Why does this topic suddenly feel more real
When people search for stem cell clinics and stem cell diabetes, they are usually not looking for general science. They are looking for one serious answer: has stem-cell-based treatment moved from theory into something that actually changes life with diabetes? That question has become more understandable in the past few years, because the strongest recent data of success stem cell treatment have come from type 1 diabetes studies showing restoration of endogenous insulin production in selected patients. At the same time, that progress is still much narrower and more carefully controlled than many clinic pages imply.
What stem cell diabetes actually means
The clearest current target is type 1 diabetes.
The best dream of stem cell diabetes today is basically a type 1 story. Much of the regenerative framework in type 1 diabetes revolves around beta-cell loss, and so its central problem is not merely glucose-lowering — it is restoring beta-cell function or replacing insufficient tissue with sufficient tissue such that proper insulin-making machinery comes back. Most of the biggest recent advances are all about islet replacement, hence your naive vagueness on your part.
There is already one FDA-approved cellular therapy, but it is limited.
The most critical reality check is that Lantidra, the first allogeneic pancreatic islet cellular therapy, has already been approved by the FDA under limited indications for adults with type 1 diabetes who continue to experience recurrent severe hypoglycemia despite intensive management and education about their condition. Lantidra is a specific therapy for a particular, small group of people, specifically since it uses the islet cells from deceased donor pancreases, so it is not even applicable to every person with diabetes, rather than an ‘anyone walk-in stem cell clinic’ service.
The newer excitement is about stem-cell-derived islets.
Where the field becomes even more interesting is in stem-cell-derived islet therapy. A 2024 Nature review described recent clinically meaningful metabolic benefits from transplanted stem-cell-derived tissue, and the 2025 New England Journal of Medicine study on zimislecel pushed that conversation further by reporting physiologic islet function after transplantation of allogeneic stem-cell-derived islet cells in people with type 1 diabetes.
What the data of successful stem cell treatment actually show
The best current numbers are encouraging, but they belong to selected patients
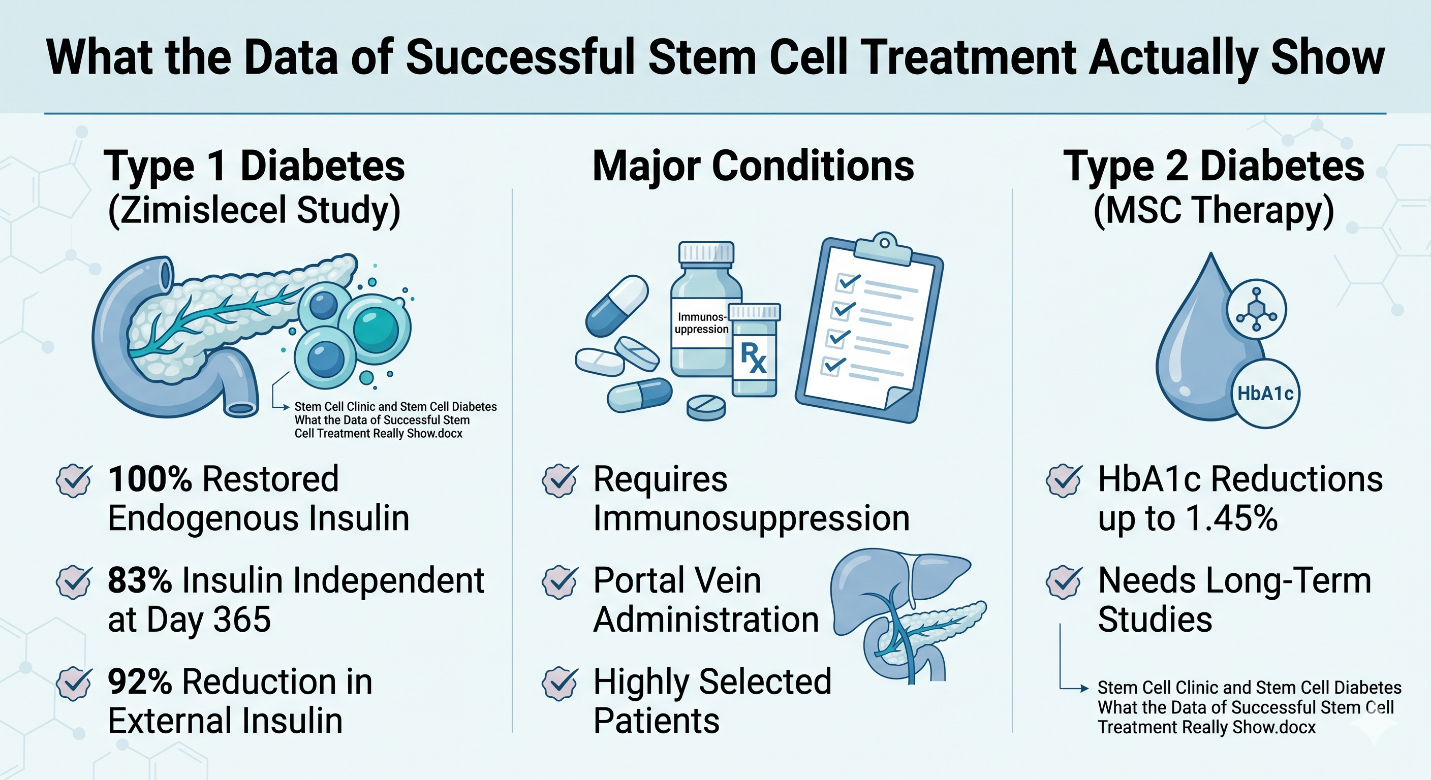
If someone asks for the strongest current data on successful stem cell treatment, the most cited recent example is the 2025 Zimislecel phase 1–2 study. In the full-dose cohort followed for at least a year, all 12 participants showed restoration of endogenous insulin secretion, elimination of severe hypoglycemic events, and achievement of glycemic targets; 10 of the 12 participants, or 83%, were insulin independent at day 365, and external insulin use fell by a mean of 92%. Those are remarkable data. But they come from a small, highly selected group of people with type 1 diabetes and impaired awareness of hypoglycemia, not from the broad diabetes population.
These successes still come with major conditions.
This is the part that matters just as much as the headline numbers. Zimislecel was given through the portal vein along with a transplant-style immunosuppression regimen, and the study itself was still early-phase and short-term. That means the strongest current “success” data do not describe a simple outpatient wellness procedure. They are describing sophisticated transplant medicine with meaningful follow-up, careful patient selection, and ongoing questions about long-term durability and immune protection.
Type 2 diabetes is a different and less settled story.
If the conversation expands beyond type 1 diabetes, the evidence becomes much less definitive. A 2025 umbrella review on mesenchymal stem cell therapy found improvements in glycemic control, particularly in type 2 diabetes, with HbA1c reductions of up to 1.45% and reduced insulin requirements, but it also emphasized the need for standardized protocols and long-term studies. In other words, there is a signal, but not yet the kind of success data that should be mistaken for a general cure story.
Why is this still not the same as any stem cell clinic claim
A clinic offering treatment is not the same thing as a therapy being established
This is where the phrase stem cell clinic can become misleading. The FDA continues to warn patients and consumers to avoid unapproved products from human cells or tissues that are marketed online for the treatment or cure of many medical conditions, noting that the agency may not have reviewed their quality, safety, purity, or potency. That warning matters a lot in diabetes, because real progress in regulated research can make unproven commercial claims sound more believable than they deserve to sound.
The best clinic is not the one with the biggest promise.
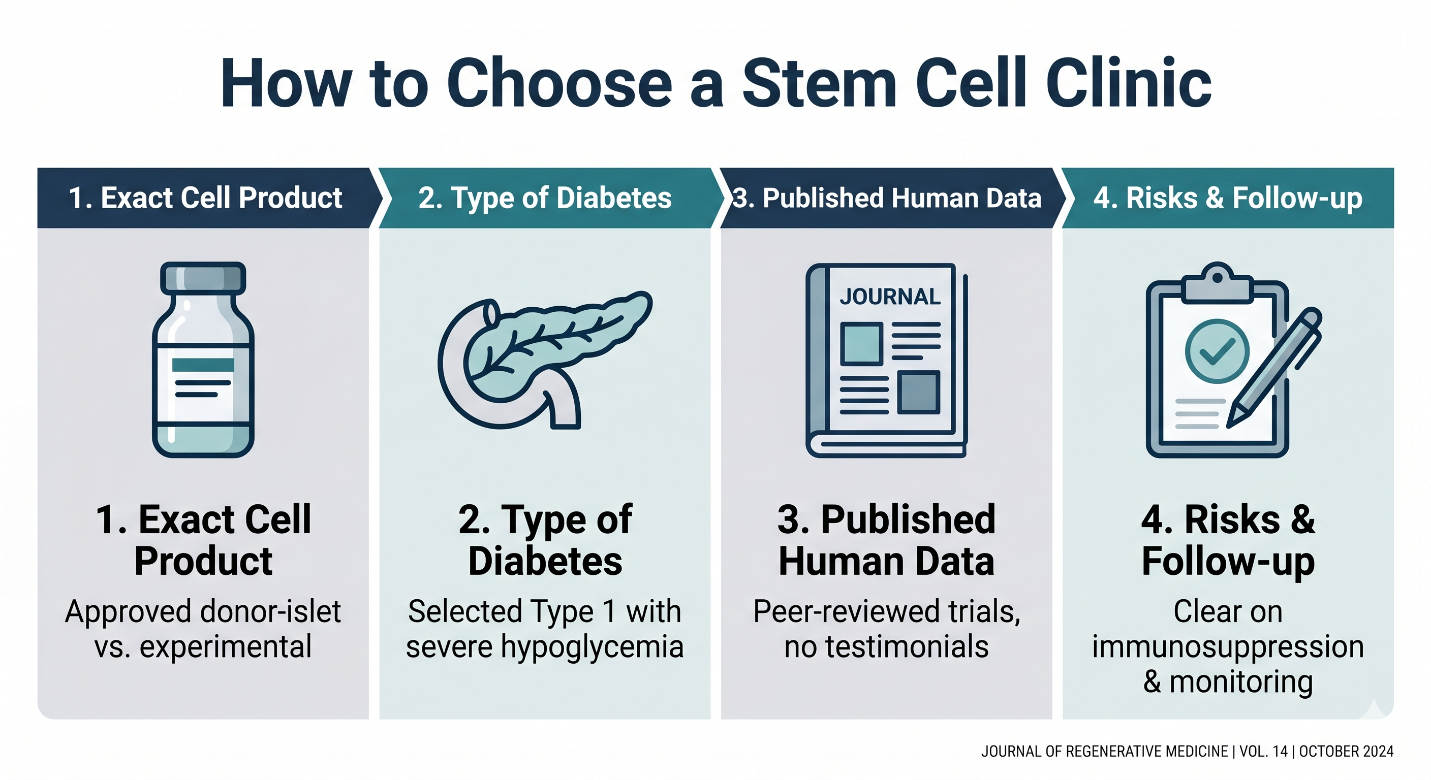
Usually, one of the most credible clinic conversations does not sound better but sharper, not more dramatic. ISSCR’s patient resources emphasize these themes in terms of rigor, oversight, and transparency, as well as whether a treatment is approved or only experimental. A credible diabetes clinic, in practical terms, should be able to define: the precise cell product (nomenclature, preparation method, dose); exact type of diabetes being treated; route and schedule of administration; evidence for requirement (or not) for immunosuppression per protocol, if relevant, as well as unpublished data where applicable.
A short, practical way to choose a clinic
A few questions matter more than glossy branding
If you want a quick way to judge a stem cell clinic, focus on a few basic questions:
What exact cell product is being used, and is it approved or only experimental? A real answer should separate approved donor-islet therapy from investigational stem-cell-derived products.
What kind of diabetes is being treated? The strongest current evidence is in selected type 1 diabetes patients with severe hypoglycemia risk, not in every person with diabetes.
What published human data support the treatment? A serious clinic should be able to point to peer-reviewed trials, not just testimonials.
What are the risks, follow-up plan, and immune requirements? If the clinic glosses over immunosuppression, monitoring, or uncertainty, that is a bad sign.
The most honest conclusion
Here is the neatest way of putting it: stem cell diabetes has ceased to be a pipedream. Current clinical data from selected type 1 diabetes programs indicate that stem-cell-derived islet therapy can restore endogenous insulin secretion and, in some patients, eliminate the need for external to substantial periods of time. However, those results are from highly controlled clinical contexts and not all clinics use regenerative language. Therefore, the most truthful answer to what is a good stem cell clinic, when somebody asks, is not the brand name. So it is a standard: the right clinic is the only one that can honestly say just how good or even moderately strong, and then exactly where each still stops.