally been approached through a number of potential benefit areas. This consists of stepping into the pace of purposeful decline, conserving motor neuron survival, lowering neuroinflammatory hobby, enhancing the microenvironment of the apprehensive device and might even support spare quality of life. These mechanisms are supported by preclinical studies and broader reviews on MSCs, explaining the continued interest. But, the words “potential benefits” are critical here. Currently, these are still at guideline level exploration stage vs. confirmation stage.
In other words, no cautious article should claim that UC-MSCs can replace lost motor neurons or halt ALS disease progression or reverse established disability. A better message would be that UC-MSCs are being explored because they may regulate inflammation or operate locally at the tissue level in ways that may matter to select patient populations, but more definitive trials remain warranted.
Safety, constraints and the significance of caution
Safety may be ahead of proof of efficacy — this is a common refrain in the ALS stem cell literature. Synopsis: A great number of studies show results indicating that MSC therapy can be feasible in neurologic disease and especially after intrathecal MSC administration, but at the same time many factors influencing heterogeneity across studies are reported and information on trial design is lacking. Different studies have applied distinct cell sources, doses, routes and follow-up schedules (Harrison et al., 2021), precluding direct comparisons of results or defining one single best protocol.
It is thus important that current use of UC-MSCs for ALS be characterized as research-based at this point, and not standard practice. Comprehensive neurologic care, respiratory monitoring, nutritional support, rehabilitation and prescription of approved medications when indicated is an evidence-supported recommendation that continues to be the most strongly suggested in people living with ALS. Those discussions on stem cells truly belong alongside that framework, not in the place of it.
Final takeaway
UC-MSCs for ALS are raised because of their match to a number of relevant disease mechanisms, particularly inflammation, immune signaling and microenvironment support. That gives them scientific credibility and clinical relevance. But the existing evidence is still not enough to put UC-MSC therapy out of investigational status. We hope the most reliable position on UC-MSCs and ALS is this: use of UC-MSCs represents a new consideration for future ALS management, but current treatment should be rooted in confirmed multidisciplinary practice until the role of stem cells is better defined through clinical investigation.

