UC-MSCs Molecular Integration and Advanced Neuro-Regenerative Interventions in Multiple Sclerosis in Thailand
The clinical trajectory for managing Multiple Sclerosis has historically centered on neutralizing the inflammatory surge to prevent relapses. However, traditional disease-modifying therapies often hit a ceiling when addressing the progressive neurodegeneration that defines the disease’s later stages. This therapeutic gap has catalyzed the evolution of stem cell therapy in Thailand, with a particular focus on Umbilical Cord-derived Mesenchymal Stem Cells (UC-MSCs). These neonatal cells are no longer viewed merely as passive immune suppressors but as active biological architects capable of rewriting the pathological script of the central nervous system through sophisticated molecular interventions.
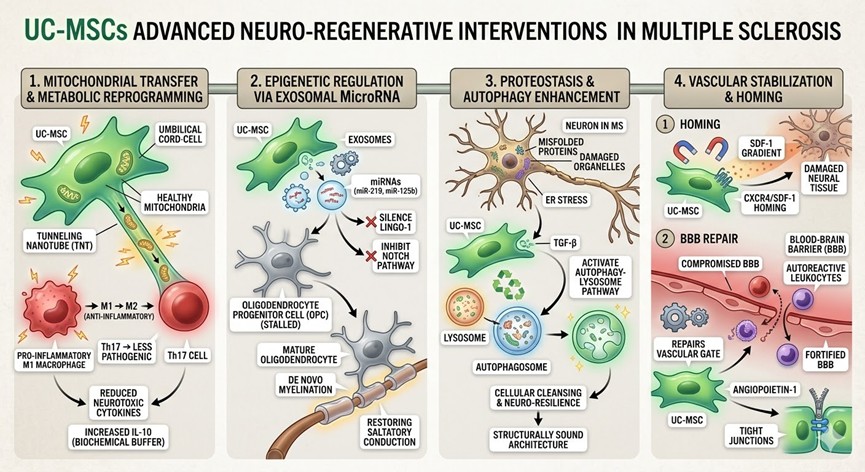
1.Mitochondrial Transfer and Metabolic Reprogramming
A frontier mechanism currently gaining traction in the study of UC-MSCs is the Immunometabolism Shift. In a patient suffering from Multiple Sclerosis, the immune landscape is dominated by pro-inflammatory M1 macrophages and pathogenic Th17 cells that rely on anaerobic glycolysis for energy. Recent high-resolution cellular studies indicate that UC-MSCs can actively donate healthy mitochondria to these distressed cells through Tunneling Nanotubes (TNTs).
This mitochondrial transfer effectively reboots the metabolic profile of the immune microenvironment. By restoring oxidative phosphorylation in resident microglia and infiltrating T-cells, UC-MSCs induce a phenotypic switch from the aggressive M1 state to the anti-inflammatory M2 state. This metabolic reprogramming results in a sustained reduction of neurotoxic cytokines while simultaneously increasing the secretion of Interleukin-10 (IL-10), creating a biochemical buffer zone that protects vulnerable axons from further demyelination.
2.Epigenetic Regulation via Exosomal MicroRNA
The advancement of stem cell therapy in Thailand has shifted significant attention toward the secretome, specifically the role of extracellular vesicles or exosomes. These nano-scale messengers derived from UC-MSCs carry a cargo of specific MicroRNAs (miRNAs) that act as epigenetic regulators. In the context of Multiple Sclerosis, the failure of remyelination is often linked to the arrest of Oligodendrocyte Progenitor Cells (OPCs) due to inhibitory signaling pathways like LINGO-1.
Cutting-edge research reveals that UC-MSCs-derived exosomes deliver miR-219 and miR-125b directly to these stalled OPCs. These miRNAs silence the LINGO-1 gene and inhibit the Notch signaling pathway, which are the primary molecular brakes preventing myelin repair. By removing these biological obstacles, stem cell therapy in Thailand facilitates de novo myelination—the actual reconstruction of the myelin sheath—thereby restoring the saltatory conduction of nerve impulses that is lost during the progression of Multiple Sclerosis.
3.Proteostasis and the Enhancement of Autophagy
Neurodegeneration in Multiple Sclerosis is characterized by the accumulation of misfolded proteins and damaged organelles within neurons, leading to Endoplasmic Reticulum (ER) stress and eventual apoptosis. UC-MSCs intervene in this process by modulating Proteostasis or protein homeostasis. Through the secretion of Transforming Growth Factor-beta (TGF-β), these stem cells activate the autophagy-lysosome pathway within neurons.
This cellular cleansing process allows neurons to break down and recycle toxic protein aggregates that accumulate during chronic inflammation. By enhancing the neuron’s innate ability to maintain its internal protein environment, UC-MSCs provide a layer of neuro-resilience. This ensures that even in the presence of low-level inflammation, the neural architecture remains structurally sound, effectively slowing the transition from relapsing-remitting Multiple Sclerosis to secondary progressive Multiple Sclerosis.
4.Vascular Stabilization and the SDF-1 Gradient
The infiltration of the central nervous system in Multiple Sclerosis depends on a compromised Blood-Brain Barrier (BBB). UC-MSCs exhibit a highly refined homing capability driven by the CXCR4/SDF-1 chemokine axis. These cells sense the SDF-1 gradient emitted by damaged neural tissues and migrate with high precision toward the site of the lesion.
Upon arrival, UC-MSCs do not just linger; they actively repair the vascular gate. By releasing Angiopoietin-1 and specialized junctional adhesion molecules, they reinforce the tight junctions of the endothelial cells forming the BBB. This sealing effect is crucial as it prevents the influx of further autoreactive leukocytes into the CNS. This dual action—migrating to the damage and then fortifying the barrier—makes stem cell therapy in Thailand a uniquely comprehensive approach to halting disease activity at its entry point.
5.The Clinical Landscape of Regenerative Medicine in Thailand
The application of UC-MSCs for Multiple Sclerosis in Thailand has moved into a highly specialized phase. Leading clinical centers now utilize Low-Oxygen expansion techniques to maintain the stemness and potency of UC-MSCs, ensuring they retain their neonatal vigor before administration. The protocols have evolved to include synergistic delivery routes; while intravenous infusion manages systemic autoimmunity, intrathecal administration—delivering cells directly into the cerebrospinal fluid—is used to bypass the BBB and maximize the concentration of neurotrophic factors around spinal cord lesions.
This precision medicine approach is often combined with advanced neuro-rehabilitation to prime the brain for the regenerative signals provided by the stem cell therapy in Thailand. By integrating cellular biology with clinical expertise, Thai specialists are establishing new benchmarks for safety and efficacy, focusing on long-term disability stabilization and the improvement of the Expanded Disability Status Scale (EDSS) scores in patients who have exhausted conventional options.
The paradigm of treating Multiple Sclerosis is undergoing a fundamental transformation, moving away from simple immune suppression toward a multi-targeted regenerative strategy. The deep molecular mechanisms of UC-MSCs—ranging from mitochondrial transfer and epigenetic regulation via exosomes to the stabilization of the blood-brain barrier—offer a sophisticated arsenal against the complexities of Multiple Sclerosis. As stem cell therapy in Thailand continues to refine these protocols and harness the full paracrine potential of umbilical cord derivatives, the goal of reversing neurological deficits is becoming a scientific reality. The dialogue between these primitive cells and the damaged nervous system represents the future of neurology, providing a sustainable path toward healing and recovery for those living with Multiple Sclerosis.

