Why this topic needs a calmer, more honest explanation
When families search regenerative medicine Thailand and autism, they are usually not searching for theory. They are searching for possibility. Autism spectrum disorder is a neurodevelopmental condition that affects how people communicate, interact, learn, and behave, and it is also highly diverse from one person to another. That diversity is exactly why this topic deserves more precision than marketing usually gives it. The science is real enough to discuss seriously, but not mature enough to flatten into simple promises.
What stem cell research in autism is actually studying
Most clinical studies are not about neural stem cells
It is the first distinction that counts. Real-world autism trials tend to focus on umbilical cord blood cells or mesenchymal stromal/stem cells (MSCs) — not neural stem cell. Journals describing clinical research in autism year-on-year continue to characterize the early work with MSCs and related cell therapies as “exploratory,” most reports are small, heterogeneous studies reporting safety plus short-term behavioral scales rather than lasting clinical outcomes. So the term regenerative medicine Thailand is perhaps appearing like expansive, but the scientific R&D base from inside of it actually is more narrower and quite certain.
Neural stem cells matter more in the research lab than in current clinical care
The keyword neural stem cells is scientifically interesting, but it needs to be used carefully. Right now, neural stem cells are much more important in preclinical research and disease modeling than in established autism treatment. Reviews on autism and stem cell models describe induced pluripotent stem cells and neural progenitor or neural organoid systems as valuable tools for studying how neurodevelopment changes in autism and for testing future therapeutic ideas. More recent animal-model work with neural stem cells is still early and preclinical. That means neural stem cells are absolutely part of the autism research conversation, but they are not the same thing as a clinically validated autism therapy.
Why stem cells became part of the autism conversation at all
The biological rationale is broader than “replacing brain cells”
This is where many articles become too simplistic. The clinical interest in stem cells for autism has generally not been based on the idea that doctors are replacing missing neurons in a straightforward way. The more common rationale in published reviews is that certain stem-cell-based approaches, especially MSCs, may affect inflammation, immune signaling, trophic support, or the broader neural environment. That is a very different claim from “repairing autism,” and it is a more defensible one scientifically.
Early trials suggest feasibility, not settled efficacy
The widely cited report Abstract A phase I trial of intravenous human umbilical cord tissue mesenchymal stromal cells in children with autism states that infusions are safe and feasible, thus warranting a transition to a second-phase efficacy testing. Later systematic reviews and meta-analyses indicated possible effects on the CARS (Childhood Autism Rating Scale) and similar behavioral scales, but they also noted ongoing weaknesses in the field: limited sample numbers, differing cell sources [if a stem-cell-based therapy is produced], treatment schedules ranging from weeks apart to much longer intervals across studies, variability in study design. That seems to be the most reasonable way of evaluating evidence available today. You see signal, but with a level of uncertainty.
Where the current evidence is strongest — and where it is weakest
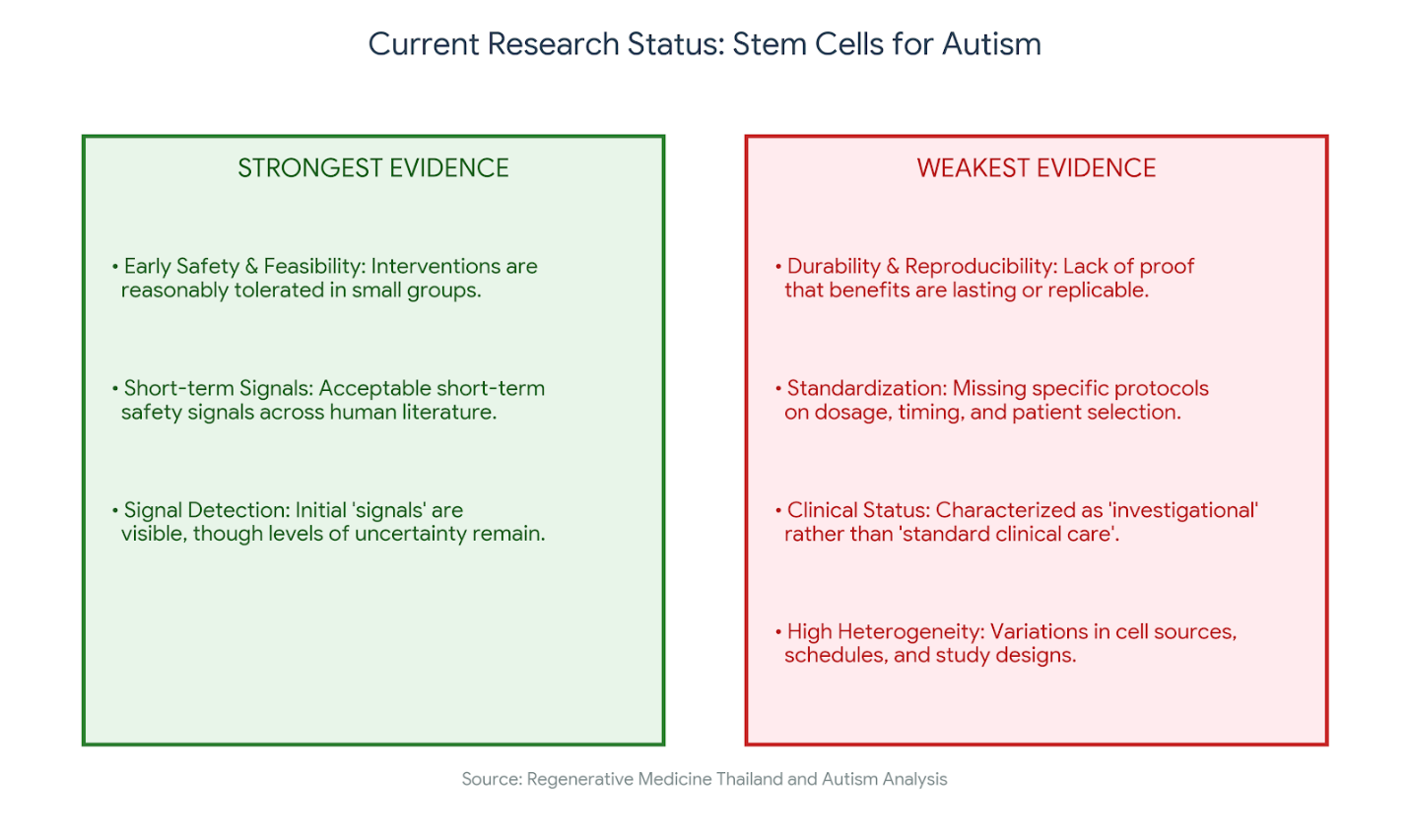
Strongest: early safety and feasibility
Across the existing human literature, the most consistent message is that early stem-cell studies in autism have mainly been trying to answer whether these interventions appear feasible and reasonably tolerated in small groups. Reviews of autism cell-therapy studies generally describe acceptable short-term safety signals, but they also stop well short of saying the field has achieved routine clinical status. That difference matters, especially when the wording on some websites sounds much more certain than the papers do.
Weakest: proof that benefits are durable, reproducible, and clinically standardized
The weakest part of the field is still precisely what families want to know most: Who benefits, how much (specifically), for how long and under which protocol. Although the pooled results of the 2022 meta-analysis were promising, this does not eliminate the inherent heterogeneity of trials. Subsequent review literature still characterizes autism stem cell therapy as investigational, rather than standard care. Regenerative medicine Thailand and autism is a subject you could always reflect in any piece of information.
What a trustworthy clinic conversation should sound like
More specific than hopeful
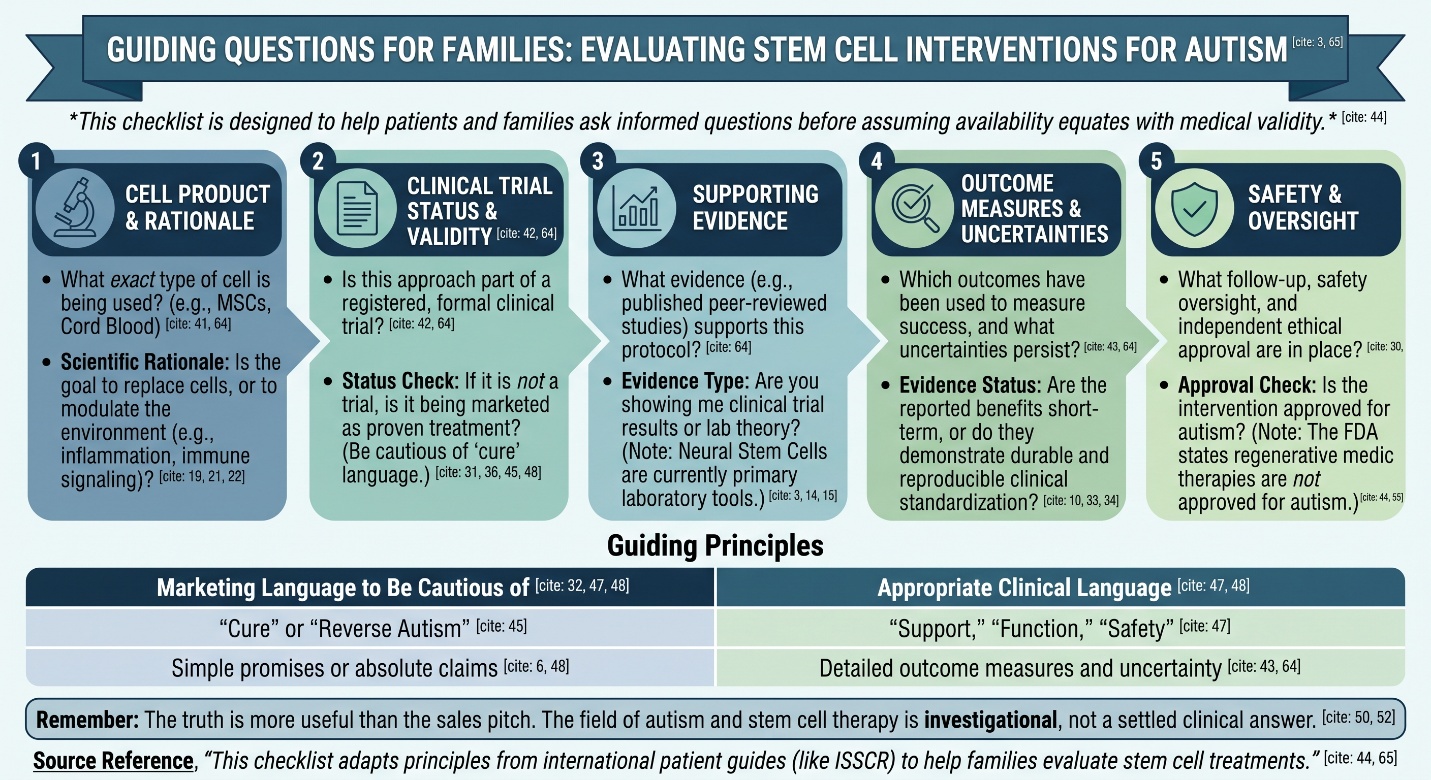
An appropriate regenerative medicine Thailand speech should be tailored to the point where individuals do not speed up, they slow down. For example, it should clarify what cell type is involved (if relevant); whether the approach forms part of a formal clinical trial; which outcome measures have been used and any uncertainties that persist. According to the FDA, regenerative medicine therapies are not approved for autism and that is why the ISSCR patient guide was designed: To assist patients and their families in asking more informed questions regarding stem cell treatments or clinical trials before falsely assuming availability equates with validity.
More respectful than “cure” language
This part matters too. Autism is a neurodevelopmental condition, not a simple lesion waiting to be reversed. That is one reason serious sources frame the conversation around support, function, safety, and evidence rather than promises of cure. When the language becomes too absolute, it usually tells you more about the marketing than the medicine.
The most honest conclusion
The truth is more useful than the sales pitch. Stem cell research in autism is active, and some early clinical studies — mostly involving MSCs or cord-blood-related approaches — suggest feasibility and possible short-term improvement on selected behavioral scales. But neural stem cells currently belong far more to the worlds of laboratory modeling, mechanistic research, and early preclinical exploration than to routine autism care. So if someone asks what the best current reading of regenerative medicine Thailand and autism should be, it is this: the science is interesting, the questions are legitimate, but the field is still investigational and not yet a settled clinical answer.
Frequently Asked Questions
1) Are stem cell treatments for autism already approved?
No. The FDA says regenerative medicine therapies have not been approved to treat autism.
2) What kinds of stem cells are usually studied in autism trials?
Most published clinical studies have focused on umbilical cord blood cells or mesenchymal stromal/stem cells, not neural stem cells.
3) Do clinical studies show any benefit at all?
Some small studies and meta-analyses have reported improvements on standardized behavioral scales, but the evidence is still limited by small sample sizes, mixed protocols, and lack of long-term standardization.
4) Where do neural stem cells fit, then?
Right now, neural stem cells are more important in autism research models and preclinical studies than in established clinical treatment. They help scientists study neurodevelopment, test hypotheses, and explore future therapeutic ideas.
5) What should families ask before considering any stem cell clinic?
They should ask what exact cell product is being used, whether the treatment is part of a registered clinical trial, what evidence supports that protocol, what outcomes are measured, and what follow-up and safety oversight are in place. The ISSCR patient guide is specifically designed to help families ask those questions.