A Neurological disorder can affect how a person moves, speaks, thinks, remembers, sleep, and interacts with the world. Other conditions have a slow course over the years. Some occur acutely after injury, a stroke, or an inflammation (or immune attack). The hardest time for patients and families is not only when the patient gets diagnosed, but also what happens after.
This is one reason there are so many instances these days where the phrase stem cell appears in discussions touching on neurology. When standard care feels limited, anyone living with Parkinson’s disease, multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS), spinal cord injury or other neurological illness often looks for more options. There are exciting scientific approaches in regenerative medicine, but the language is also a careful one.
A sensible discussion of stem cells and Neurological disorder should never sound like a promise of a miracle. The nervous system is complex. Skin and bone can heal, but neurologic tissues of the brain and spinal cord do not usually undergo such repair. Potential future applications of stem cell science for neurological care must be viewed in the context of data, safety, patient selection, and realistic expectations.
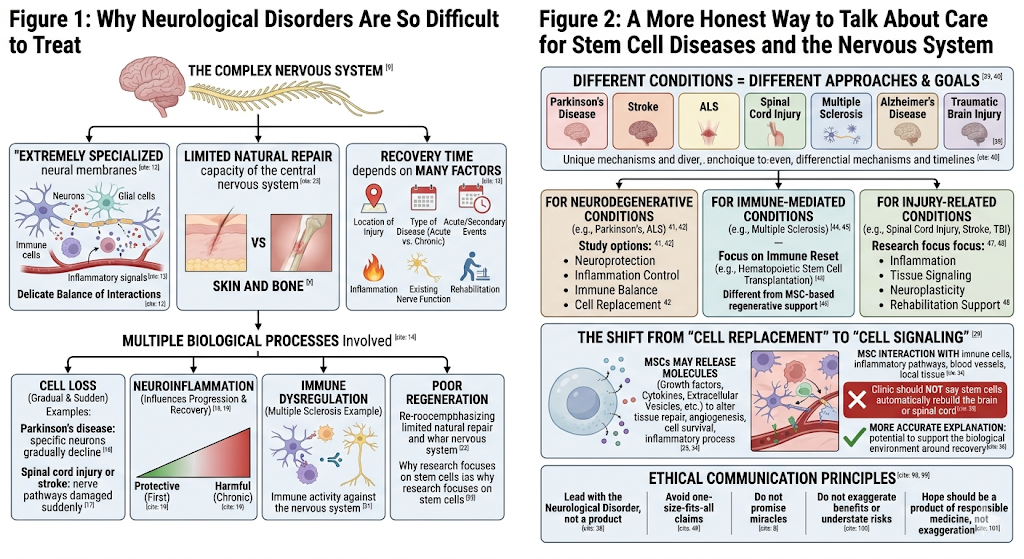
Why Neurological Disorders Are So Difficult to Treat
The neural membranes are extremely specialized. Neurons and glial cells interact with immune cells, blood vessels, and inflammatory signals in the most delicate balance. The time taken for recovery depends largely on the location of injury, type of disease (acute or chronic) and acute/secondary events within the healing process, including timing ageing inflammation, existing nerve function, rehabilitation, etc.
Multiple Biological Processes Involved with a Neurological Disorder:
Cell Loss
In diseases such as Parkinson’s disease, specific neurons gradually decline. In spinal cord injury or stroke, nerve pathways may be damaged suddenly.
Inflammation
Neuroinflammation can influence disease progression and recovery. In some cases, inflammation may be protective at first but harmful when it becomes chronic.
Immune Dysregulation
Conditions such as multiple sclerosis involve immune activity against parts of the nervous system.
Poor Regeneration
The central nervous system has limited natural repair capacity. This is one reason researchers are interested in stem cell approaches for neurological conditions.
Stem Cell Science in Neurology: What Is Actually Being Studied?
Stem cells can also mean so many things. In the arena of neurological research, one encounters at least a few cell types in which scientists have interrogated: mesenchymal stem/stromal cells (MSC); neural-stem-cells; induced pluripotent stem cells or iPSCs, and/or any derived specialized field that is extrapolated from stem cells.
The immunomodulatory properties of mesenchymal stem cells (MSCs) are the subject of a 2025 review in Signal Transduction and Targeted Therapy. They found that these MSC effects might be due to the presence of growth factors, cytokines, extracellular vesicles, and other bioactive molecules altering tissue repair rate, angiogenesis degree/quality component, cell survival factor components, or inflammatory process characteristics.
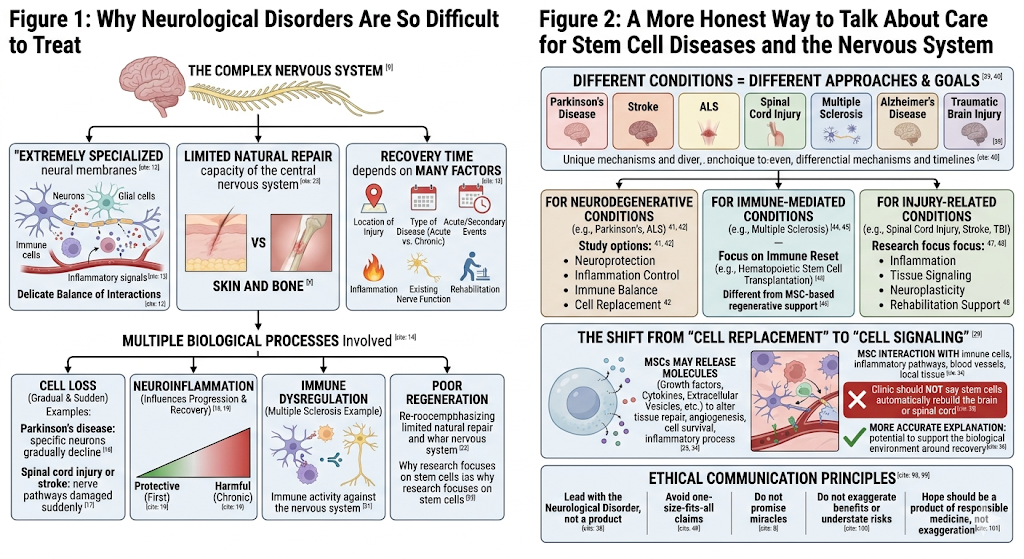
The Shift From “Cell Replacement” to “Cell Signaling”
Many patients imagine that a stem cell enters the body, travels to the brain, and becomes a new nerve cell. In some advanced research fields, cell replacement is truly being studied. For example, researchers are exploring stem-cell-derived dopamine neurons for Parkinson’s disease.
But for many neurological applications, especially MSC-based approaches, the main scientific discussion is different. It is more about signaling than replacement. MSCs may release molecules that interact with immune cells, inflammatory pathways, blood vessels, and local tissue environments.
This is important because a clinic should not say that stem cells automatically rebuild the brain or spinal cord. A more accurate explanation is that stem cell therapy is being explored for its potential to support the biological environment around recovery.
A More Honest Way to Talk About Care for Stem Cell Diseases and the Nervous System
The conversation regarding regenerative medicine should lead with the Neurological disorder first, and never come in on a product. Parkinson’s disease, stroke, ALS, spinal cord injury, multiple sclerosis, Alzheimer’s disease, and traumatic brain injury are all different conditions. Each of these things will be with different mechanisms and timelines, as well as goals of care.
For Neurodegenerative Conditions
For example, in the case of Parkinson’s disease or ALS, a study involving neuroprotection versus inflammation control vs immune balance v/s cell replacement might be conducted. Nonetheless, these strategies are still extremely dependent on the conditions.
For Immune-Mediated Conditions
In multiple sclerosis, some stem cell-related approaches, such as hematopoietic stem cell transplantation, are more focused on immune reset. This is very different from MSC-based regenerative support.
For Injury-Related Conditions
In spinal cord injury, stroke, or traumatic brain injury, research may focus on inflammation, tissue signaling, neuroplasticity, and rehabilitation support.
This is why any article about stem cells and Neurological disorder must avoid one-size-fits-all claims.
Stem Cell Side Effects: The Part Patients Should Not Skip
The keyword stem cell side effects is not just an SEO phrase. It is one of the most important parts of patient education.
A 2024 systematic review of MSC-based therapies for neurological disorders reviewed 43 clinical manuscripts. It reported that many studies described the procedures as generally tolerable, with common mild events such as fever, headache, mild injection-site pain, or nausea. However, the review also emphasized that many studies had limitations such as small sample sizes and short follow-up periods, making long-term safety questions difficult to answer fully.
Commonly Reported Stem Cell Side Effects
Depending on the cell type, route of administration, patient condition, and procedure, possible stem cell side effects may include:
fever
headache
nausea
fatigue
injection-site discomfort
temporary pain
inflammatory reactions
infection risk
immune response
procedure-related complications
These should not be exaggerated, but they should not be hidden either.
Serious Safety Concerns
Regenerative medicine products have been marketed for the treatment of neurological disorders without FDA approval, but such safety concerns may include tumor formation and any neurologic event (e.g., stroke), as well as bacterial infections; inflammatory or immune reactions; unwanted cell growth in many forms (eg. Skin tissue); contamination and failure of therapy to provide expected benefits, according to warnings from U.S.
According to the FDA, stem cell products generally have to be approved and that the currently-approved (in the U.S.) stem includes blood-forming cells from umbilical cord blood for diseases of impaired production rather than generic neurological purposes.
Why Route of Administration Matters
When discussing stem cell side effects, the route of administration matters. Intravenous infusion, intrathecal injection, local injection, and neurosurgical transplantation carry different levels of complexity and risk.
Intravenous Delivery
Although IV delivery is less invasive than procedures that involve the nervous system, it continues to require sterility and screening for infectious diseases as well as monitoring by medical professionals during administration in order to address potential immune- or infusion-related reactions.
Intrathecal Delivery
Intrathecal delivery injects material into the CSF space. Due to its closeness to the nervous system, higher procedural caution is warranted making it more suitable for experienced clinicians with strict sterile technique and careful patient selection.
Direct Brain or Spinal Procedures
Cell transplantation into the brain or spinal cord is much more specialized and generally should be part of a regulated clinical trial, performed under appropriate research conditions in hospitals.
An ethical clinic should provide an explanation of how the route impacts these considerations, alongside potential stem cell side effects.
What a Responsible Stem Cell Program Should Include
A serious program for stem cell and Neurological disorder support should be structured around medical safety, not marketing excitement.
Diagnosis Review
The clinic should understand the exact neurological diagnosis, disease stage, imaging findings, medications, symptoms, and specialist reports.
Risk Screening
Patients should be screened for infection, immune disease, clotting risk, cancer history, uncontrolled illness, and other factors that may increase complications.
Cell Quality Transparency
The clinic should explain the cell source, donor screening, sterility testing, viability, identity testing, processing standards, and release criteria.
Realistic Goals
Goals may include comfort, function, inflammation balance, rehabilitation participation, or quality-of-life support. A clinic should not promise cure, reversal, or guaranteed neurological recovery.
Follow-Up and Outcome Tracking
Progress should be measured using neurological function, mobility, fatigue, cognition, speech, daily living ability, and quality-of-life tools when appropriate.
The Ethical Side of Stem Cell Neurology
Neurological patients are vulnerable since a lot of these conditions have limited treatment strategies. Which makes ethical communication very important! Stem Cell Research and Its Clinical Translation I – A Responsible Publishing in Cell Stem Cells, the International Society for Stem Cell Research suggests that stem cell research and clinical translation assume appropriate rigor, oversight, transparency, ethical integrity, and evidence-based development.
The ISSCR further cautions that promises of possible benefit are sometimes overstated and risks and clinical challenges understated, as well as the fact that companies promoting stem cells for unproven applications can take advantage of partial public comprehension.
Hope is important in any Neurological disorder. But hope should not be a product of exaggeration, but rather responsible medicine.
Conclusion: Stem Cell Neurology Needs Hope, Evidence, and Safety
With exemplary consideration for the sustainable relationship of stem cell science and Care For Neurological disorder, how to invest concrete time in one of regenerative medicine’s most thrilling areas. Immune modulation, neuroinflammation, cell signaling, and neuroprotection are important novel targets for researchers that extend beyond just replacing cells in selected conditions.
However, this field has to be addressed very cautiously. Potential side effects from stem cells are mostly viewed in the context of poorly regulated or processed products and those that are marketed outside established evidence. Before making any decision, patients should inquire about the diagnosis, cell types, route of administration (intra-arterial or otherwise), safety testing protocols, medical supervision/security available during treatment, and what realistic functional outcomes can be expected.
The meaningful future of neurological regenerative medicine that exists will not be constructed with shortcuts. It will be founded on science, transparency, judicious patient selection, and clinics that respect both the promise and the limits of stem cell care.