- Introduction to Kidney Function Decline
Healthy kidneys do much more than produce urine. They play a role in fluid balance, electrolytes, acid-base status, blood pressure and waste management. If kidney structure or function is abnormal for at least three months, it is chronic kidney disease (CKD). CKD is usually measured through estimated glomerular filtration rate (eGFR) and albuminuria, and it can progress from mild impairment to kidney failure if the underlying injury persists.
Conventional kidney care remains essential. Standard management consists of blood pressure control, blockade of the renin-angiotensin system, if appropriate with SGLT2 inhibitors, risk-factor control and treatment of the underlying cause. Nonetheless, many patients gradually lose their kidney function over time, prompting increasing research interest in regenerative and biologically targeted strategies. These include umbilical cord-derived mesenchymal stem cells (UC-MSCs) and selected peptide-based therapies that have been investigated for their potential anti-inflammatory, anti-fibrotic, or tissue-supportive effects.
- Why Kidney Function Worsens Over Time
The pattern of injury is similar in many kidney diseases. The continual assault from inflammation, oxidative stress, endothelial dysfunction, microvascular damage, tubular injury and fibrosis progressively impairs the kidney’s ability to filter blood efficiently. As fibrosis increases, kidney tissue stiffens and functions poorly, making late-stage damage hard to undo.
That’s one reason that kidney care increasingly aims not just to manage symptoms but also to slow the biological processes causing progression. In CKD, albuminuria and declining eGFR are more than laboratory abnormalities; they are markers that the filtration barrier and tissue environment of the kidney are under strain. Any supportive therapy under consideration should therefore be evaluated in relation to its potential for reduction of inflammation, protection of surviving cells, and preserving function as an adjunctive approach to standard nephrology care.
- Limits of Conventional Treatment Alone
Current medical therapy can greatly enhance outcomes, but it still does not resolve the entire issue completely. Many such treatments have been developed to minimize pressure on the kidneys, control diabetes or hypertension, and manage complications like anemia, acidosis or fluid overload. These are essential and evidence-based, but will not directly restore damaged kidney tissue.
That has sparked interest in therapies that may more directly affect the kidney microenvironment. That’s not to say that regenerative medicine supplants standard care. Instead, the question for research is whether biological therapies may serve an adjunct in certain patients by limiting inflammatory signaling, inhibiting fibrosis and aiding in preservation of viable renal cells. Currently, this is an area of active investigation and clinical evidence is still lacking compared with preclinical literature.
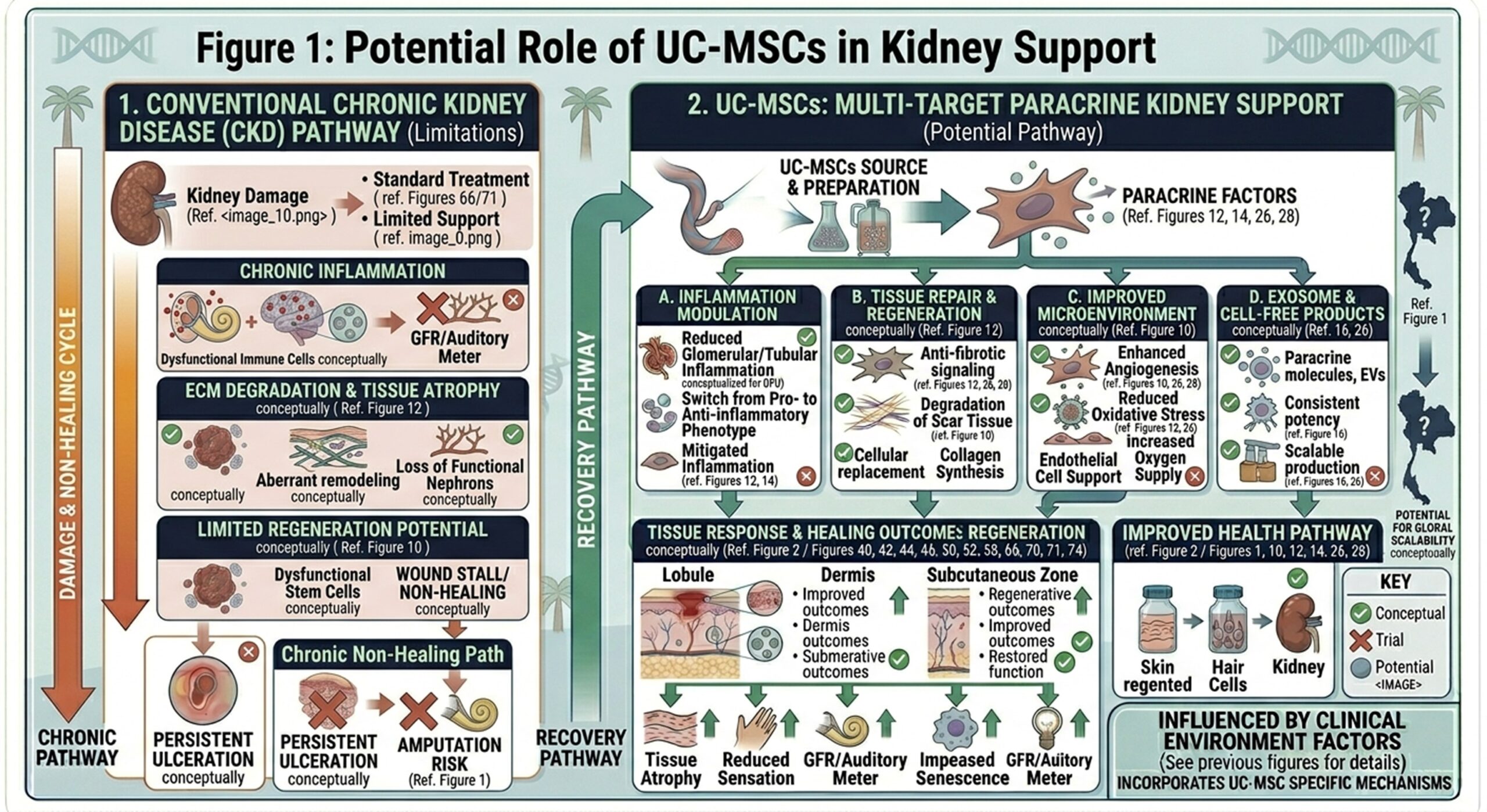
- Potential Role of UC-MSCs in Kidney Support
UC-MSCs are being investigated because mesenchymal stem cells have the capacity to secrete bioactive factors affecting immunity, inflammation, angiogenesis and tissue repair signaling. In several kidney disease models, MSCs have demonstrated reduced inflammatory cytokine levels, reduced oxidative stress activity, decreased collagen deposition and fibrosis, and improvement in serum markers of renal injury. Importantly, many researchers believe their primary beneficial action is paracrine support rather than direct replacement of kidney cells.
Interestingly, umbilical cord-derived cells are particularly appealing in that they are readily accessible, possess lower immunogenicity compared with some other cell sources, and have been extensively investigated in regenerative research. Studies of CKD and renal injury indicate that MSC-based approaches may help modulate the immune environment and support residual kidney tissue. But reviews also stress that hopeful data in animals has yet to yield conclusive clinical evidence for widespread routine use in patients.
Clinically relevant putative benefits being studied include supportive properties on eGFR stability, reduction of inflammatory injury, improvement in albumin leakage, and attenuation of fibrotic progression. While these options may be scientifically intriguing, they must still be conveyed with care: UC-MSCs do not constitute an approved treatment for CKD and the source of cells, manufacturing quality, dosing, and route of administration all matter.
- Potential Role of Peptide-Based Strategies
“Peptides” is a broad category, so it’s crucial to not use a one-size-fits-all approach when considering and using peptide therapies. Some peptide-based drugs already play meaningful roles in kidney-related care while others are still experimental. For instance, GLP-1 receptor agonists (a group of peptide-based medication mainly indicated for type 2 diabetes and obesity) have demonstrated cardiovascular and kidney protection in individuals who have CKD, particularly when these patients have diabetes. Recent trials and meta-analyses indicate that these agents may reduce clinically-relevant kidney end points in appropriate patients.
Other peptide approaches are still further behind in development. Peptide therapy in kidney disease reviews describe proposed renoprotective mechanisms that may include the reduction of tubular apoptosis, podocyte protection, blunting of inflammatory signaling, and reduction in fibrosis. C-peptide has also been investigated as a potential renoprotective agent, especially in diabetic kidney disease settings; however, the available evidence base remains limited and not yet fully adequate to draw wide clinical conclusions.
That creates a useful clinical distinction: some peptide-based therapies have already found their way into the mainstream treatment of patients to reduce kidney risk, while others remain investigational and should be treated as such. When evaluating how to use peptides for the sake of kidney function, I submit that the critical question is not whether “peptides” are good for you or not as a blanket statement but rather which peptide is being referred to, in what patient population and disease state, and with which level of evidence of efficacy.Profile.

