Figure A: Regenerative Medicine Framework for UC-MSC Support in Psoriasis
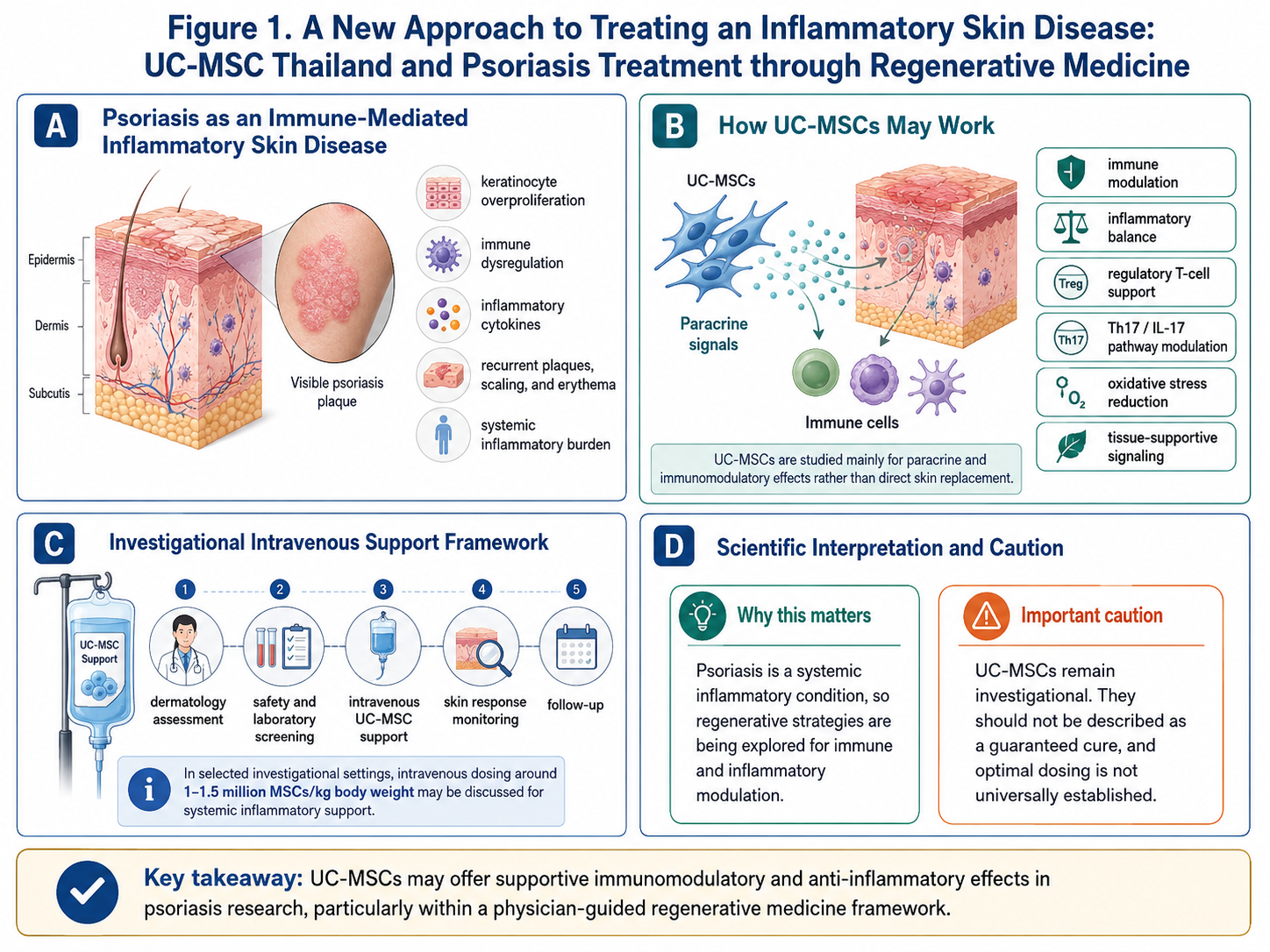
Psoriasis, is often identified by the factual. Heavily scaled and red skin plaques along with itching that recurrence but psoriasis is not a skin disease when viewed from an academic perspective. It is a chronic immune-mediated inflammatory disease with complex interactions between both the classical immune system and keratinocytes along with numerous other inflammatory cytokines and systemic pathways. Central inflammatory networks in psoriasis such as the IL-23/Th17/IL-17 axis have been identified to play a central role in the disease, and have therefore become targets of interest within contemporary psoriasis research.
Since psoriasis is a chronic and relapsing disease, a number of patients look for supplementary complementary treatments that may assist topical therapy, phototherapy, oral immunosuppressants or biologic therapy. The above explains to one extend as why UC-MSC Thailand, stem cell for psoriasis and UC-MSCs for inflammatory skin disease have been some invisible neglected area of search on internet amongst patients looking forward to regenerative medicine.
Nevertheless it needs to be addressed with caution. They should not promise a definitive cure for psor- iasis, nor be presented as a substitute for dermatology care. A more realistic explanation involves further investigation into the ability of UC-MSCs to contribute to immune modulation, inflammatory homeostasis- restoring signaling, as well as systemic tissue supportive signalling.
Introduction to UC-MSC Thailand for Psoriasis
Umbilical cord-derived mesenchymal stem/stromal cells (UC-MSCs) play an important role in regenerative medicine due to their secretion of bioactive factors that mediate immune activities, inflammation, oxidative stress, tissue repair signaling and cellular communication.
Such findings are potentially relevant in psoriasis, which is characterized by immune dysregulation, aberrant inflammatory signaling, epidermal hyperactivation and chronic cutaneous inflammation. Psoriasis: Human umbilical cord-derived MSCs in clinical Research due to their immunomodulatory properties. Conclusions: A phase 1/2a single-arm study was done including patients with psoriasis after infusions if intravenous injection and found no apparent serious treatment-related safety issues during the follow-up of the study although emphasized that larger controlled studies are required for conclusive results.
Patients seeking UC-MSC Thailand are generally interested in medically supervised Regenerative Programs, International Coordination and Lab-supported Cell Prep. However, at the end of the day medical quality, physician assessment, cellular testing and honest patient education trumps location or marketing language.
Potential Mechanisms by Which UC-MSCs Work in Psoriasis
UC-MSCs are least referred here as they explicitly “replenish” injured skin. The more scientific explanation is paracrine signaling. Therefore, UC-MSCs can release a variety of growth factors, cytokines, extracellular vesicles and other bioactive molecules that will interact with nearby tissues and immune cells.
In psoriasis research, the mechanisms involved with UC-MSCs are being investigated for their possible affects on:
immune modulation
inflammatory balance
Th17-related inflammatory activity
regulatory T-cell support
cytokine signaling regulation
oxidative stress reduction
skin microenvironment support
tissue repair communication
The mechanism of these drugs is of academic interest because in contrast to psoriasis simply being a skin surface aberration, it is mediated by immune dysregulation. In a phase 1/2a study conducted in individuals with diabetes over the course of roughly six months, evidence of immune changes following UC-MSC treatment was identified, as follows: changes in Treg cells (not observed) and Th17 levels at baseline and response status; however, further confirmation is still warranted.
Why systemically delivered UC-MSCs are discussed
Intravenous method of UC-MSC is mostly talked about, since psoriasis itself can be regarded as a systemic inflammatory disease; not only localized cutaneous disorders. When the purpose of therapy is not solely local skin repair, intravenous delivery can be proposed – for deconstruction to systemic immune and inflammatory modulation.
Investigational protocols Some investigational protocols mention a dose of approximately 1–1.5 millions MSCs per kg BW particularly for intravenous inflammatory support. In a human psoriasis study, the 1st stage protocol (Stage-1) used target dose of 1.5 × 10⁶ unmanipulated UC-MSCs/kg/infusion with prolong repeat infusion according to the design of the study, Some other dose groups were also considered and the investigators cautioned that because of limited long-term safety data, the need for caution with global dose standards.
Hence, it is excellent to say this dose gives the best result but rather more academic and safe would be:
Intravenous injection of UC-MSCs (about 1–1.5 million MSCs per kg body weight) could be occasionally considered as an experimental systemic immunomodulation treatment on an individual basis with patients with psoriasis, while conclusions cannot yet be drawn regarding the best-dose at this time point worldwide.
Evaluate What Before Supporting UC-MSC?
The first step to start a responsible UC-MSC Thailand psoriasis treatment chain or figured out by doing this is based on thorough medical survey. Important assessments may include:
psoriasis type and severity
PASIs score or the body surface area infected
flare frequency
itching, scaling, and plaque distribution
previous medication response
history of biologic therapy
psoriatic arthritis symptoms
autoimmune background
infection screening
liver and kidney function
inflammatory markers
metabolic profile
current medications
dermatology consultation
This assessment aids clinicians in determining if the patient has localized mild disease, moderate-to-severe plaque psoriasis, systemic inflammation, treatment-refractory disease or probable psoriatic arthritis. Regenerative support should not be viewed as a universal approach, thus patient selection is crucial.
A Regenerative Framework Supporter: UC-MSC Thailand
An example of a medically responsible framework would be the following:
Step 1: Dermatology assessment → Step 2: Inflammatory and safety screening → Step 3: UC-MSC support under the guidance of physicians → Step 4: Skin response monitoring to optimize doses and timing → Step 5: Long-term follow-up
A more robust structure than the presentation of UC-MSC as a single miracle therapy. Psoriasis is a chronic condition with the potential for long-term management, including skin care, trigger avoidance, lifestyle support and dermatology-specific systemic medications.
Regulatory caution is also important. The U.S. FDA has issued warnings regarding certain regenerative medicine products (including stem cells & exosomes) that are being marketed with unsubstantiated claims, or without evidence and oversight from the FDA;
Conclusion
Since psoriasis is an immune-mediated chronic inflammatory disease with systemic biological characteristics, UC-MSC (umbilical cord-derived mesenchymal stem cells) Thailand and other MSC-based therapies such as ucMSC for psoriasis have strongly now become major concerns in the regenerative medicine field. UC-MSCs have been studied for their possible immunomodulatory, anti-inflammatory and paracrine signalling functions.
In some experimental settings for inflammatory modulation, an intravenous injection of UC-MSCs protocol consisting of ~1–1.5 million/kg body weight might be considered, with one intervention in psoriasis delivering 1.5 × 10⁶/kg. Even UC-MSCs should not be characterized as a universal cure, and no optimal dose has been defined.
The most legitimate way would be to have very controlled, accurate patient selection, physician oversight and quality-controlled cell preparation with realistic expectation and follow up. Responsible regenerative medicine in psoriasis care should enable scientific innovations while being forthright about limitations.