Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder, which shows pancreatic β-cell failure alongside insulin resistance. Pharmacological therapies are vital to controlling the disease, but mainly focus on regulating glycemia rather than addressing the upstream pathways driving disease progression. Given the high demand for better alternatives, regenerative medicine approaches using MSCs Stem Cells and UC-MSC have attracted a lot of interest. Such cells have been investigated for immuno-modulatory properties as well as systemic anti-inflammatory effects and paracrine support of pancreatic function. This review article addresses the pathophysiology of T2DM, and discusses the limits of conventional treatments with new emerging roles in MSC-based therapies for clinical applications in Thailand.
- Introduction
Type 2 Diabetes Mellitus is a significant global public health problem, fueled by lifestyle changes, an aging population, and increasing prevalence of metabolic disorders. Diabetes is characterized by chronic hyperglycemia due to impaired action of insulin, and progressive β-cell failure. Although lifestyle interventions and pharmacological agents are popular, these measurements do not protect from long term disease progression.
Diabetes complications like nephropathy, neuropathy or cardiovascular disease hold a great impact in morbidity and mortality. The existing treatment options concentrate on metabolic control, rather than endogenous pancreatic restoration. This has led to increasing interest in therapeutic approaches that target the underlying biology of the disease.
Regenerative medicine, particularly MSC-based approaches, is being explored as a potential adjunctive strategy aimed at modifying the disease environment rather than solely managing symptoms.
- Pathophysiology of Type 2 Diabetes Mellitus
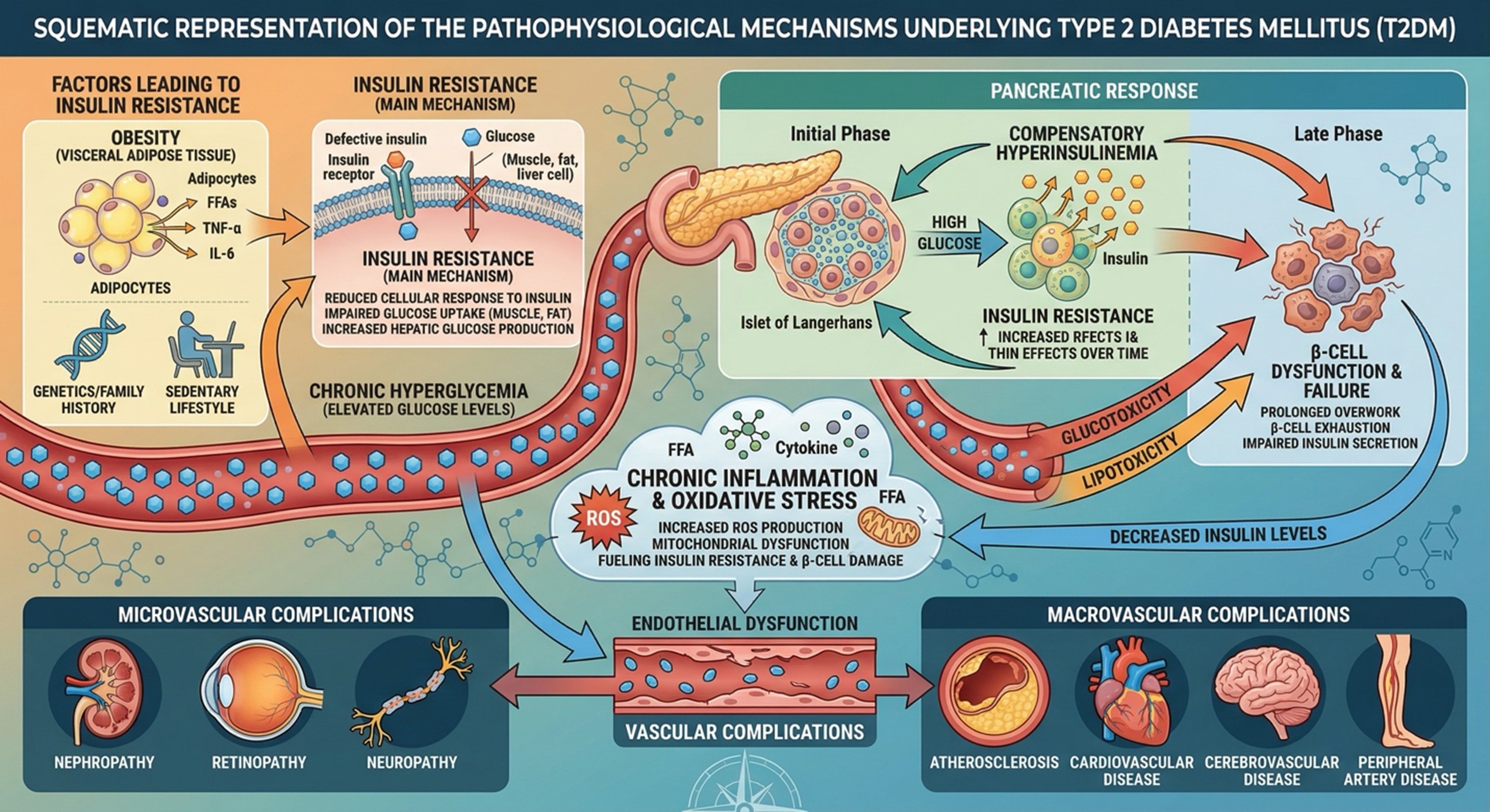
The progression of T2DM is a multifaceted process regulated by metabolic and inflammatory signaling pathways. One notable early feature of the disease is insulin resistance in peripheral tissues, including muscle and adipose tissue. Pancreatic β-cells respond by secreting additional insulin to maintain glucose homeostasis.
At some point, this compensatory mechanism fails. Chronic exaggerated exposure to glucose and lipids are recognised as causing oxidative stress and mitochondrial dysfunction through glucotoxicity and lipotoxicity. These processes result in the death of β-cells and impaired insulin secretion.
In addition, chronic low-grade inflammation caused by cytokines such as TNF-α and IL-6 further disrupts the insulin signalling cascades. Prolonged exposure to elevated Glucose leads to loss of vascular endothelium function and initiation, progression of microvascular/macrovascular complications.
The gradual depletion of functional β-cell mass is a key event in the progression toward disease, when endogenous glucose regulation fails to maintain homeostasis.
Limitations of Conventional Therapeutic Strategies
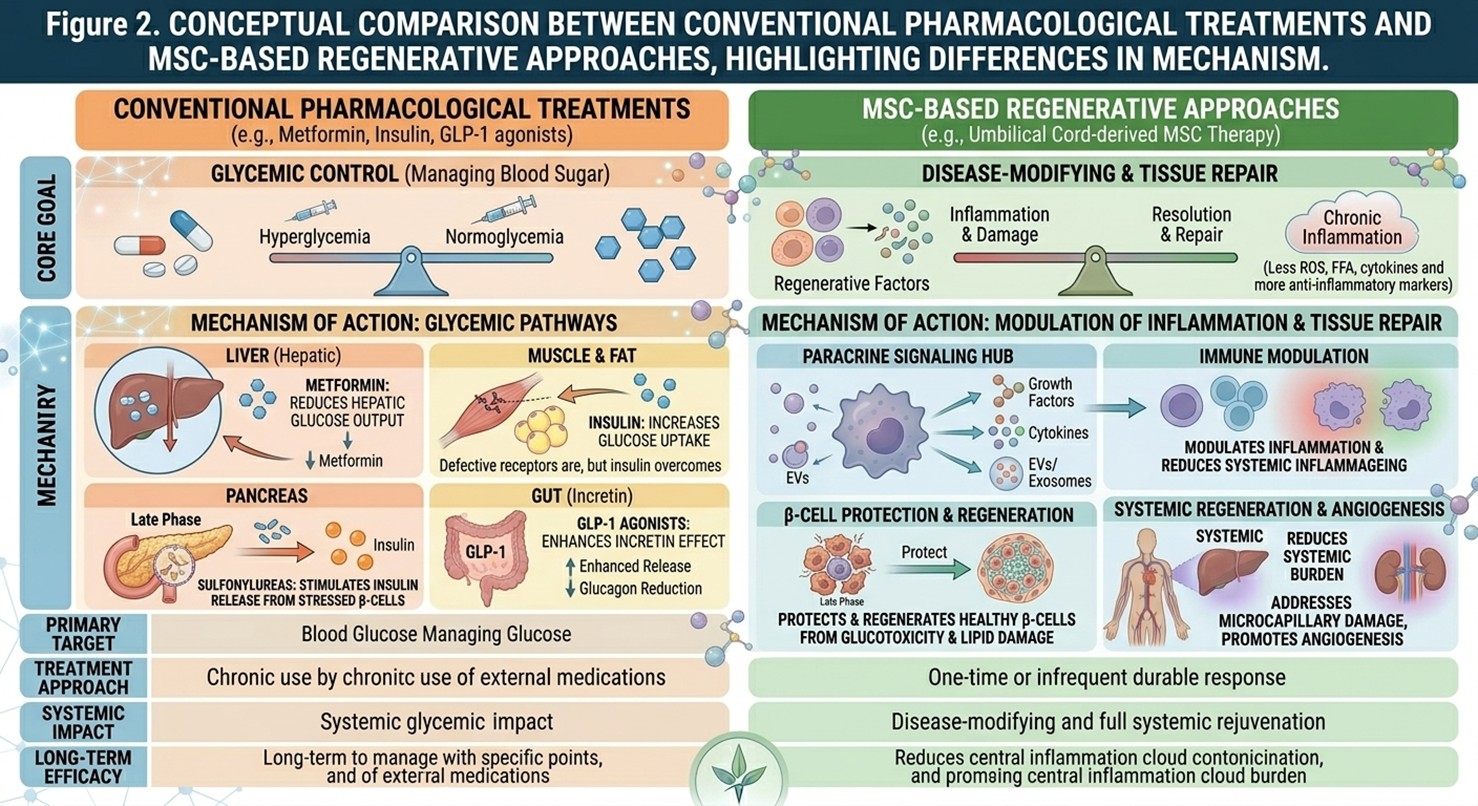
T2DM is commonly treated using various oral hypoglycemic agents alone or in conjunction with insulin therapy. These approaches are effective at decreasing blood glucose and improving glycemic metrics such as HbA1c. However, they do not address the underlying drivers of β-cell dysfunction or chronic inflammation.
Class side effects include hypoglycemia, weight gain and GI adverse events. Also, naturally the natural history of type 2 diabetes is marked by failing β-cell function over time resulting in enlarging treatment regimens.
These classical therapies are also non-regenerative, which is very important. They do not restore pancreatic structure or function, and they do not reverse metabolic derangements at the cellular level. This highlights the need for strategies that focus on endogenous disease modifying pathways.
MSC Heterogeneity and Effects on MSC Therapies
Therefore, mesenchymal stem cells (MSCs), especially deriverd from umbilical cord tissue (UC-MSCs) is being studied for their regeneration and immunomodulation activity. These cells are thought to exert their functions at least in large part by a paracrine mechanism and not direct replacement of tissue.
MSCs secrete pro- and anti-inflammatory cytokines, growth factors, and extracellular vesicles that can modulate the tissue microenvironment. Proposed mechanisms include:
- Modulation of inflammatory pathways
- Reduction of oxidative stress
- Promotion of angiogenesis
- Support of cellular repair processes
- Regulation of immune responses
Several studies have found that several MSCs (mesenchymal stem cells) available in human tissues may be effective for T2DM, given their potential to increase insulin sensitivity, retain β-cell function and epithelial repair of pancreatic tissue. Preclinical studies indicate that factors secreted from MSCs can mitigate inflammatory infiltration and promote β-cell survival, but the translation to clinics still needs to be explored.
Clinical Applications in Thailand and translation
Thailand has become a hub for regenerative medicine in the region and offers a range of specialized clinics with laboratory and cell-processing capabilities. Such facilities follow globally acceptable standards in the handling and monitoring of cells.
Although MSCs stem cell can be delivered by diverse means (including subcutaneous injection, topical application, and ectopic administration), the most commonly employed route for clinical investigation involve systemic delivery of MSC stem cell systemically 95, 96. These strategies seek to influence systemic inflammation and enhance metabolic homeostasis.
The early clinical experience indicates improvement of metabolic markers and patient reported outcomes in selected cases. However, those therapies remain investigational and outcomes may differ. Advanced medical technology of Thailand integrated with caring hospital environments has prompted to its faster growing as a popular choice for regenerative treatment approaches.
Future Perspectives
The applicability of MSC stem cell in T2DM marks a change toward disease-modifying approaches. Research is ongoing to better understand:
- Mechanisms of action on a molecular level
- Optimal dosing and administration protocols
- Long-term safety and efficacy
- Patient selection criteria
New strategies such as exosome-mediated therapies may also expand the application, facilitating parental stem cells to deliver regenerative signals without including these cells directly into target tissue.
Such approaches require testing on large scale clinical trails to establish their therapeutic utility and define a role in standard medical practice.
Conclusion
Type 2 Diabetes Mellitus: A Continuing Global Burden. And though conventional therapies remain central to glycemic control, they do not address the biological drivers of disease progression. MSC-derived regenerative strategies could be used to modulate inflammation, promote tissue repair and enhance metabolic homeostasis. Hong Kong, Thailand and clinical applications reflect that there is increasing interest in such therapies as part of an integrative treatment strategy.
Nevertheless, these approaches should be viewed interrogative and additional work is needed to determine their clinical long-term value. As regenerative therapies progress, MSC type treatments will potentially become a larger area of metabolic disorder management.

