Why does this topic get so much attention
When people use ” stem cell therapy Thailand” to look up ovarian health, they’re usually not longing for abstract lab science. They’re in search of a genuine reply to a quite human fear: diminishing ovarian reserve, fertility treatment with poor outcome anyway, or ovarian function that may be going beyond what was planned. From a scientific perspective, the discussion always involves ovarian ageing, diminished ovary reserve, and premature ovarian insufficiency; all of these are closely related by way the follicular pool ceases functioning and/or fails to form properly. But the trouble in this field is that it’s both too heartfelt and too complicated at the same time–exactly why we need calm interpretation rather than breezy certainty.
The word follicular matters more than it may seem. Ovarian function is not just about whether ovulation happens. It depends on the health of follicles at multiple stages, the quality of the oocyte inside them, the surrounding granulosa cells, the vascular environment, and the hormonal signals that support maturation. Recent reviews on ovarian aging describe the process as a decline in both follicle quantity and quality, with the rate of follicle depletion accelerating over time and the ovarian microenvironment becoming more fibrotic, inflamed, and less supportive of follicle survival.
Understanding the ovarian side first
- Ovarian reserve is not the same as fertility
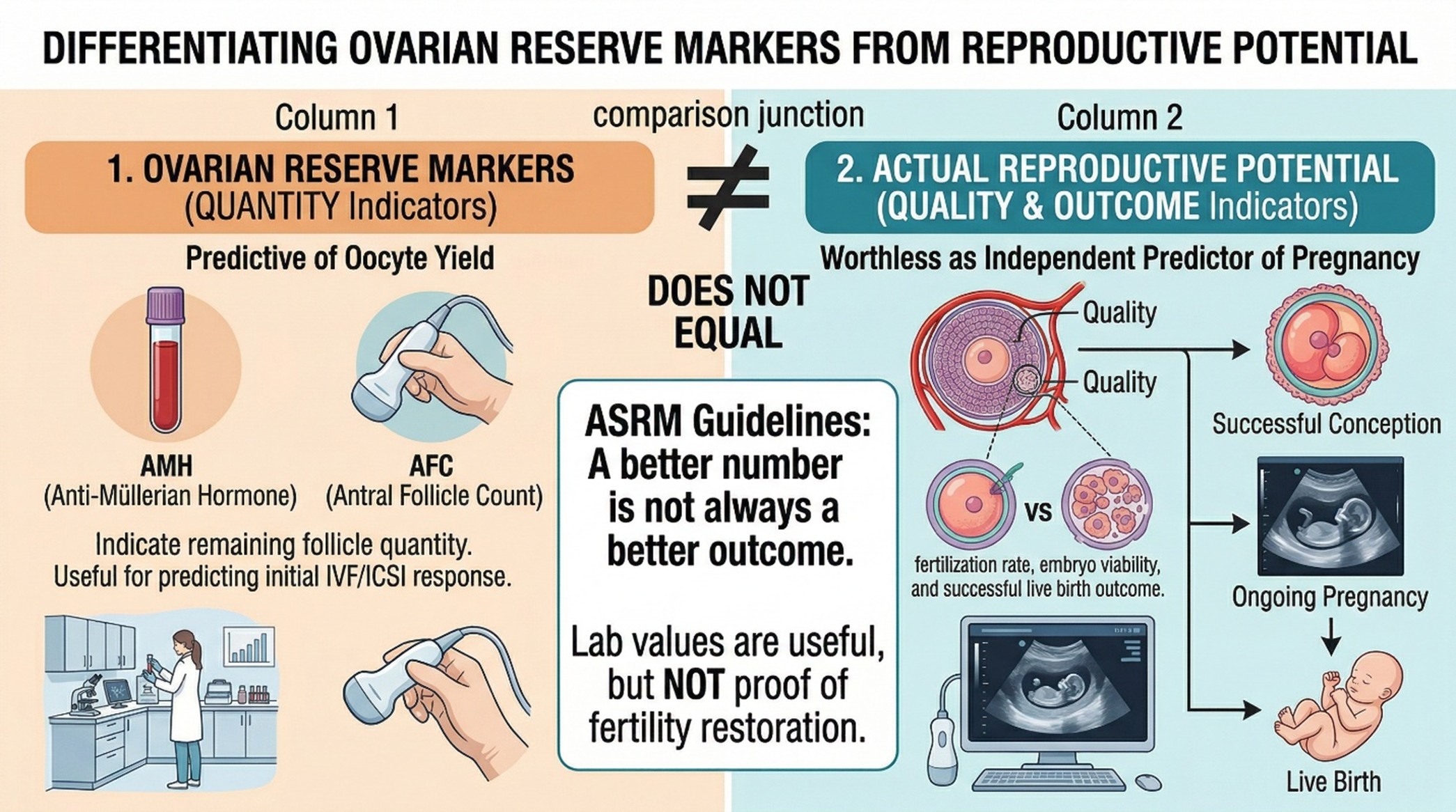
The ASRM provides one of the most helpful clarifications in this entire field: Ovarian reserve refers to the number of eggs remaining in the ovary, not a guarantee for reproduction. Currently, AMH and antral follicle count are the simplest, most sensitive markers of ovarian reserve, but ASRM also states that these markers are good predictors for oocyte yield and a worthless independent predictor of reproductive potential.
That distinction is extremely important in any stem cell therapy Thailand discussion, because improvements in a lab value or ultrasound count do not automatically mean improved live birth outcomes.
- Follicular biology is where the real story sits
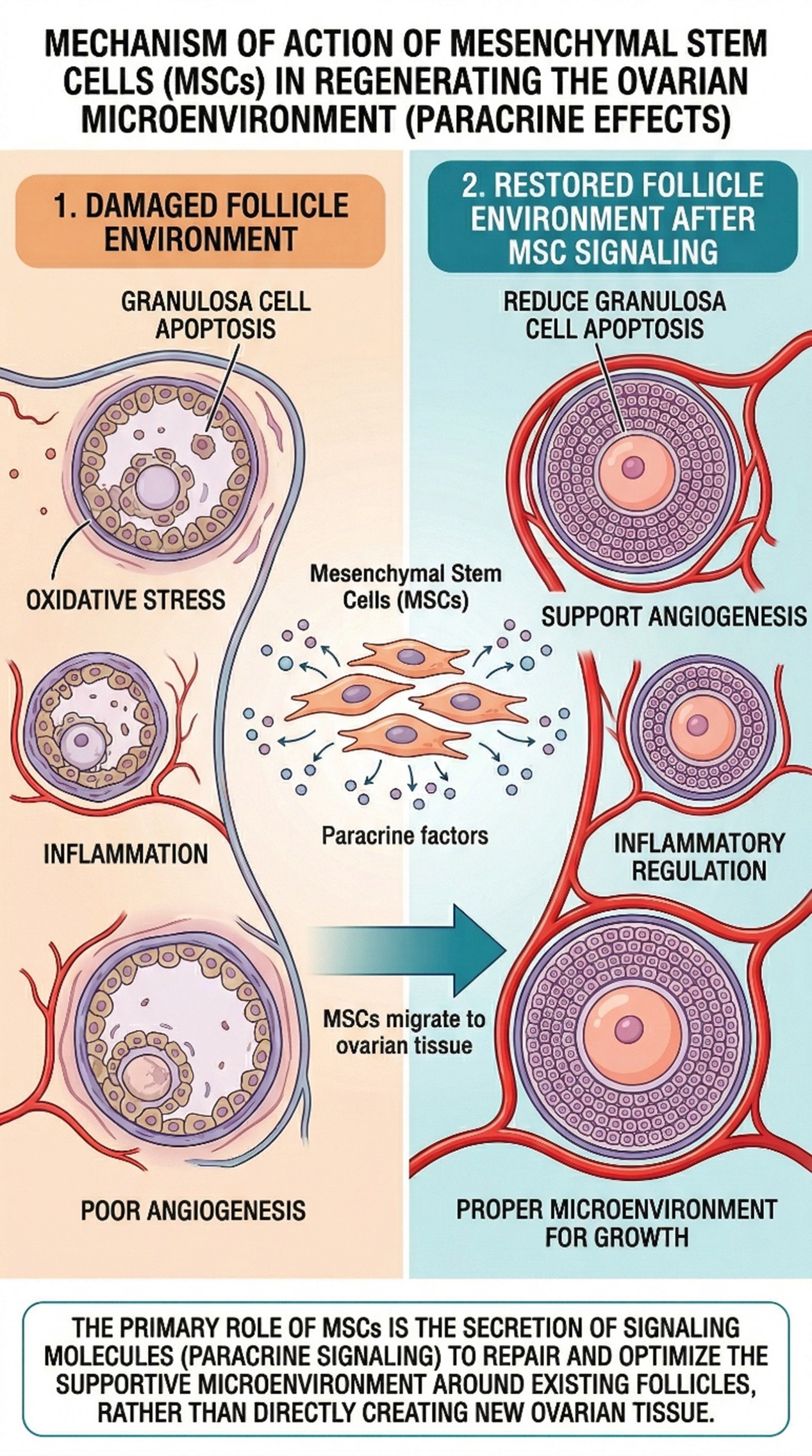
The ovarian follicle is not just a container for an egg. It is a dynamic microenvironment that supports oocyte development, hormone production, and coordinated maturation. In ovarian aging and premature ovarian insufficiency, the system becomes less resilient. Follicular development can be disrupted by granulosa-cell injury, oxidative stress, inflammation, reduced angiogenesis, and changes in the ovarian niche. This is why the secondary keyword follicular fits naturally in this topic: most regenerative claims in ovarian medicine are really claims about protecting or restoring the follicular environment.
Where stem cell therapy enters ovarian medicine
- The scientific rationale is real
The reason stem cells continue to be studied in ovarian medicine is not hard to understand. According to reviews published in 2025, mesenchymal stem cells are thought to have relevance in ovarian dysfunction because they might migrate toward the injured ovarian tissue. They can help hormone production and thus reduce apoptosis in granulosa cells. They are also able to support angiogenesis, make proper inflammatory regulation possible at sites where initialize to develop into follicles, and present an environment that serves for such growth. It should be emphasized that most of these plausible roles are believed to reside exclusively in paracrine signaling, rather than a matter of simply creating fresh ovarian tissue. That makes the field scientifically interesting, even if it does not make it clinically settled.
- Why “ovarian rejuvenation” became such a powerful phrase
There is a reason phrases like “ovarian rejuvenation” and stem cell therapy Thailand have become so prominent online: they sound as though modern reproductive medicine may finally be able to reopen a window that biology was closing. But even ASRM’s own 2026 roundtable on ovarian aging describes “ovarian rejuvenation” as unproven. Those wording matters. It does not dismiss the research. It simply acknowledges that exciting mechanisms and small early studies are not the same thing as an established fertility treatment.
What the current evidence actually supports
- There are early human studies, but the field is still investigational
This is the part that needs the most discipline. Reviews of cellular therapy in primary ovarian insufficiency and poor ovarian response continue to evaluate efficacy, safety, and limitations, which tells you immediately that the field is still under active investigation rather than settled clinical routine. Other recent reviews note that stem cell approaches show promise in preclinical models and early clinical reports, including signals in ovarian structure, hormone levels, and follicular development, but they also emphasize that the evidence base remains fragmented. Different studies use different cell sources, routes, endpoints, and patient populations, which makes comparison difficult.
- Guidelines remain much more cautious than marketing
The most important reality check comes from the current POI guideline developed by ASRM and collaborating societies. It states that women with POI should be informed that there are no interventions that have been reliably shown to increase ovarian activity and natural conception rates. That is a very strong sentence, and it should sit near the center of any honest article on ovarian stem cell claims. Likewise, a European recommendations document on add-ons in reproductive medicine lists stem cell therapy for premature ovarian insufficiency or diminished/poor ovarian reserve as not recommended for routine clinical use.
- Follicular improvement is not the same thing as proven pregnancy benefit
This is where many articles become too eager. A study may report changes in AMH, estradiol, FSH, antral follicles, or ultrasound appearance. Those findings can be interesting. They can even be clinically meaningful in the research setting. But they are not automatically the same as consistent improvement in pregnancy or live birth. ASRM’s ovarian reserve opinion is very useful here because it repeatedly separates follicular markers from broader reproductive potential. In plain language: a better number is not always the same as a better outcome.
- The follicular pool can sometimes be measured, but not fully “reset.”
Because follicular language sounds technical, it often persuades people quickly. But the biology is more modest than many headlines suggest. Follicles are finite, ovarian reserve declines with age, and the ovarian microenvironment also changes over time. Current reviews describe follicular depletion, oxidative stress, fibrosis, and altered immune signaling as part of ovarian aging. That is exactly why researchers are interested in stem cells, but it is also why blanket reset claims should be treated carefully. The problem is not just one missing switch. It is an entire aging tissue context.
- Better questions lead to better decisions
In practical terms, the most helpful questions are not “Can stem cells fix my ovaries?” but rather: Which specific disease is being treated? POI, diminished ovarian reserve, poor ovarian response, or a woman who ate her egg count, as well as age-related ovarian decline, are not identical disorders. What criteria are considered successful? Hormones, antral follicle count, oocyte yield or spontaneous ovulation, the number of embryos transferred, and miscarriage rate and pregnancy rate are all quite different. And as for the guideline landscape? At present, the best available guidance on this subject is still rather more cautious than what might be inferred from some of the shriller celebrations in stem cell therapy Thailand.
- The most honest conclusion
The honest conclusion is neither dismissive nor overexcited. There is a real scientific basis for studying stem cells in ovarian medicine, especially around the follicular microenvironment, granulosa-cell survival, angiogenesis, inflammation control, and endocrine support. But there is still a major gap between mechanistic promise and reliable routine clinical benefit. For now, the strongest evidence says this field is investigational, ovarian rejuvenation remains unproven, and any serious discussion of stem cell therapy Thailand should be grounded in that reality first.
Frequently Asked Questions
1) What does “ovarian” usually mean in stem cell discussions?
Usually, it refers to ovarian aging, diminished ovarian reserve, poor ovarian response, or premature ovarian insufficiency rather than one single disease category. In practice, the conversation often revolves around whether ovarian function and the follicular environment can be supported or partially restored.
2) Why is the keyword “follicular” important here?
Because follicles are the functional units that support oocyte development and hormone production. Many regenerative claims in ovarian medicine are really claims about improving follicular development, reducing granulosa-cell injury, or supporting the ovarian microenvironment.
3) Does stem cell therapy reliably increase natural conception in POI?
Current international guidance says no intervention has been reliably shown to increase ovarian activity and natural conception rates in women with POI. That is why stem cell approaches remain investigational rather than standard care.
4) Are stem cell treatments for poor ovarian reserve recommended for routine use?
Not for routine clinical use. European recommendations on add-ons in reproductive medicine list stem cell therapy for POI or diminished/poor ovarian reserve as not recommended for routine clinical use.
5) If AMH or antral follicle count improves, does that mean fertility is restored?
Not necessarily. ASRM notes that AMH and AFC are useful markers of ovarian reserve and expected oocyte yield, but they are poor independent predictors of reproductive potential and should not be treated as direct proof of fertility restoration.