Insomnia is defined as a persistent difficulty with falling asleep, remaining asleep or achieving adequate quantities of restorative sleep despite having the opportunity to do so. Chronic insomnia can lead to daytime sleepiness, difficulty managing emotions, poorer cognitive performance and work productivity, decreased quality of life. Contemporary sleep medicine is moving away from viewing insomnia simply as a symptom of nighttime hyperarousal, to recognizing it as a disorder with chronic deregulation at the physiological, cognitive, emotional and even neurobiological levels.
In this context, biological and regenerative approaches that promote the internal environment implicated in chronic disturbance of sleep have gained considerable interest. Umnical cord-derived mesenchymal stromal/stem cells (UC-MSCs) are being investigated for their potential immunomodulatory, anti-inflammatory, paracrine and extracellular vesicle-related signaling properties in regenerative medicine. The efficacy of UC-MSCs stem cell therapy for insomnia However, then UC-MSCs stem cell therapy for insomnia should be described in academic caution. Currently, it needs to be categorized as an investigational and supportive concept rather than a paradigm treatment or cure for insomnia.
Regenerative medicine and clinical programs focused on wellness have gained the attention of healthcare professionals across the globe and patients with complex chronic conditions who consider integrative approaches, particularly in Thailand. A systematic clinical discussion centered on insomnia should be a blend of standard sleep medicine, behavioral therapy, lifestyle optimization, medical evaluation and judiciously articulated biological support.
Insomnia: A Clinical Overview
Insomnia can be multi-dimensional in terms of health. Patients may complain of trouble falling asleep, frequent awakening throughout the night, early morning awakening, have non-refreshing sleep, have symptoms of daytime tiredness, irritability and problems with concentration or memory that impact their social or occupational function. As time passes, chronic insomnia can initiate interactions with anxiety, depression, chronic pain, and metabolic dysfunctions and cardiovascular risk factors as well.
The disorder is typically perpetuated in a cycle: worrying about sleep leads to conditioned arousal, leading to irregular sleep scheduling and maladaptive behaviors, along with the activation of physiologic processes of stress. As a result, boosting sedative medication is rarely adequate for managing insomnia. It usually involves a systematic approach that targets both sleep behaviour and the arousal systems that prevent an individual from sleeping.
UC-MSCs for insomnia need to be framed as an adjunct therapy, not as substitute for guidelines in a pathway through much of the applicable evidence: UC-MSC stem cell therapy effectiveness Rather, it could be enabled as an experimental biological retention approach for consideration within a framework of neuroinflammation, stress-system regulation, immune signaling, and the global sleep-wake microenvironment.
Pathophysiology and Significant Disease Consequences
One of the main pathogenetic concepts in insomnia is hyperarousal. This refers to heightened activation of neural, autonomic, endocrine, and cognitive systems that prevent the brain and body from transitioning stable sleep. Hyperarousal may present with increased sympathetic activity, hyper-reactivity to stress, changes in cortical activation and dysregulated hypothalamic-pituitary-adrenal axis (HPA axis).
One important agent is the hypothalamic-pituitary-adrenal (HPA) axis, which mediates the body’s stress response. When this system is chronically overactive, the cortisol rhythm may be less adaptive for initiation and maintenance of sleep. This might explain difficulty falling asleep, interrupted sleep and shallower sleep.
Even chronic sleep disruption might affect immune and inflammatory signalling. Sleep loss is associated with changes in IL-6, TNF-α and other inflammatory mediators that impact daytime fatigue, alertness levels, and systemic stress responses.
Biologically, insomnia may therefore involve multiple interacting mechanisms:
Hyper-aroused CNS
HPA-axis dysregulation
Autonomic nervous system activation
Altered sleep-wake signaling
Stress-related inflammatory activity
Impaired restorative sleep architecture
Cognitive and emotional dysregulation
This does not imply that the vast majority of people with insomnia have structural “brain damage.” In academic terms, chronic insomnia may represent a disordered state of arousal, stress, circadian, emotional and inflammatory processes that are abnormally activated.
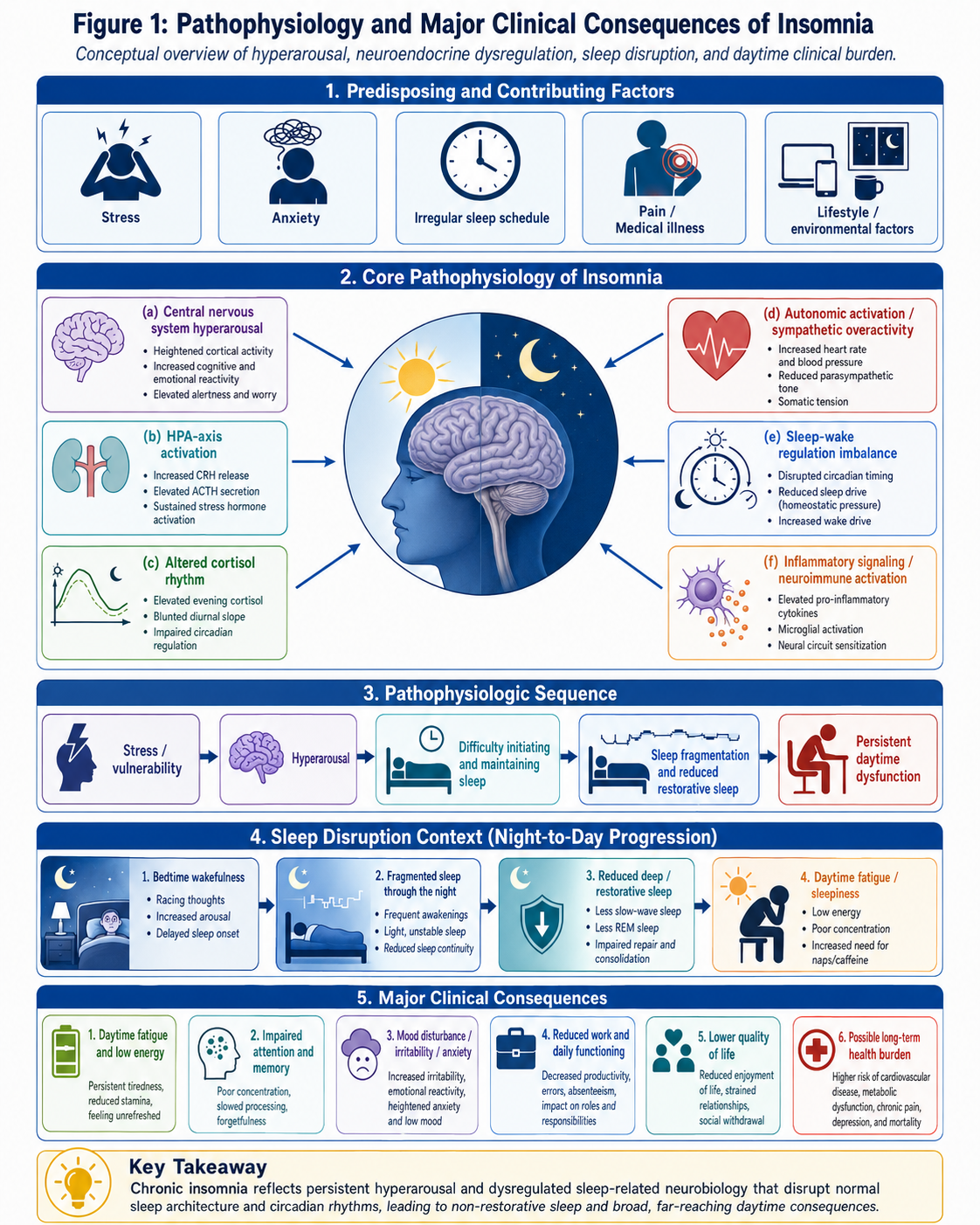
This schematic depicts important mechanisms involved in the perpetuation of chronic insomnia: central nervous system hyperarousal, activation of the HPA-axis and alteration of cortisol rhythm, autonomic activation (“upstream” mechanisms), sleep–wake disturbance, inflammatory signaling and daytime fatigue and impaired cognitive-emotional function (specifically how those symptoms are manifest)
Existing Conventional Therapies
Global or international concept of insomnia treatment involves non-pharmacological and pharmacological means in clinical practice. Cognitive Behavioral Therapy for Insomnia, or CBT-I, has an extensive evidence base supporting the efficacy as a first-line treatment; it targets both behavioral and cognitive mechanisms underpinning insomnia perpetuation. CBT-I may involve sleep restriction therapy, stimulation control, cognitive restructuring and/or relaxing tactics, sleep scheduling.
Consider pharmacological treatment in selected patients – particularly if insomnia is severe, requires short-term improvement or access to behavioral therapy is not immediately available. Available pharmacotherapy includes medications such as benzodiazepine receptor agonists, orexin receptor antagonists, melatonin receptor agonists, sedating antidepressants in appropriate situations and other clinician-directed options. Medication choice should weigh patient age, comorbidities, adverse-effect profile and duration-of-use risk-benefit balance according to clinical guidelines.
Long-term use of sedative-hypnotic medication has some limitations, including tolerance, dependence, risk for morning sedation, cognitive impairment, falls and other side effects in sensitive populations. Due to risks of sedation, dependence, falls and fractures, as well as cognitive effects that are specifically associated with benzodiazepines, appropriate prescribing and monitoring is especially important.
The conventional treatment does not lack effectiveness, but its limitation is that it has been limited. CBT-I and guideline-based pharmacotherapy can also be of considerable value. However, limitations are that some patients with insomnia not only persist but also major long-term medications may not well tolerated. This has sparked interest in adjunctive biological approaches such as an investigational use of UC-MSCs stem cell therapy for insomnia assistance.
UC-MSCs as an Experimental Completion Support Concept
UC-MSCs become the object of study as they may produce bioactive molecules, cytokines, growth factors, and EVs that can influence both immune cells and tissue microenvironments. UC-MSCs are not often viewed as a new source of replacement cells in many discussions on regenerative medicine. They are more often associated with paracrine signaling, immunomodulation, anti-inflammatory communication and effects mediated by extracellular vesicles.
This notion is still premature and under investigation for insomnia. That biological rationale is itself predicated on the idea that neuroimmune signaling may be a powerful therapist of chronic sleep disturbance, though also points towards dysregulation in the stress system and inflammatory imbalance. In terms of clinical achievements, even though some preclinical studies have been conducted with human UC-MSC-derived extracellular vesicles (exosomes) and changes in behavioral traits associated with sleep deprivation or inflammation during the sleep/wake cycle have shown promising efficacy, this is not a designated treatment for insomnia.
A medically responsible formulation would be:
The biological support for UC-MSCs will be explored in specific insomnia-related programs, particularly as they affect immune modulation, inflammatory balance and neurobiological support. Nevertheless, insomnia clinical evidence is still scarce, and outcomes are never assured.
However, UC-MSCs have recently emerged as a new biological treatment alternative for insomnia and may also participate in the sleep-wake mechanism.
5.1 Neuroinflammatory Balance
The alteration in inflammatory signaling that may result from chronic sleep disruption. This is a strictly bioinformatics based paper derived primarily from databases operating under high throughput functional genomics to characterize UC-MSCs already shown to exert the capacity both in regulating immune cell activation, as well as verifying that passage it definitely alters inflammation mediator secretion. In a context of insomnia support, instead of the proposed goal to “repair the brain,” aim to balance out the neuroimmune environment.
A cautious academic wording is:
Paracrine and immunomodulatory signals may contribute to the potential role of UC-MSCs in supporting inflammatory balance, although few studies have tested this concept in insomnia.
5.2 HPA-Axis and Stress-System Support
Insomnia is frequently linked to increased stress reactivity and HPA-axis activation. UC-MSCs was not established as a direct key modulator of cortisol rhythm or sleep architecture. Nonetheless, as MSCs are investigated for immunomodulatory and anti-inflammatory properties, it is possible that scholars will entertain the notion of whether systemic biological homeostasis could impact stress-related pathways indirectly.
A responsible statement is:
The association between UC-MSCs and HPA-axis regulation may be proposed as a potential concept but has to be confirmed in controlled clinical studies on insomnia.
5.3 Exosome and Paracrine Signaling
UC-MSC-derived extracellular vesicles are rich in proteins, lipids, RNAs and microRNA-related signals with a capacity of cell-to-cell communication. These vesicles are currently being studied in the context of regenerative medicine research as a method of impacting inflammatory and neurological microenvironments. In MSC studies, there is increasing interest in application to neurological disease models but translation into regular clinical practice still needs input.
For insomnia, the research need should be treated as an open question rather than a useable treatment pathway.
5.4 Providing an Ambient in Sleep-Wake Microenvironment
The sleep-wake system consists of interplay between the circadian rhythm and stress biology, neurotransmitter balance, immune signaling and behavioral conditioning. In the larger context of this framework, UC-MSCs may be considered in relation to biological adjuncts or aids. Rather, there should be no mention of glowing chronobiotically or fixing neurotransmitter pathways unless supported by clinical data.

