Current rationale, clinical uncertainty and investigational potential of umbilical cord mesenchymal stem cells (UC-MSCs) & double-filtration plasmapheresis in ALS
ALS is a progressive motor neuron disease and, based on the latest evidence, care remains multidisciplinary with psychological support plus approved drugs (ruluzole, edaravone and tofersen for SOD1-related ALS). Tuesday’s conclusion is that stem-cell treatment was not a standard ALS care, and the FDA maintains that it has not approved regenerative medicine therapies for use in ALS cases.
What is the reasoning for using UC-MSCs in ALS?
Because mesenchymal stem cells modulate neuroinflammation, immune signaling and the neural microenvironment largely via paracrine and immunomodulatory effects rather than by “replacing” lost motor neurons, UC-MSCs are being investigated. Recent ALS stem-cell reviews characterized MSCs as scientifically interesting and relatively safe to investigate further, however they cautioned that efficacy is unknown, and protocols remain diverse.
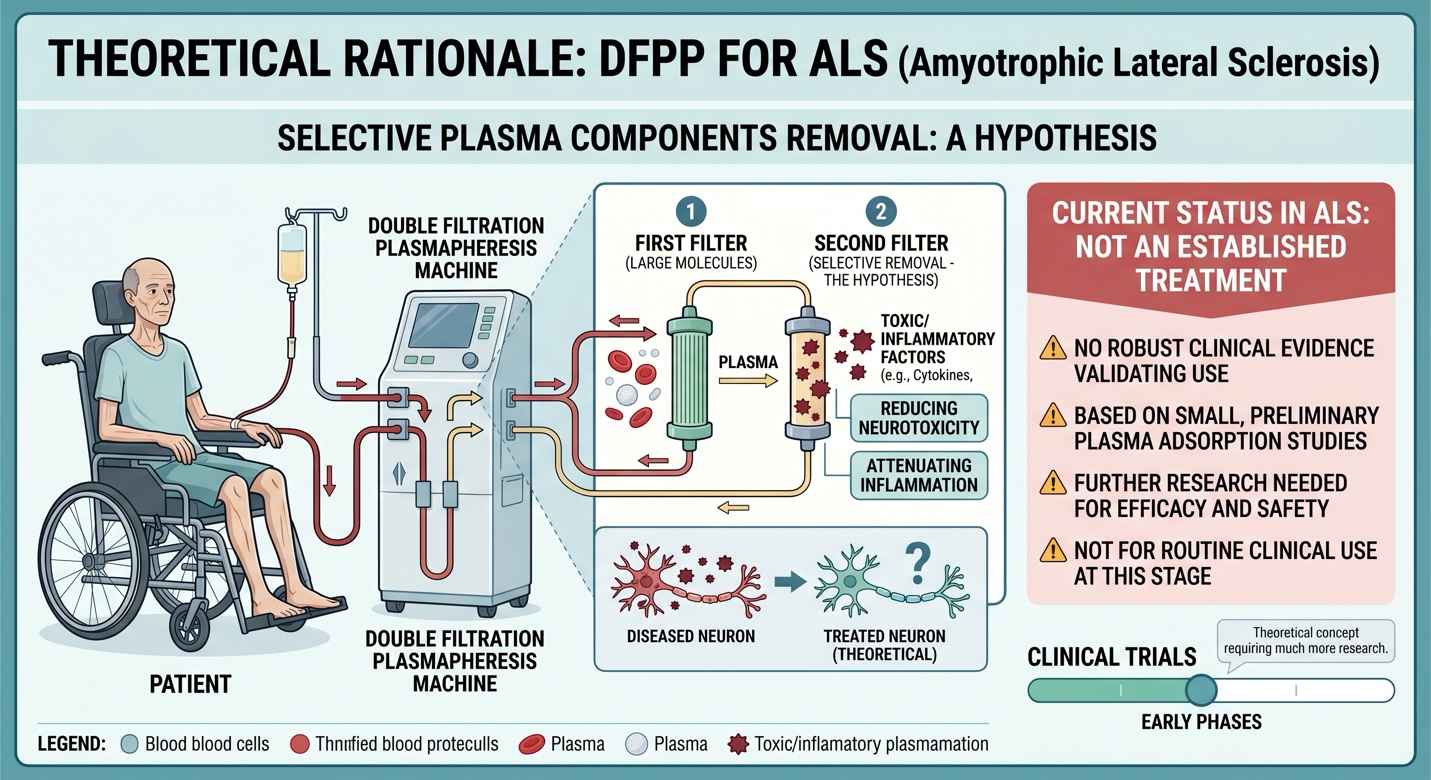
DFPP in ALS: Why would you do it?
The theoretical basis of DFPP is distinct: It seeks to eliminate certain plasma constituents, which some investigators hypothesize may contain factors that mediate inflammation or toxicity. But as a reminder: DFPP is not an FDA-approved treatment for ALS. I searched for ALS clinical evidence that proved DFPPs were a validated treatment. The only human signal that I could find in a plasma adsorption paper was a small preliminary study suggesting short-term effects, which is nowhere near enough evidence to warrant routine use of ALS.
What is the role of UC-MSCs and DFPP?
Scientifically, the paired modality can be conceptualized as a hypothesis-driven investigational scheme: DFPP may be viewed as an intervention that aims to alter the circulating inflammatory milieu whereas UC-MSCs may be considered an intervention that imparts downstream immunomodulatory and neuro-supportive signaling. It is a possible theory, but right now it is far stronger as idea than as clinical fact. It was not that more robust clinical evidence has demonstrated the efficacy of the UC-MSC + DFPP combination in treating ALS.
The most important caution
Even stem-cell products reaching furthest in ALS trials have failed to provide strong enough evidence to proceed into routine therapy. For debamestrocel (an MSC-based ALS product), randomized placebo-controlled studies were both described as failing to demonstrate efficacy on the primary endpoints in FDA briefing materials and the FDA determined that there was no substantial evidence of effectiveness for ALS from the available clinical data. That does not mean all of the MSC research is garbage, but it does indicate that claims should be cautious.

