The chronic inflammatory bowel disease (IBD), Crohn’s disease, is characterized by recurrent intestinal inflammation with progressive tissue injury causing a long-lasting decline in quality of life. CD may involve any region of the GI tract and usually has a relapsing–remitting clinical course, but some patients develop strictures, fistulas, abscesses, deficiencies in nutrients/energy and experience frequent hospitalisation. While most patients benefit from standard of care therapy, disease control remains difficult to achieve in some patients whose disease is refractory or structurally complicated, and others who have repeatedly relapsed. The limitations in current therapies have led to increasing scientific interest in regenerative medicine, with a specific focus on stem cell-based approaches that might provide immunomodulation and mucosal repair.
Pathogenesis and Clinical Burden of Crohn’s Disease
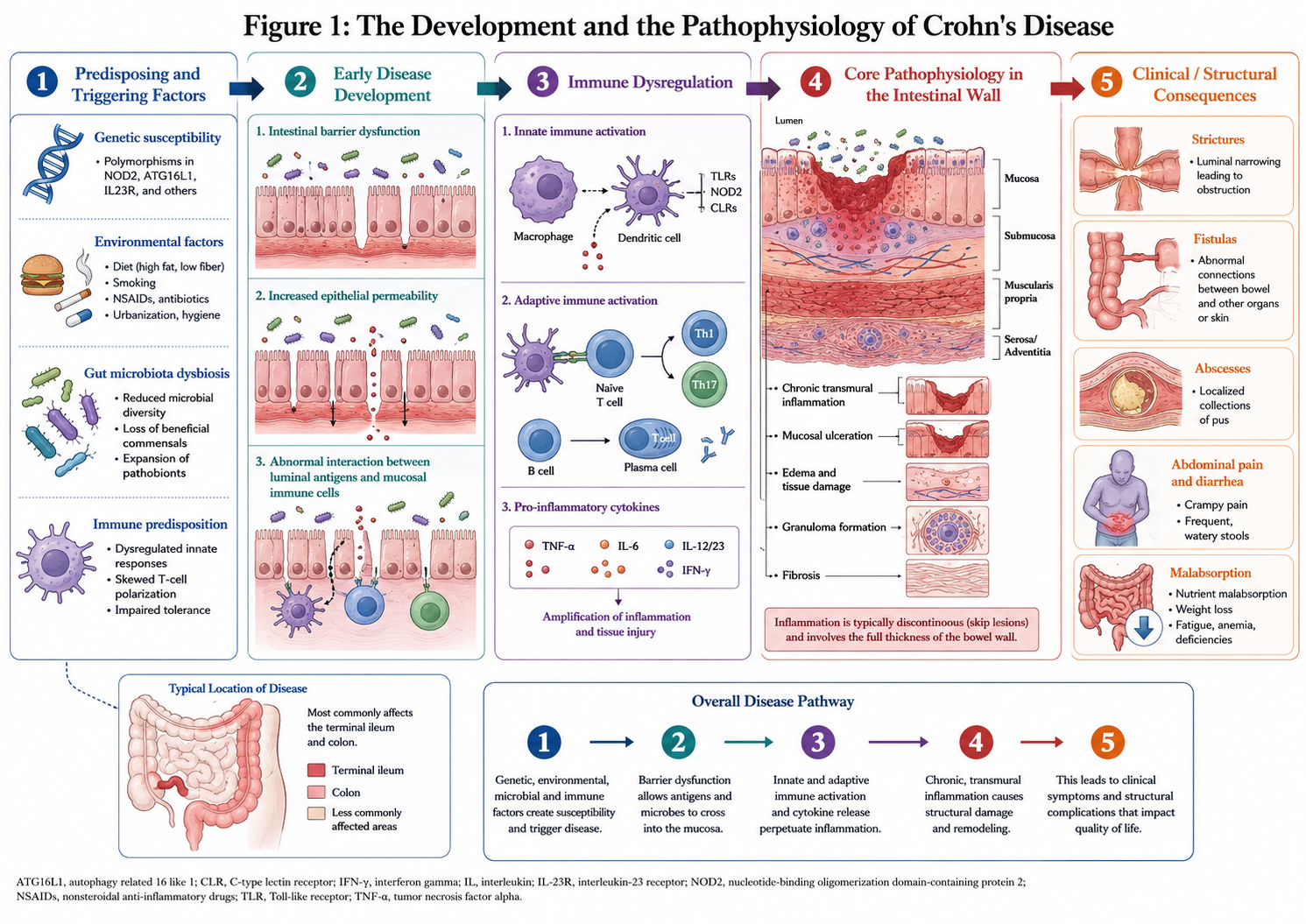
Crohn’s disease (CD) is the consequence of multilayered interactions among genetic susceptibility, environmental exposure as well as microbial factors and deregulated mucosal immunity. This causes the immune system to be continuously activated instead of balanced tolerance toward intestinal contents and commensal microbiota, leading to chronic inflammatory injury to the bowel wall. Unlike more supersticial inflammatory derangements. Crohn’s disease almost always involve transmural inflammation (where the inflammatory process goes all the way through the intestinal wall). This is partly the reason as to why the disease may evolve from ulceration to strictures, penetrating lesions, fistulas and abscess formation.
At the tissue level, Crohn’s disease is characterized by dysregulated cytokine signalling, infiltration of immune cells as well as epithelial barrier damage and repetitive injury without full repair. In due course, such conditions can affect nutrient uptake, modify bowel function, and cause fibrotic structural changes to the intestine. Clinically, abdominal pain, diarrhea, fatigue, weight loss and anemia along with fever and decreased activity can be observed in patients. Background The global burden of the disease is not only gastrointestinal but systemic, psychosocial disease that can impact work and social participation in addition to mental well-being and long-term daily function.
Figure 1: The Development and the pathophysiology of Crohn’s disease
Conventional Treatment and Its Limitations
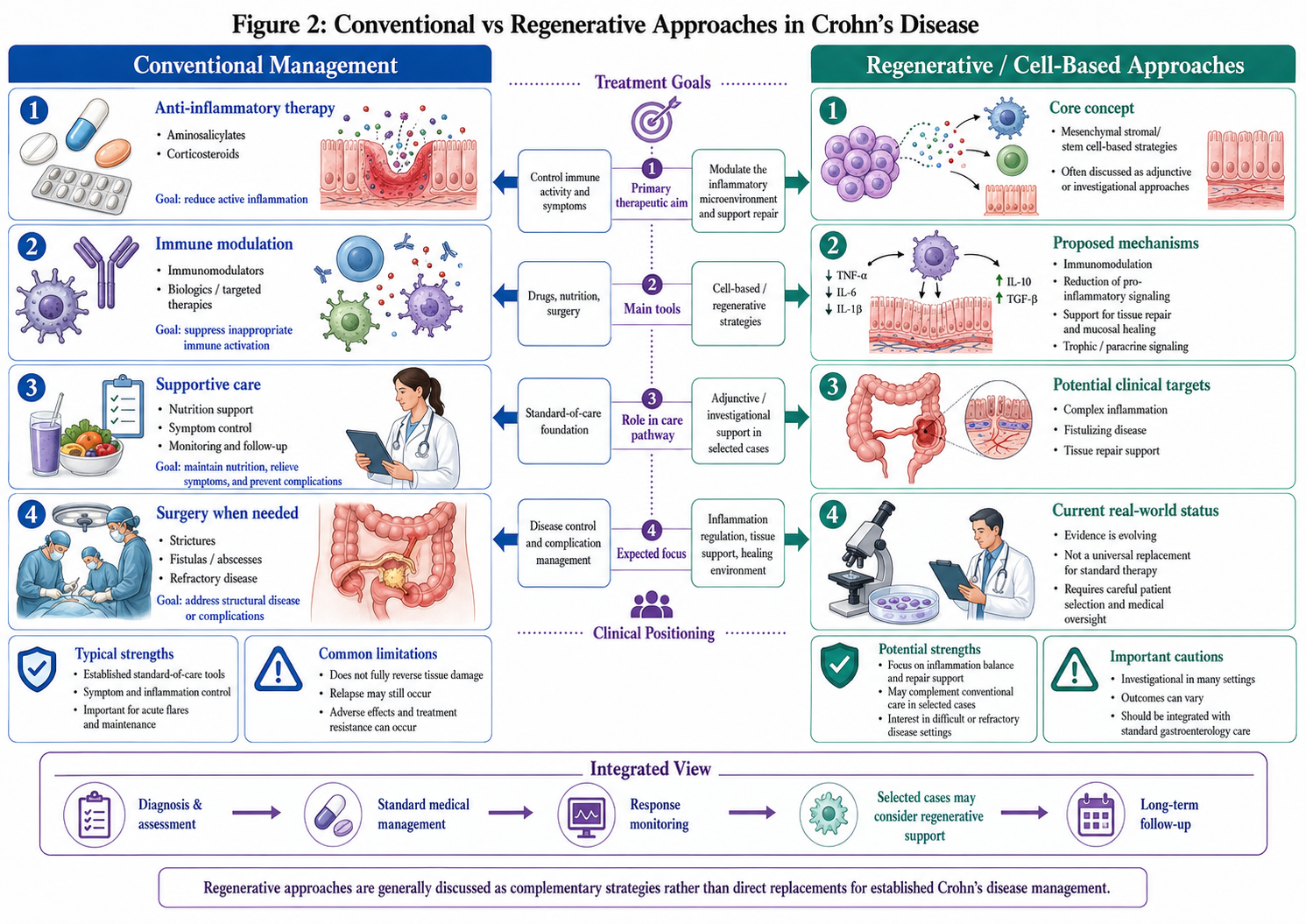
Crohn’s disease, which are based mainly on controlling inflammation, inducing and maintaining clinical remission, and preventing complications. Standard therapies range from corticosteroids and immunomodulators to biologic therapies; nutritional support and surgical intervention when indicated. Medically-oriented procedures are still relevant to contemporary practice, and they can be very successful for properly selected patients.
However, important limitations remain. Some patients do not achieve an adequate response, and many that responded become refractory over time. Chronic immunosuppression can also pose clinically significant risks such as infection and therapy-related toxicity. Surgery may be needed for obstructing, fistulous or abscessing disease and medically refractory disease but it is not curative and postoperative recurrence continues to present a well-recognized problem.
This is why Crohn’s disease remains a gap between short-term control of inflammation and long-term restoration of intestinal homeostasis. That gap has fueled interest in biologically based therapies that may have more effect than being a mere symptom-focused treatment.
Why Stem Cell Therapy Is Unfurling
Because of its immunomodulatory and supportive properties, stem cell-based therapy has been applied in Crohn’s disease. Of these cell types, umbilical cord–derived mesenchymal rarefied or stem cells (UC-MSCs) have drawn special attention. They are not primarily being studied as direct replacement cells. Instead, they are becoming more recognized as biologically active signaling cells that potentially mediate inflammation, the immune balance, and tissue repair via paracrine and vesicle-mediated mechanisms.
Potential mechanisms are proposed by which H2 can exert its effects through altering T-cell activity, downregulating pro-inflammatory cytokine signaling, promoting anti-inflammatory macrophage polarization, inducing epithelial barrier repair and/or enhancing local tissue recovery. In this regard, UC-MSCs are being proposed as a potential tool to move the intestinal microenvironment from chronic immune-mediated damage toward more regulated repair.
In Crohn’s disease, this notion is of particular relevance because the successful control may not just depend on short-term silencing but rather ongoing suppression of inflammation. Re-establishing mucosal integrity, strengthening epithelial resilience and breaking the circuit of repeated injury with incomplete healing may also be important.
The Regenerative Rationale in Crohn’
Regenerative medicine perspective provides a therapeutic paradigm unlike pharmacologic suppression alone. Instead of restricting attention to blockade of inflammation, it poses the question of whether cellular therapies may help to remodel the very tissue milieu in which they will be reconstituted. These adaptations lead to unique functional characteristics of the IBD inflammatory response, Aspects that should be considered in crohn’s disease include: mucosal healing, epithelial repair, inflammatory regulation and maintenance of intestinal structure.
UC-MSCs are a logical choice in this setting, since they may operate via numerous biologically relevant trajectories simultaneously. Loved or hated their paracrine signalling profile has been associated in experimental settings with anti-inflammatory and immune regulatory effects, tissue-protective signals, and local repair processes. As such, this has rendered them a focus of active interest — particularly in the context of complex or refractory disease with comorbid tissue damage and chronic inflammation.
But this field must be treated with some care. In conclusion, stem cell therapy in Crohn’s disease remains an area of active investigation that should still be classified as a narrow and advanced field of regenerative medicine rather than a panacea for standard gastroenterology care. Biologically, the most responsible message is that it looks good but we don’t know its exact clinical role (what patients are ideal candidates; optimal dosing), and whether the response will be durable over time.
Figure 2: Conventional vs Regenerative on Crohn’s Disease
Thailand and Development of Regenerative medicene
Some may be aware that Thailand has been an increasingly recognizable hub for much state-of-the-art medical expertise and translational biomedical development. This has stimulated an increasing interest in the role played by the country in regenerative medicine research and clinical application, within the context of stem cell therapy. Thailand has also actively engaged in the cell-based therapy area, through a combination of developing laboratory infrastructure, as well as medical expertise and international patient influx.
However, the academic significance of this development is not as a promotional positioning but rather as an opportunity to carefully structured and clinically supervised regenerative strategies. This field will continue to evolve, and the long-term impact of Thailand’s contribution will depend on research excellence, standardization efforts, safety oversight, and transparent clinical outcome reporting.
Conclusion
The incuutivity and chronicity of Crohn’s disease — a particularly intractable form of inflammatory bowel disorder — hath lasting effects through patients. Standard therapies remain a cornerstone of treatment, however they are insufficient to fully address the multifaceted nature of chronic immune dysregulation, tissue damage and/or structural progression. Thus stem cell-based therapy has become an enticing field given its potential for immune modulation and intestinal healing via releases of bioactive mediators.
UC-MSCs is a particularly compelling option due to its scientific rationale as a regenerative platform. Their suggested functions in inflammatory modulation, macrophage polarisation as well as epithelial barrier support and also tissue recovery make them very pertinent to the pathophysiology of Crohn’s illness. Nevertheless, these compounds are still best interpreted through an investigational and evidence development lens for clinical use.
A slightly more scholarly conclusion is that stem cell therapy does not replace conventional Crohn’s disease management per se, but such therapies may help in the future development of better-targeted treatment paradigms directed specifically towards repairing the gut and restoring mucosal function. Continuing research may one day help regenerative medicine change the way we conceptualize complex inflammatory bowel disease in selected patients.