Development of regenerative medicine liver fibrosis clinical trials in Thailand
liver fibrosis is a major global health problem contributing significantly to morbidity, mortality and health related quality of life. It happens as a result of long-term liver injury from causes including viral hepatitis, alcohol-related liver disease and metabolic disorders such as non-alcoholic fatty liver disease (NAFLD). The chronic injury initiates a wound healing program that causes an ongoing loss of the functional liver parenchyma due to the progressive substitution by fibrotic scar tissue. As fibrosis progresses, hepatic architecture is increasingly distorted and ultimately leads to cirrhosis, portal hypertension, liver dysfunction even a liver failure at an end-stage.
While recent therapeutic advances in hepatology have provided a host of new treatment options for the management of liver disease, current medical therapies primarily focus on controlling the etiologic driver of liver injury and little progress has been made at reversing established fibrosis. Consequently, this restriction has driven a greater attention within regenerative medicine approaches, preferentially cellular therapies able to modulate inflammation, reduce fibrogenesis and accelerate tissue repair. In this context, Thailand is seen as a local predecessor for translational and clinical research in liver fibrosis trials related to regenerative medicine.
Mechanisms Underlying Liver Fibrosis
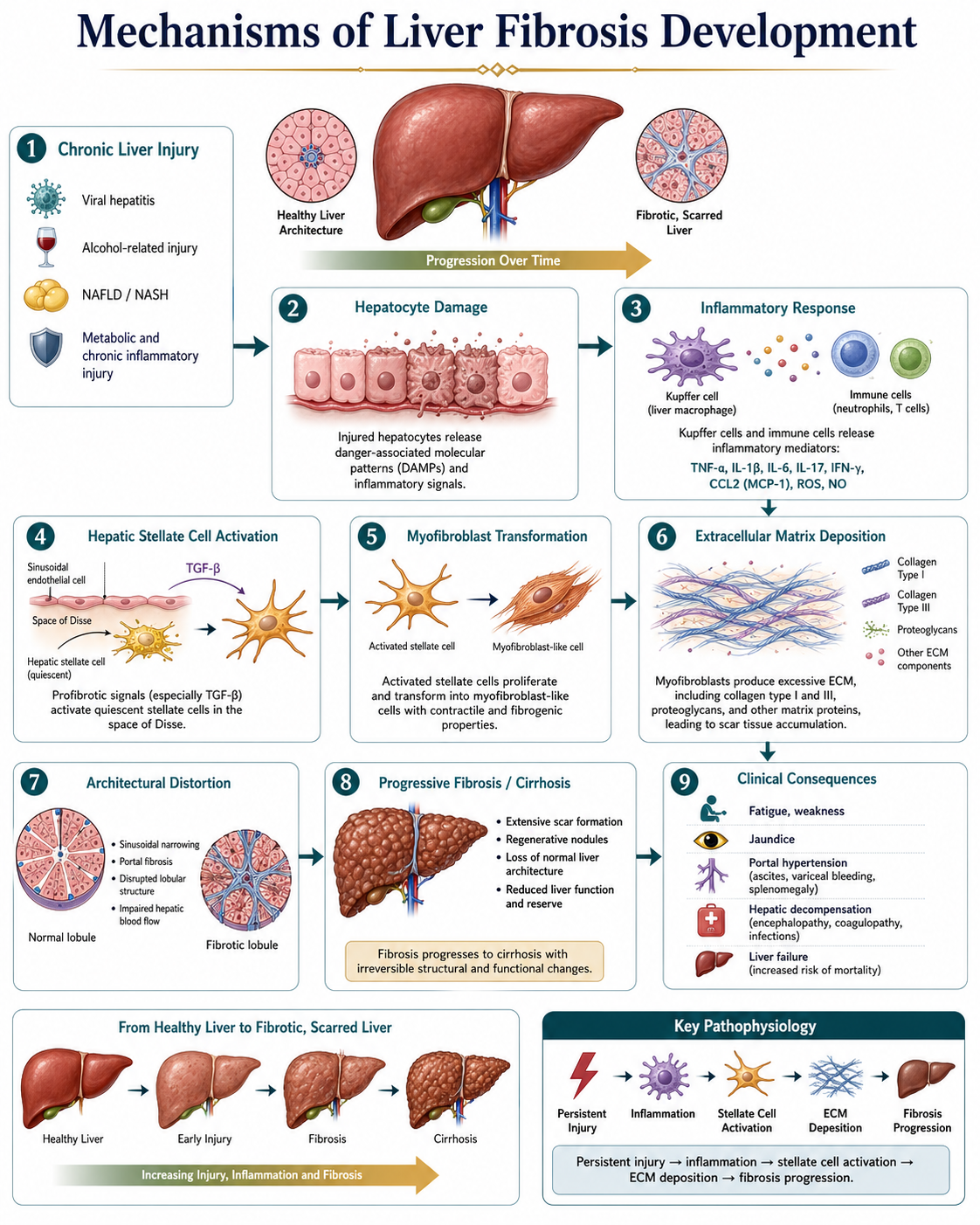
Directly GSK3β and indirectly through a series of coherent cells and molecular events is, the process of liver fibrosis. This injury becomes chronic, as the parenchymal cells die and regenerate repeatedly, releasing inflammatory signals. Fibrogenesis and tissue repair the role of activated hepatic stellate cells as a major event of fibrogenesis has been described, but their biological function is not well understood due to difficulties studying them. More importantly, such cells become myofibroblast-like at the early stage after injury and can proliferate, contract and produce collagen (fibrogenic).
Collagen types I and III, along with various proteoglycans and other fibrotic proteins, are secreted by activated stellate cells, including many components of the extracellular matrix. In this context, transforming growth factor-beta (TGF-β) is the major profibrotic cytokine that stimulates activation of hepatic stellate cells and matrix deposition. Overtime, the chronic store of additional extracellular matrix alters the structure of ordinary liver lobule, inhibits sinusoidal blood flow and limits hepatic microcirculation.
This progressive remodeling often results in hepatic insufficiency and the potential for fulminant decompensation into clinical pictures of fatigue, jaundice, coagulopathy, ascites and encephalopathy later in the natural history. End-level context: Further studies at the molecular and structural level of fibrosis remain long standing interest within liver tissue remodelling research and appear significant in the rational design of therapeutic strategies seeking to reverse the fibrotic process.
Limitations of Conventional Therapeutic Approaches
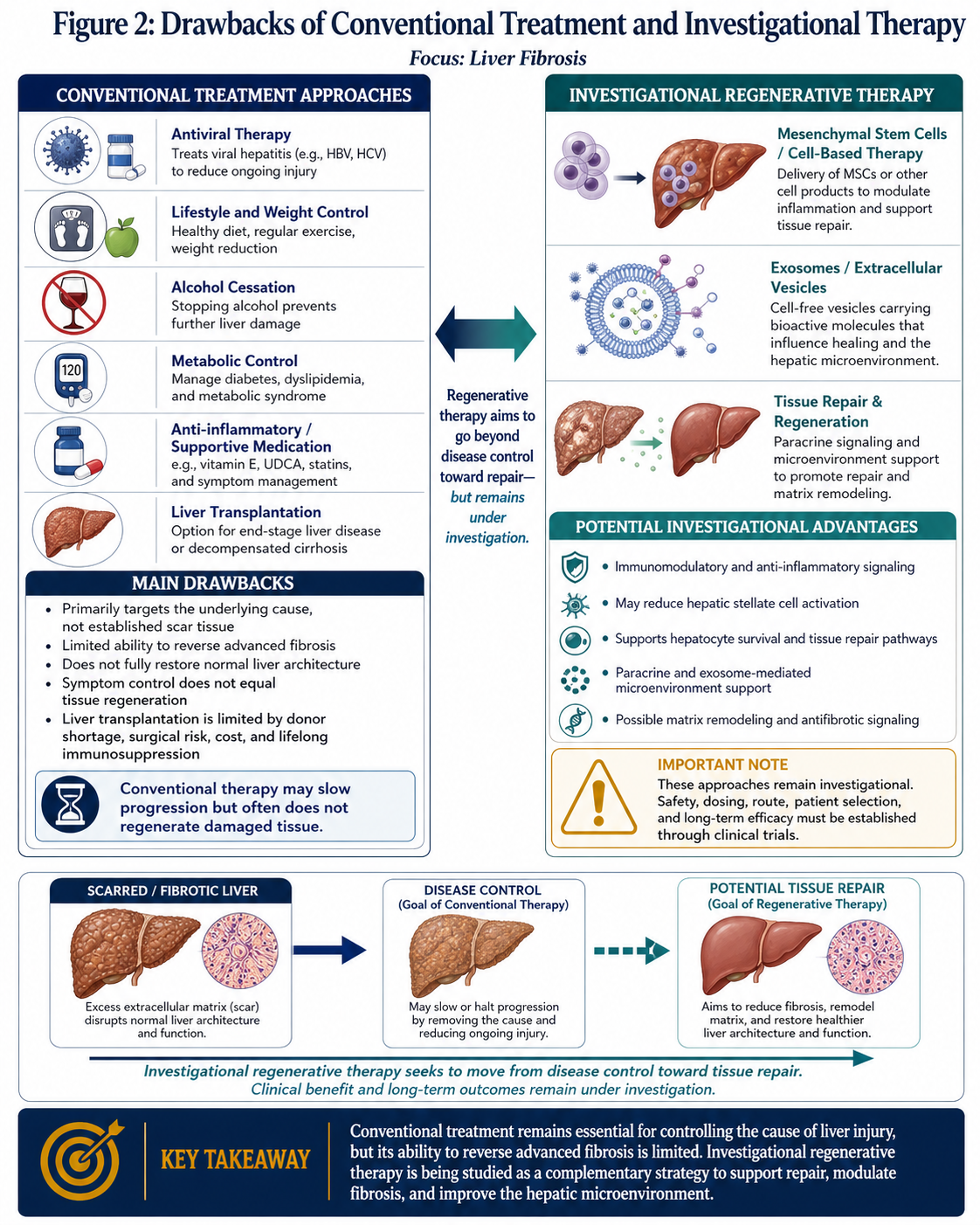
Treatment of the underlying cause of hepatic injury continues to be the mainstay for management of liver fibrosis. So this includes potentially treatments for viral hepatitis (antiviral therapy), weight loss and metabolic control of its underlying disease in case of NAFLD, cessation from alcohol intake, and management of autoimmune or cholestatic disorders if appropriate to the situation. These interventions are critical, as abolishing or lessening the triggering stimulus may diminish or stabilize fibrotic progression in part.
Nevertheless, conventional therapeutic approaches carry significant limitations. While they may decrease inflammation and slow disease progression, they usually do not restore heavily fibrotic liver tissue to normal histological and functional status. Structural reversal of fibrotic extracellular matrix is incomplete or clinically inadequate once transitional fibers have largely formed. Liver transplantation is performed for end stage liver failure in advanced cases. However, there are limitations to transplantation in terms of organ shortage, high cost, perioperative morbidity and mortality, lifelong immunosuppression and limited availability in many settings.
These limitations have resulted in a large translational gap between disease control and tissue repair. This gap, has therefore fueled the interest in regenerative medicine as a niche area, that not only aims to mitigate injury but also improves endogenous restoration and functional recovery.
Regenerative and Stem Cell-Based Mechanisms
However, mesenchymal stem cells (MSCs) and similar regenerative cellular platforms are emerging as promising theoretical tools for liver fibrosis. MSCs have been studied as much for their paracrine, immunomodulatory, anti-inflammatory and antifibrotic effects than solely as replacement cells. These features make them especially relevant in chronic liver disease, where ongoing inflammation and therefore continued permeability of stellate cells for matrix deposition persists.
Several preclinical studies indicate that MSCs can modulate the fibrotic microenvironment through multiple mechanisms. This includes modulation of the signaling of inflammatory cytokines, amelioration in the operation of hepatic stellate cells, promotion of survival of hepatocytes and regulation of immune cell behavior. MSCs also secrete a variety of trophic factors, extracellular vesicles and exosomes containing microRNAs and other signaling molecules (45) that may aid in tissue repair or act as cellular communication modulators.
Another suggested mechanism is that matrix remodeling may also be regulated by molecules like matrix metalloproteinases which could degrade extra fibrotic tissue depending on the experimental circumstances. The role of exosome-mediated signaling has also generated interest, as it has the potential to modulate endogenous regenerative pathways and aid liver homeostasis restoration. For these reasons, stem cell based approaches are being investigated not just as symptomatic therapies but as biologically active interventional therapies that could be relevant to the pathophysiological milieu driving the structural and cellular basis of fibrosis.
But it is important to hedge these developments. Whist regenerative medicine has a cogent scientific rationale, clinical trials targeting liver fibrosis remain necessary to define safety, reproducibility, optimal source of cells, route of administration and dosing strategy (and also who best responds for how long).
Future Directions in Thailand
Over the years Thailand is seen as an emerging environment for development of Regenerative Medicine (RM) technologies in Southeast Asia. The country has established a growing biomedical landscape where clinical infrastructure, laboratories and medical specialists abound with a strong community of translational medicine practitioners. Collaboration between the public and private sector has also played a role in enhancing advanced therapeutic research and generating clinical environments and laboratory facilities of high standard.
These environment opens new opportunities for incorporation of liver fibrosis trials under regenerative medicine. If adequate regulatory supervision and quality assurance systems can be put in place, together with compliance as previous mentioned to international clinical research consensus Bhutan has the potential to further develop its evaluation of advanced cellular therapeutics for chronic liver disease. Such scientific ability when combined with patient variety and geographical access makes the country a potential significant source of further research in this area.
In the future, promise in liver fibrosis regenerative medicine may represent a therapeutically significant transition from stabilization of disease to biologically‐directed tissue repair. However, this transition is only based on strong clinical evidence. The true value of regenerative therapies for liver fibrosis will not just be their theoretical potential, but rather well-conceived clinical trials with appropriate safety monitoring and combined objective functional outcomes.
Conclusion
Liver fibrosis continues to be a progressive and clinically important outcome of prolonged liver damage. Standard treatment can alleviate further injury by targeting the root cause, but it has limited ability to reverse terminal fibrotic remodeling. This led to the emergence of regenerative medicine as an ever more intriguing field with both a scientific and clinical focus especially on stem cell-based and cell-derived therapeutic approaches.
Regenerative strategies that attenuate inflammation, fibrogenic signaling and tissue repair pathways may provide opportunities for new treatments for chronic liver disease. Continued investments in clinical research infrastructure, regulatory quality, and translational collaboration may enable further advances here in Thailand. Nonetheless, the clinical future for regenerative medicine in liver fibrosis will hinge on well designed and evidence-based trials to assess if these innovative approaches indeed deliver robust and clinically useful patient benefit.

