Why diabetic wounds are so difficult to heal
Diabetic wounds, particularly diabetic foot ulcers, are notoriously difficult to heal because multiple pathologies can simultaneously present: poor blood flow, neuropathy, infection risk, chronic inflammation, recurrent pressure and impaired tissue repair. The International Working Group on the Diabetic Foot states that any method of advanced wound-healing intervention should only be employed in conjunction with gold-standard multidisciplinary care, never as a replacement. Within that standard of care framework, the basics include local debridement, pressure offloading, revascularization when indicated and treatment of infection as appropriate.
Why UC-MSCs are being studied for diabetic wound healing
UC-MSCs (Umbilical Cord Mesenchymal Stromal cells) are under investigation because they might positively modulate local inflammation, vascularization and the local wound ambience through paracrine signalling. Recent reviews characterize MSC therapy for the treatment of chronic wounds as highly promising based on early-phase studies but also make clear that this area of cell-based therapy remains investigational and that larger, more rigorous trials are needed before treatment standards can be established. The FDA also continues to caution patients against unapproved human cell or tissue products being sold for a range of diseases as the agency may not have evaluated their quality, safety, purity or potency.
Potential benefits of UC-MSCs for diabetic wounds
Benefits should be framed as potential or reported, but not assured results, when presented. Due to their immunomodulatory activity, UC-MSC-based approaches have been linked to faster wound closure, improved granulation and epithelialization, as well as a healthier healing environment in a few selected patients involved in clinical and translational studies. For example, a 2022 phase I pilot study in patients with diabetic foot ulcer plus peripheral arterial disease reported positive long-term outcomes after treatment with UC-MSC combined therapy (Lin et al., 2022), while promising healing results using perilesional injection has been described for a phase I/II clinical trial with derivatives of native MSCs from umbilical cord tissue (UC-MSC; Dziubinski et al., 2015). The results are encouraging, but they do not yet create a universal standard of care.
What dose has actually been studied?
Currently, there are no standard UC-MSC doses for the treatment of diabetic wound. So, the most helpful way to discuss dose is to discuss published investigational protocols.
For the human UC-MSC pilot study in diabetic foot ulcer with peripheral arterial disease used 1 million MSCs/kg per doses, and at least an upper limit of up to 2 million MSCs/kg per doses. All patients were well above 50 kg, practical dose was 50 to 100 million cells per treatment most received a total of 3 treatments for total combined of 30-50 million cells.
In the same study, local dose delivery was performed with injective local around wound edge and wound base using small aliqout of 2 mL UC-MSC suspension (e.g., 0.5 to 1.0 mL directly injected per injection for every 1.5 cm² of injured area). That detail is helpful because it indicates the protocol wasn’t just “10 million cells” but rather a plan for structured wound-based local delivery.
A phase I/II study (2025) used a human UC-MSC derivative by perilesional injection instead of whole cells. For that protocol, patients received 5 mL weekly for up to 10 sessions. Mentioning it but only if you’re flagging it as derivatives protocol versus whole-cell UC-MSC dose.
Should you suggest local injection, IV infusion, or both?
The preclinical work performed overall suggests that local or perilesional delivery is the most compelling wound route to focus on based upon the human diabetic wound literature. In the 2022 UC-MSC pilot study, cells were injected subcutaneously into tissue adjacent to the lesion and into the wound base on Day 0. The authors ultimately found that healing “relied primarily on topical injection,” although the study employed IV treatment as well. This makes local delivery the main route to highlight for an article on diabetic wound, with most evidence aligned.
Avoid generalizing and, if you have to mention IV, be specific. The same study did not specify a standard intravenous systemic drip as the predominant method. Instead, following local treatment on Day 0, patients received regional intravenous infusion into veins of the affected foot on Day 7 and then another IV infusion on Day 28. Because their patients had nonhealing diabetic foot ulcers and significant peripheral arterial disease not suitable for revascularization, the authors employed that combined strategy.
That said, the most defensible phrasing is as follows: local or perilesional injection can be described as the dominant wound-directed strategy; regional IV infusion can be mentioned as a selective adjunct for patients with ischemic limbs (eg, leg ulcer [DFU] with PAD), to specialist care. This is a significantly stronger stance than offering IV alone as an adjunct to a general diabetic wound protocol.
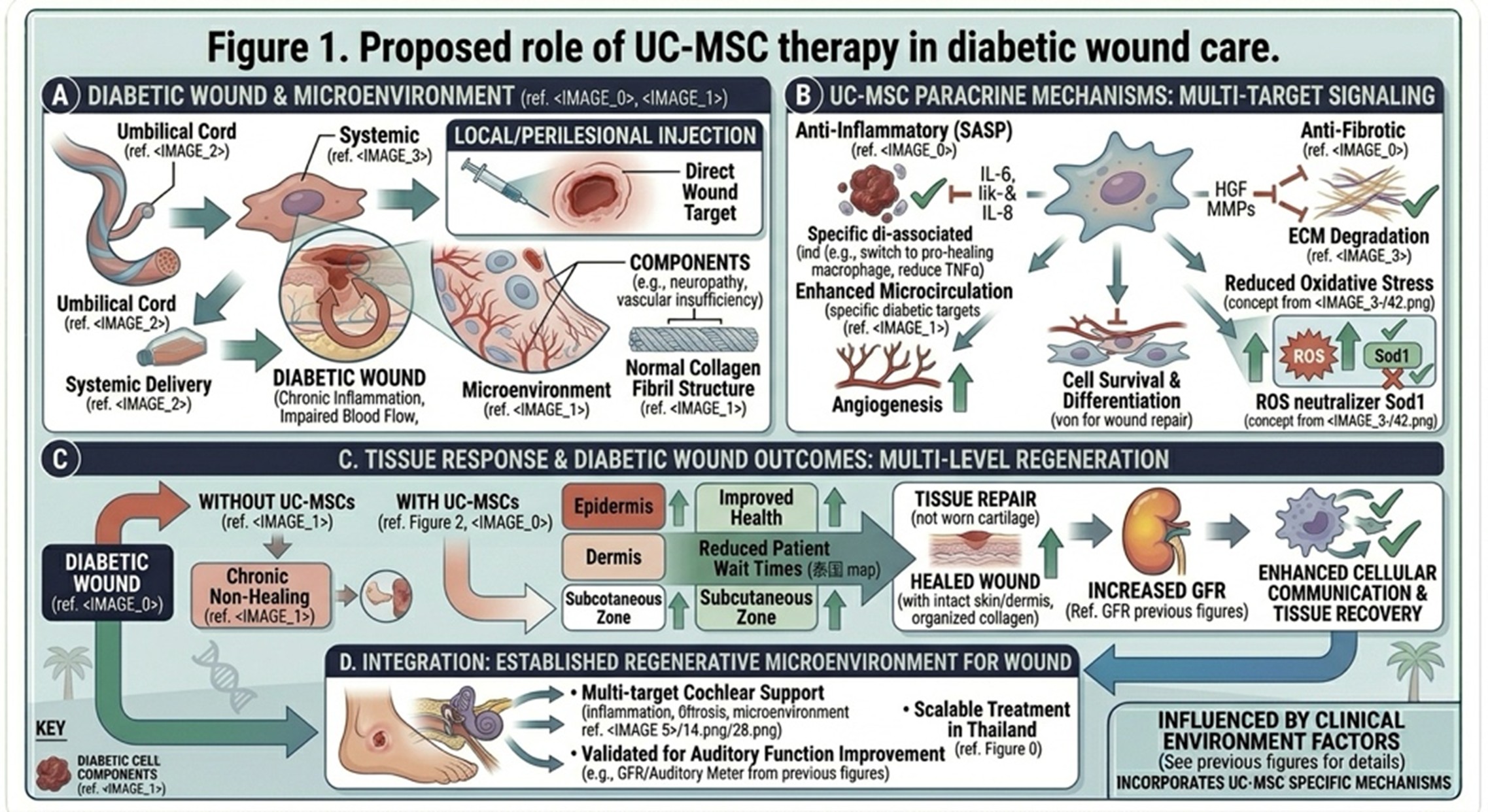
Diabetic wounds often heal poorly because of chronic inflammation, impaired blood flow, neuropathy, infection risk, and reduced tissue repair capacity. Current wound-healing guidance says advanced therapies should be used alongside gold-standard care such as debridement, offloading, infection control, and revascularization assessment when needed. In published UC-MSC diabetic foot studies, local or perilesional injection has been the most directly wound-targeted route, with some protocols adding regional intravenous infusion in selected patients with peripheral arterial disease.

