Why does this topic pull people in so strongly?
When people search for Natural Cellular Regeneration and ovarian health, they are usually not looking for abstract science. They are looking for a way to make sense of diminishing ovarian reserve, premature ovarian insufficiency, repeated IVF disappointment, or the feeling that reproductive time is moving faster than expected. That emotional weight is exactly why this topic needs careful language. The research is real, but it is also very easy to oversimplify.
What “Natural Cellular Regeneration” should mean here
It is a biological idea, not a guaranteed clinical outcome.
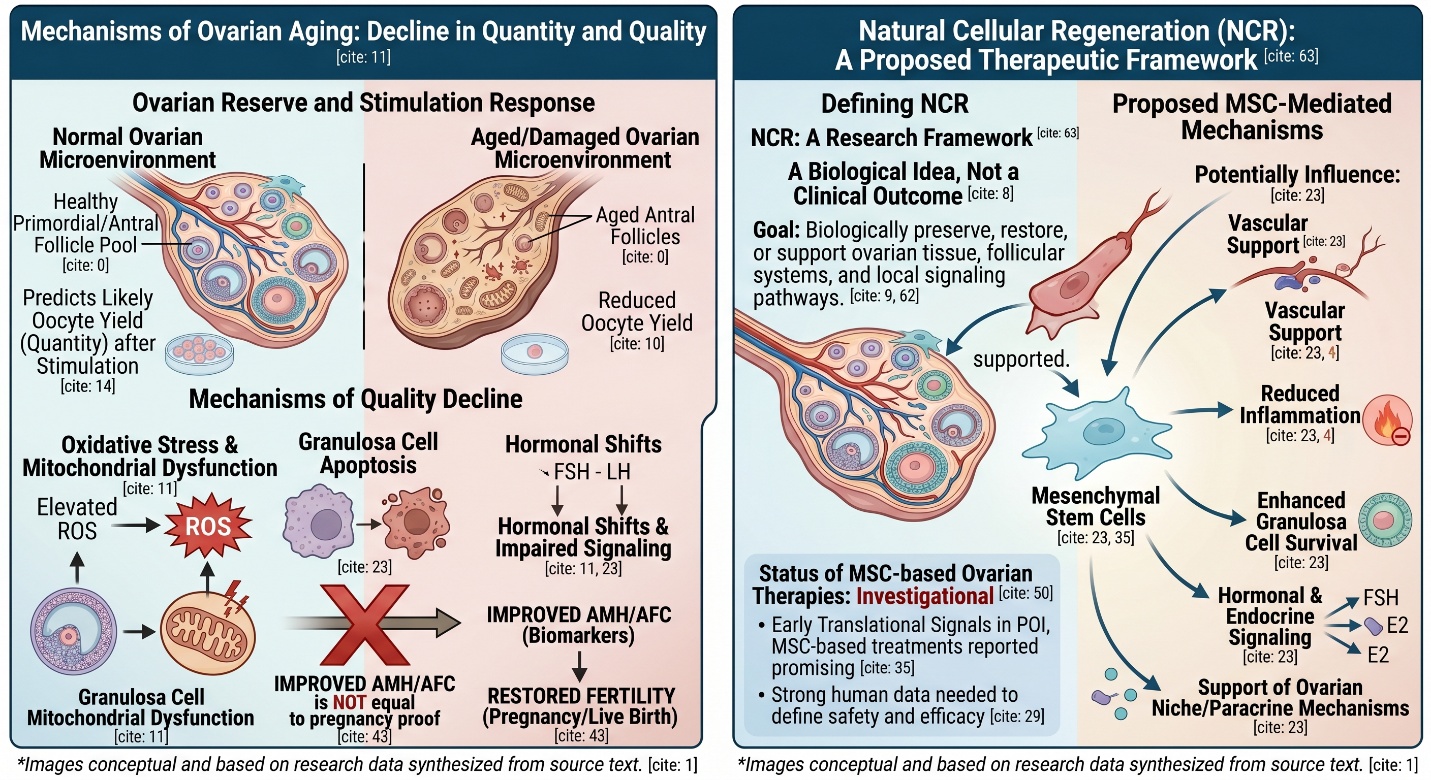
In ovarian medicine, Natural Cellular Regeneration is best understood as a way of talking about whether ovarian tissue, follicular support systems, and local signaling pathways can be preserved, restored, or biologically supported. That sounds attractive because ovarian aging is not just about “running out of eggs.” Recent reviews describe it as a decline in both follicle quantity and quality, influenced by oxidative stress, mitochondrial dysfunction, hormonal shifts, and changes in the ovarian microenvironment.
Ovarian reserve is not the same thing as fertility.
This is one of the most important distinctions in the whole discussion. ASRM defines ovarian reserve as the number of oocytes remaining in the ovary, and says markers such as AMH and antral follicle count are useful for predicting likely oocyte yield after stimulation. But ASRM is also clear that these markers are poor independent predictors of reproductive potential. That means an improved number on paper does not automatically equal restored fertility or a higher live birth rate.
Why did stem cells enter the ovarian conversation at all
The main clinical targets are POI and poor ovarian response
Most serious work in this area focuses on women with premature ovarian insufficiency (POI) or diminished/poor ovarian reserve, especially when conventional IVF outcomes are limited. The 2024 international POI guideline states that POI substantially reduces the chance of natural conception, while also noting that intermittent ovarian activity can still occur in some non-surgical cases.
The rationale is real, but the field is still early.
It is not hard to see why stem cells have gained scientific attention. Mesenchymal stem cells have therefore been studied to potentially influence the survival of granulosa cells, inflammation, vascular support, as well as endocrine signaling related and more broadly the ovarian niche through paracrine mechanisms rather than mere tissue replacement per se(45). The scientific reasoning behind keyword stem cells ovaries fertility is then starting to fall into place, with researchers posing the question of whether cellular therapies can be used to support an ovarian environment that has become locally less responsive or prematurely aged.
What the current evidence actually shows
There are early human signals, but they are still early.
A 2025 clinical cohort study of intraovarian transplantation for UC-MSC in women with POI is also one of the more discussed recent studies. Higher antral follicle count, a greater number of oocytes retrieved in the first stimulation cycle following treatment, and increased frozen embryos were reported for the treated group compared to the control group. The authors hailed the strategy as promising but also specifically urged larger multicenter studies to define long-term safety and efficacy more precisely. And that is the crux: it makes for an interesting signal, but not a routine clinical certainty.
Systematic reviews are still evaluating the field, not closing the case.
A 2025 systematic review on cellular therapies in primary ovarian insufficiency and poor ovarian reserve was designed to evaluate efficacy, safety, and limitations. That wording tells you a lot about where the field still stands. It is strong enough to justify formal evidence synthesis, but not mature enough to be treated as established standard practice. Reviews published in 2025 also continue to describe MSC-based ovarian therapies as promising, especially in preclinical and early translational settings, while still emphasizing the need for stronger human data.
What the guidelines say matters most
The current guideline tone is more cautious than clinic language.
This is the part readers deserve to hear clearly. The 2024 international POI guideline states that women with POI should be informed that there are no interventions that have been reliably shown to increase ovarian activity and natural conception rates. That is a very strong sentence, and it should sit close to the center of any honest article on ovarian stem cell claims.
Stem cell therapy is not recommended as a routine IVF add-on.
The ESHRE good practice recommendations on add-ons in reproductive medicine also state that stem cell therapy for premature ovarian insufficiency, diminished/poor ovarian reserve, or thin endometrium is not recommended. The reason is not that the field is meaningless. Although the evidence remains limited, most human studies are small and observational, and the biologic rationale and safety profile are still not clear enough for routine use.
Biomarker improvement is not the same as pregnancy proof.
This is where nuance really matters. If a study reports improved AFC, AMH, hormone values, or oocyte yield, those findings may still be meaningful. But ASRM’s ovarian reserve guidance remains very useful here: ovarian reserve markers help estimate response to stimulation, not reproductive potential by themselves. In fertility medicine, surrogate markers and meaningful reproductive outcomes are not interchangeable.
How to read the phrase “stem cells ovaries fertility” more intelligently
It is a real research question, but still not a settled therapy category
The phrase stem cells ovaries fertility is awkward as language, but the question behind it is real: can cell-based therapies help restore some part of ovarian function or improve fertility-related outcomes in women with POI or poor ovarian reserve? The best answer right now is that this is an active area of reproductive research with real biologic promise, but it remains investigational rather than routine.
Better questions usually reveal the quality of the offer.
And a more down-to-earth way to think about this domain is: What specific diagnosis does it treat? POI does not equate to poor ovarian response, diminished ovarian reserve, or age-related decline in the ovary. What outcomes are being measured? End-points are not equal (hormone values, antral follicles versus oocytes retrieved and embryos frozen vs. ig pregnancy upwards live birth So what does the evidence-based medicine say right now? For the moment, professional advice is tamer than marketing talk.
The most honest conclusion
The reality is neither dismissive nor breathless. What is most compelling about the area of ovarian research this approach explores can be encapsulated in its title: Natural Cellular Regeneration—that biology may eventually sustain richer function with less invasive options than currently afford. And yes, there have been early human signals in stem cells ovaries fertility research that validate serious consideration. However, the existing evidence base is small, and recommendations based on some outcomes remain cautious, while ovarian stem cell interventions are yet to be established in standard fertility treatment. For the time being, this represents grave yet still experimental medicine.
Frequently Asked Questions
1) What does “Natural Cellular Regeneration” mean in ovarian health?
It refers to the idea that ovarian tissue, follicles, and the surrounding microenvironment might be biologically preserved or supported. In current science, it is more of a research framework than a proven treatment category.
2) Are stem cells already a routine fertility treatment for poor ovarian reserve?
No. Current recommendations say stem cell therapy is not recommended for routine use in POI or diminished/poor ovarian reserve.
3) Have any human studies shown positive results?
Yes. Early human studies, including a 2025 cohort study of intraovarian UC-MSC treatment in POI, reported improvements in AFC, oocyte retrieval, and frozen embryo outcomes. But those studies are still early and do not establish standard care.
4) If AMH or AFC improves, does that mean fertility is restored?
Not necessarily. ASRM says ovarian reserve markers can help predict oocyte yield, but they are poor independent predictors of reproductive potential.
5) What is the clearest guideline message right now for POI?
Women with POI should be informed that there are no interventions that have been reliably shown to increase ovarian activity and natural conception rates.