Diabetes Mellitus Type 2 (DMT2) is one of the common chronic metabolic diseases in worldwide which represents a clinical and health care challenge for contemporary health care systems. Diabetes mellitus is characterised by a state of chronic hyperglycaemia due to increased insulin resistance and/or impaired insulin secretion and ultimately progressive β-cell failure. Chronic patients who did not receive treatment may ultimately associated with systemic complications from ineffective glucose regulation including; chronic kidney disease (disease-specific for diabetic nephropathy), nerve damage (peripheral neuropathy),eye diseases(retinopathy), cardiovascular problems because of inadequate wound healing and decreased qualify-of-life.
In addition, lifestyle modification, oral hypoglycaemic agents, injectables and insulin with structured monitoring have all greatly improved the management of diabetes using conventional treatment. Nevertheless, and in spite of all these advances, many patients continue to suffer from progressive metabolic decline, requiring increasing doses of diabetes medicines that become intolerable or render long-term glycaemic control difficult. These limitations have encouraged a lot of research into regenerative medicine, and more specifically into the potential use of mesenchymal stem/stromal cells as an additional treatment for metabolic disease.
Within the new area of stem cell therapy Thailand, mesenchymal stem/stromal cells (MSCs) (umbilical cord-derived MSCs [UC-MSCs]) are studied due to their capacity to adjust immune reaction, inhibit swelling and arouse other tissues using a paracrine signaling system. They should be seen as a new and experimental treatment, not a replacement for traditional diabetes care. The primary academic issue is that, if UC-MSCs could potentially help to preserve β-cells from apoptosis, reduce chronic inflammation and improve insulin sensitivity with other measures for a more favorable metabolic microenvironment.
Diabetes Mellitus Type 2, The most prevalent form of diabetes, this condition is characterized by the development of insulin resistance due to repeated exposure to high concentrations of circulating glucose.
Pathophysiology of diabetes mellitus type 2 is characterized by a disorder in the pathophysiology of glucose and energy metabolism system. First, tissues such as skeletal muscle, adipose tissue and liver increase their resistance to the action of insulin. It is associated with insulin resistance which causes a reduced uptake of glucose and an increase in hepatic glucose production, resulting in hyperglycaemia.
In turn, pancreatic β-cells release more insulin. Conversely, an ongoing metabolic stress on the β-cell due to a combination of glucotoxicity, lipotoxicity, oxidative injury and inflammatory signals more gradually infiltrate cellular function. With time progression of the disease, β-cells may lose their ability to properly secrete insulin resulting in worsening hyperglycemia.
Even prolonged instances of hyperglycemia result in vascular injury. High glucose in fact increases oxidative stress, advanced glycation end products (AGEs), endothelial dysfunction and finally chronic low grade inflammation. The central element of this process is the involvement of mechanisms in microvascular and macrovascular diabetic complications, such as kidney disease, nerve damage and retinal injury (microvascular), coronary artery disease (CAD) and stroke (macrovascular).
Hence, Diabetes Mellitus T2 cannot be taken as a blood sugar level elevation disorder.
Clinical Caveats of Conventional Therapies
Diabetes Mellitus Type 2 is typically managed individually with glycemic control, reduction of cardiovascular risk factors, lifestyle modification and prevention of chronic complications. Main treatments include diet and lifestyle changes, weight loss or medications (metformin, insulin-sensitizing agents, insulin secretagogues [sulfonylureas], GLP-1 receptor agonists [GLP-1RA], SGLT2 inhibitors; insulins when clinically indicated).
Such treatments remain recognized as essential and evidence-based. Many patients do demonstrably improve in blood glucose control, as measured by HbA1c or other more complex metrics, cardiovascular risk markers and other aspects of disease management. But conventional methods also come with their own limitations. Adverse effects (drug class and patient profile depending) – some agents might cause hypoglycemia, GI upset, weight gain or dehydration risk.
Another important limitation is that most conventional diabetes treatments focus on ameliorating hyperglycemia rather than directly regenerating insulin-producing β-cell tissue or replacing diabetes-compromised loss of tissue. Diabetes Mellitus Type 2 is still frequently a progressive disorder, at least in patients with advanced β -cell dysfunction by modern medications that can both lower complication risk and disease burden, yet particularly those with longstanding disease or severe insulin resistance (obesity) or chronic inflammation.
As a result, it eventually sparked interest in regenerative medicine. Rather than replacing available diabetes therapy, the potential for regenerative approaches to add to existing care while targeting simultaneous paths of inflammation, immune–metabolic imbalance, vascular dysfunctions and tissue-repair signaling have been an active area of investigation.
MSC: The application of UC-MSC in regenerative medical management
Based on deep and immunogenicity, umbilical cord-derived mesenchymal stem/stromal cells (UC-MSCs) have attracted immense attention in regenerative medicine due to their unique biological feature. As non-replacement cells, UC-MSCs represent a poor paradigm. Instead, most of the effects attributed to these cells are thought to be mediated by paracrine action; secretion of cytokines, growth factors, extracellular vesicles and other bioactive molecules that act on proximal tissues/cells.
The diverse way of MSCs which are presently being study for UC-MSC in DM2. The possible mechanisms underlying such effects are modulation of chronic inflammatory processes, reduction of oxidative stress, improvement of local tissue microcirculation as well as partial preservation or even loss inhibition of pancreatic β-cell survival and regulation to immune-metabolic signaling. The role of UC-MSCs in influencing immune system might potentially be linked with metabolic homeostasis as the condition is strongly associated with chronic inflammation, which mediates insulin resistance.
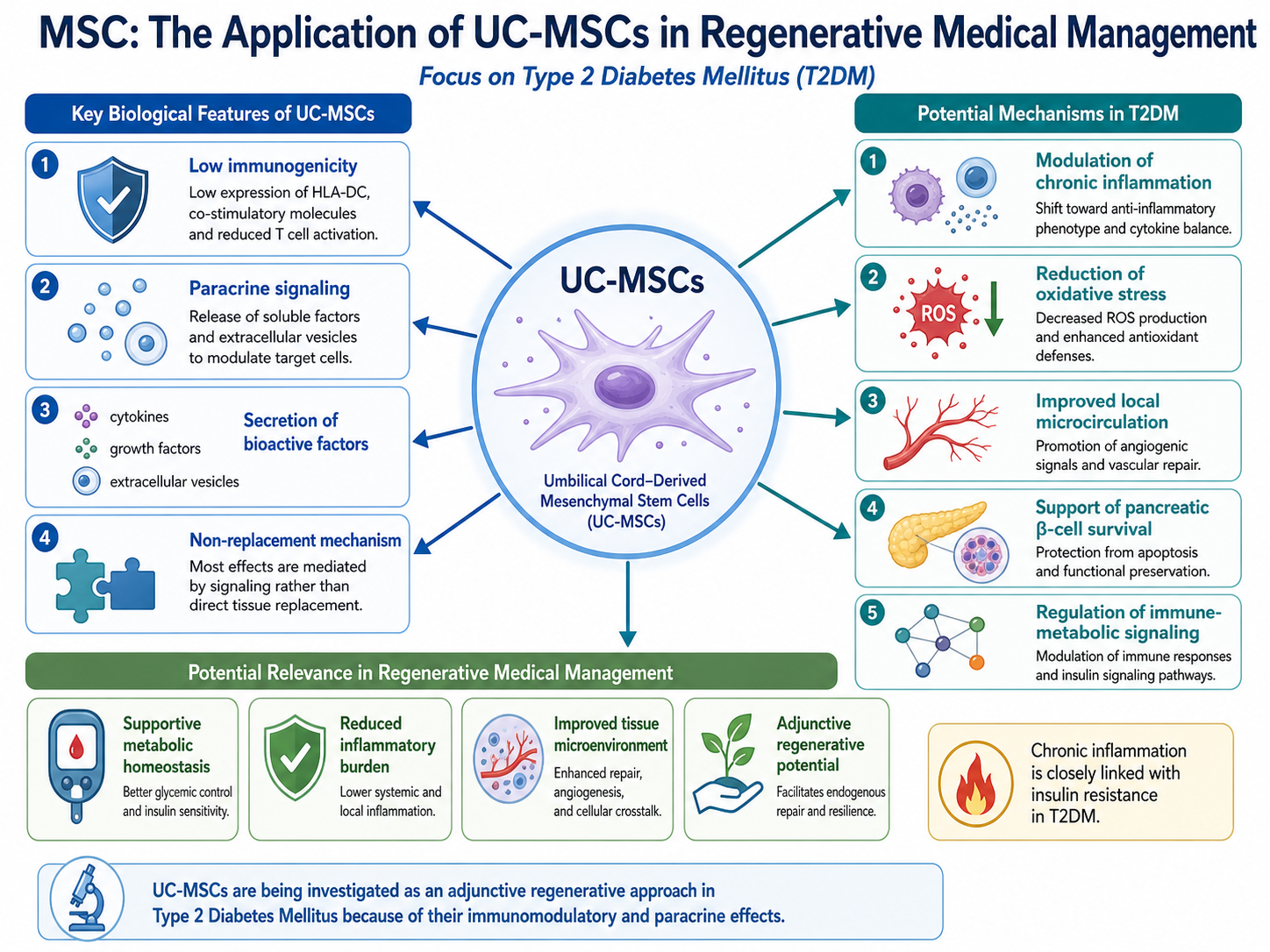
Secreted factors involved in cell communication and the tissue repair process may also allow UC-MSCs to influence the pancreatic and hepatic environments. This has been investigated in multiple case–control studies, some animal ingestion methods or insulin secretion-based measurements as β-cell function markers. However, evidence is still emerging and outcomes will likely be influenced by patient selection, disease duration, dosing of cells, route of administration, cell quality and follow-up time.
For this reason, it is essential to present UC-MSCs as either a research or an adjunctive regenerative public method. A conclusive statement that UC-MSCs can cure you of Type 2 Diabetes Mellitus and regenerate your entire pancreas or remove all needs for medication will be scientific falsehood. The more rational academic position would be that UC-MSCs at least could contribute to a healthier metabolic and inflammatory milieu, in select patients; and as such further exploration is needed to help define their role in therapy.
Traditional Stem Cell Therapy and Regenerative Stem Cell Therapy For Type 2 Diabetes Mellitus
Thailand as the Hub of Regenerative Medicine: Opportunities
The developments in the regenerative medication, medical wellness and cell-based clinical services have been warmer and hotter to make Thailand one stage better into center point perspective. Development of stem cell therapy thailand falls within a larger rajghat region-based demand for biotechnology and advanced laboratory systems with medically supervised regenerative protocols.
Applications of UC-MSC to emerging areas of diabetes mellitus type 2 require better clinical evidence, standardized manufacturing processes, extensive quality control and sterility testing, cell characterization and potency assay in the phase I & II setting and defined follow-up protocols. These elements are essential to enhance the scientific integrity and clinical safety of cellular therapies.
The future of MSC-based diabetes care could be more precise. Future programs would be more sophisticated, including duration of disease, baseline HbA1c and fasting glucose & insulin resistance as well as biomarker (C-peptide level) along with body composition data (visceral fat area) combined with inflammatory markers (hsCRP and a few others), cardiovascular risk factors (BP and lipid profile), kidney function tests by urine albumin > creatinine ratio for excretion of small-type proteins in non-RAAS stimulating drug subjects etc. medications history also from family background to lifestyle related aspects incorporated. Other than through protocol consistency across the continuum of patients. Evaluating this may help clinicians know which patients would do better on regenerative support, as well as what end points should be followed over time.
The evolution of Thailand as a providores for the provision of medical care is continuing more broadly with appropriate levels of patient education and required clinical pratice assurance to support regulated regeneration medicine assisted by evidence based physician manged lifecycle reviews as markers of identifying good practice. In this scenario, a Thailand clinic specializing in stem cells should be transparent about the potential value and limitations of UC-MSC based approaches for disease management.
Conclusion
Diabetes Mellitus Type 2 is a heterogeneous metabolic disorder, which involves the dysfunction of multiple tissues (insulin resistance, β-cell dysfunction, chronic inflammation and oxidative stress and vascular disorders. Although conventional therapeutic agents have been and still are the mainstay of evidence-based diabetes care, numerous patients suffer from progressive disease, medication burden, or residual metabolic inadequacy.
UC-MSCs represent a rapidly developing area in regenerative medicine with rapid immunomodulatory, anti-inflammatory and paracrine signaling effects as well as the option to provide patient-independent. In DM2, they are scientifically relevant by virtue of their potential multi-faceted actions in β-cell protection and regeneration, the amelioration of the inflammatory microenvironment and tissue repair signaling/metabolism coupling to many aspects of multi-organ metabolic management.
However, descriptors should guard against excessive clinical reliance on UC-MSC based care. It should not be portrayed as a cure or alternative to conventional diabetes management. Or it might be an official free service requiring careful patient selection, well controlled cell processing, physician supervision and reproducible blinded follow-up.
As research advances the advancement of stem cell treatment Thailand may pave the way for a more customized strategy to metabolic management. These findings, however, are at least incomplete for the moment and the wisest path does not lie in promises of reversing disease but preferably in coming together to ground regenerative medicine as part of a patient-centered scientific approach to diabetes care.

