Alzheimer’s disease is a progressive condition that gradually impairs memory, cognition, and the ability to perform everyday activities. At the biological level, it is defined by the accumulation of amyloid-beta plaques and neurofibrillary tau tangles, both of which disrupt communication between neurons and eventually lead to widespread cell death. Despite advances in modern medicine, current drug therapies primarily focus on symptom management and do little to stop or reverse the disease process. This limitation has driven growing interest in regenerative approaches such as Mesenchymal Stem Cell (MSC) therapy, particularly those derived from umbilical cord tissue (UC-MSCs), which aim to address the underlying causes of neurodegeneration.
The Power of Stem Cells for Alzheimer’s Disease
Stem cells exert most of their benefits through paracrine signaling. This process involves the release of biologically active molecules that support and protect existing brain cells. These include neurotrophic factors such as brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), glial cell line-derived neurotrophic factor (GDNF), vascular endothelial growth factor (VEGF), and insulin-like growth factor (IGF).
These signaling molecules play a crucial role in enhancing neuronal survival, promoting synaptic connectivity, and increasing brain plasticity—the brain’s ability to adapt and reorganize. This is particularly important in Alzheimer’s, where synaptic loss is closely linked to cognitive decline.
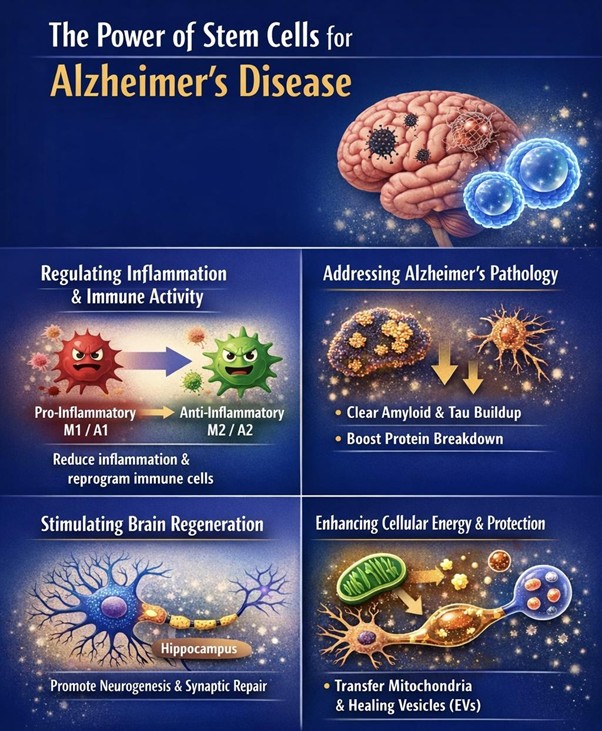
Regulating Inflammation and Immune Activity: Chronic neuroinflammation is a major driver of Alzheimer’s progression. In the diseased brain, immune cells such as microglia and astrocytes become overactivated, shifting into pro-inflammatory states that contribute to neuronal damage. Stem cells help counteract this harmful process by releasing anti-inflammatory cytokines, including interleukin-10 (IL-10) and transforming growth factor-beta (TGF-β).
In addition to suppressing inflammation, stem cells actively reprogram immune cells. They encourage microglia and astrocytes to transition from destructive phenotypes (often referred to as M1 and A1 states) into protective, repair-oriented forms (M2 and A2 states). This shift not only reduces ongoing inflammation but also creates a more favorable environment for healing and regeneration.
Addressing Core Alzheimer’s Pathology: A distinguishing feature of stem cell therapy is its ability to target the hallmark abnormalities of Alzheimer’s disease. Stem cells contribute to the reduction of amyloid-beta and tau pathology through several complementary mechanisms. They enhance the activity of microglia, enabling these immune cells to more effectively identify and remove amyloid deposits. At the same time, stem cells stimulate the production of enzymes such as neprilysin and insulin-degrading enzyme (IDE), both of which are involved in breaking down amyloid proteins.
Furthermore, stem cells support intracellular cleanup systems, including autophagy and lysosomal pathways, which help degrade accumulated toxic proteins. They also release regulatory molecules that assist in cellular repair and promote neuron survival. Through these combined actions, stem cells may help reduce the toxic burden within the brain and restore a healthier neural environment.
Stimulating Brain Regeneration: One of the most promising aspects of stem cell therapy is its potential to activate the brain’s intrinsic repair mechanisms. In Alzheimer’s disease, the natural process of neurogenesis—the formation of new neurons—is significantly impaired. Stem cells have been shown to stimulate dormant neural stem cells, particularly in regions like the hippocampus, which is essential for memory formation.
By enhancing neurogenesis and increasing synaptic density, stem cells may help rebuild neural networks that have been damaged by disease. This restoration of connectivity is critical for improving cognitive function and slowing the progression of memory loss.
Enhancing Cellular Energy and Protection: Beyond structural repair, stem cells provide important metabolic and protective support to neurons. One remarkable capability is their ability to transfer mitochondria—the energy-producing components of cells—to damaged neurons. This mitochondrial transfer can improve cellular energy production and help restore normal function in stressed brain cells.
Additionally, stem cells release extracellular vesicles (EVs), which are small particles containing proteins, lipids, and genetic material such as microRNAs. These vesicles act as carriers of therapeutic signals, delivering instructions that reduce inflammation, enhance neuron survival, and strengthen synaptic communication. MicroRNAs such as miR-21 and miR-124 play key roles in regulating these protective processes.
Emerging Clinical Evidence
Early findings are encouraging. Individual case reports have described noticeable improvements in mobility, social interaction, and coordination following stem cell treatment in advanced-stage patients. Clinical trials are also underway to evaluate the safety and efficacy of stem cell infusions in individuals with early-stage disease, with a focus on reducing inflammation and slowing cognitive decline.
In addition, experimental approaches involving direct administration of stem cells into the brain have shown reductions in amyloid and tau levels, along with signs of cognitive stabilization or improvement in small patient groups. While these results are preliminary, they highlight the therapeutic potential of this approach.
A Multifaceted Therapeutic Strategy
Stem cell therapy stands out because of its comprehensive mode of action. Rather than targeting a single pathway, it simultaneously addresses multiple aspects of Alzheimer’s pathology. These include reducing inflammation, clearing toxic proteins, supporting neuronal survival, stimulating regeneration, and improving cellular metabolism.
Looking Forward
As research continues to evolve, stem cell therapy is emerging as a promising candidate for transforming the treatment landscape of Alzheimer’s disease. Its ability to target root causes rather than just symptoms represents a significant shift in therapeutic strategy. While further large-scale clinical trials are needed to confirm long-term safety and effectiveness, the current evidence suggests that stem cell-based interventions could one day move from experimental use to standard clinical practice.
In the future, this regenerative approach may not only slow disease progression but also help restore lost cognitive function, offering new hope to patients and families affected by this challenging condition.

