Autism Spectrum Disorder (ASD) is a very complex neurodevelopmental issue which we see manifest in terms of social and communication deficits and also in the presentation of restricted and repetitive behaviors. We are seeing that neuroinflammation, immune dysregulation and changes in neural connectivity play a role in the cause of it. Presently our treatment options are mostly supportive which include behavioral interventions and management of symptoms. Recently we have looked at Mesenchymal stem cells (MSCs) in general and Umbilical cord derived MSCs (UC-MSCs) in particular for their immun modulatory and neuro supportive properties. Via paracrine action and interaction with the immune system UC-MSCs may play a role in modulating inflammation and in the neural microenvironment. This review looks at the bio basis of ASD, the issues with present interventions and the growing role of UC-MSC based therapies in the field of regenerative medicine.
- Introduction
Autism Spectrum Disorder (ASD) is a very diverse neurodevelopmental issue which is seeing growing global prevalence. It is characterized by social and communication deficits as well as repetitive behaviors and narrow interests. The cause of ASD is many factor which includes genetic, environmental, and neurobiological elements.
While still at the forefront are early behavioral and educational interventions, we do not at present have any therapies which target the base biological issues in ASD. What we are seeing is a growth in research which is putting focus on the role of neuroinflammation and immune system dysregulation in terms of its play in changed brain development and function. Regenerative medicine which includes the use of mesenchymal stem cells (MSCs) is a focus for what we are seeing as an additive treatment for these biological pathways.
- Biological Mechanisms in Autism Spectrum Disorder
The in large part in which ASD is a product is of complex interactions of the central nervous system with that of the immune system. What we have seen to be true is:
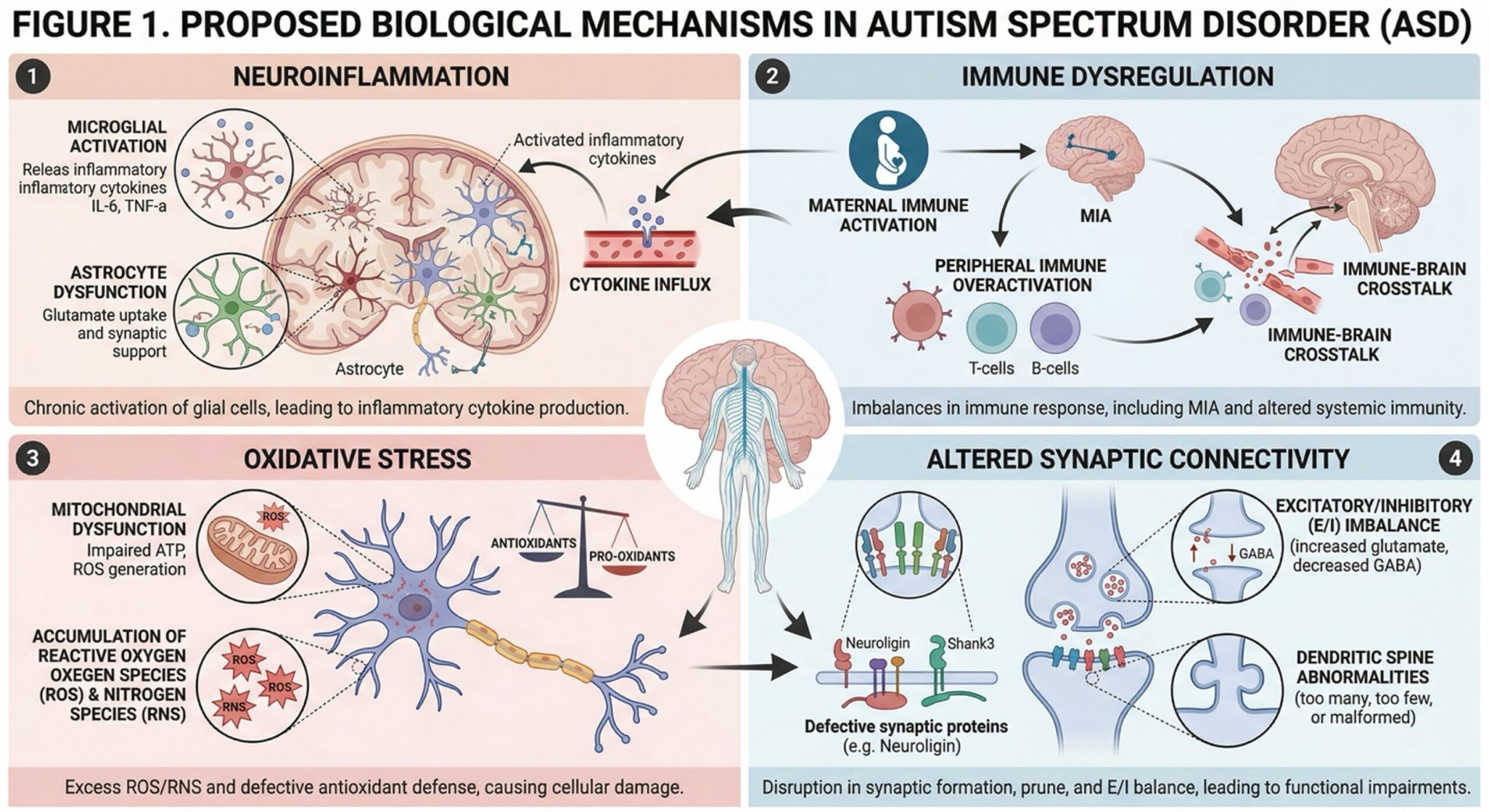
Brain inflammation
In some people with ASD we see high levels of pro-inflammatory cytokines which points to ongoing neuroinflammation.
Immune Imbalance
Changes in both innate and adaptive immune responses may play a role in abnormal neurodevelopment and synaptic function.
Neurotransmission Failure
Impaired which is reported is that impaired synaptic connectivity and plasticity play a role in the behavior and cognition of ASD.
Oxidation Stress
Increased free radical activity may play a role in neuronal damage and altered cellular signaling.
Blood Brain Barrier Integrity
Disruption of the blood brain barrier may allow for the entry of peripheral inflammatory agents into the central nervous system.
These interrelated processes present that ASD may have both neurological and immunological elements which in turn puts forth a case for therapies which target the neuroimmune axis.
- Limitations of Current Therapeutic Approaches
Today, the management of ASD approaches mainly behavioral therapies, educational genericities and pharmacological treatment concern all associated symptoms such as irritability, anxiety or hyperactivity.
Although these strategies are powerful and have potent implications for functional recovery they do not target the underlying biology such as neuroinflammation or immune dysregulation. Pharmacological options are often limited due to side effects and variable efficacy.
This has led to growing interest in therapies that could affect the brain’s biological milieu and enhance neural function.
- UC-MSCs in Regenerative Medicine for ASD
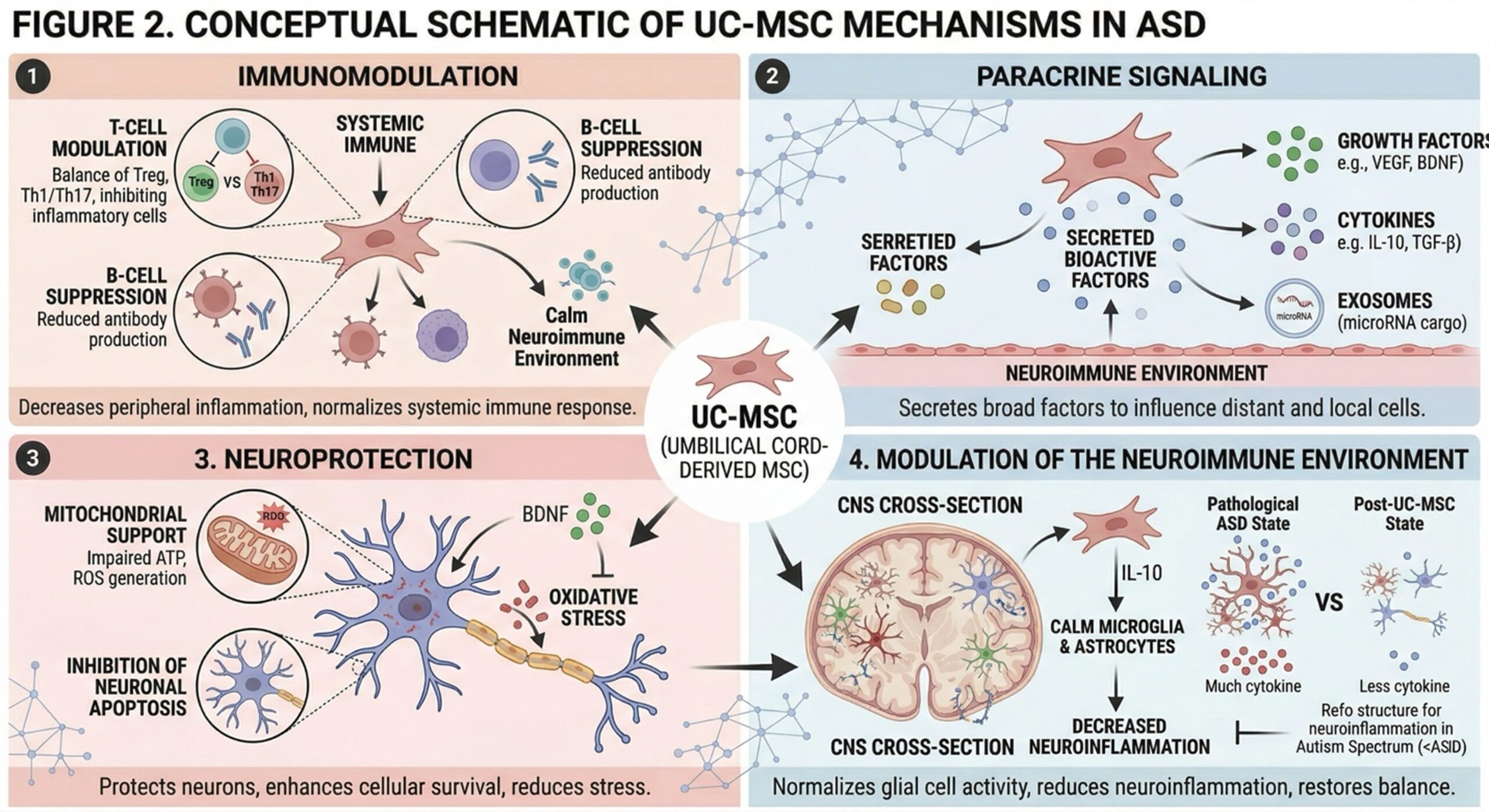
Umbilical cord–derived mesenchymal stem cells (UC-MSCs) have been studied for their immunomodulatory and neuro-supportive functions. The main function of these cells is not to substitute neurons, but rather they have effects through the secretion of bioactive molecules.
Paracrine Signaling
UC-MSCs secrete mediators such as growth factors, cytokines and extracellular vesicles that affect cellular communication and tissue homeostasis.
Immunomodulation
UC-MSCs regulate immune responses through two ways; inhibit T-cell signaling and initiate anti-inflammatory pathway.
Neuroprotective Effects
MSC-derived factors can have a neuroprotective effect through the reduction of oxidative stress and enhancement of the neural microenvironment.
Modulation of Neuroinflammation
UC-MSCs might reduce neuroinflammatory activity associated with ASD by interacting with immune cells.
Support of Neural Connectivity
Recent studies indicate that MSC-released signaling molecules have the potential to modify synaptic plastic events and CNS network activity.
These mechanisms are under investigation in preclinical and early-phase clinical studies.
- Clinical Translation and Applications
In recent years, there has been a growing clinical interest in MSC-based therapies for ASD (Autism Spectrum Disorders). Exploratory studies that evaluate safety and potential functional outcomes have surfaced. Clinically, UC-MSCs are usually delivered systemically (IV infusion) and intrathecally in some protocols.
These findings support the possible benefits of MSC-targeted therapies as indicated by changes seen in inflammatory markers and, in some studies, improvement of behavioral or functional measures. These observations, however, are variable and need larger controlled studies to be demonstrated.
Protocols combining regenerative approaches with support care have also been developed in specialized treatment centers (in regions such as Thailand). These environments grant access to complex laboratory processes and multidisciplinary clinical teams.
It is very important to note that UC-MSC therapies for ASD are investigational and not standard treatment.
- Future Perspectives
UC-MSCs for ASD are an innovative, still developing component at the neuroimmune interface. Future studies aim to:
- Elucidate mechanisms of action on a molecular and cellular level
- Determine optimal dosing and administration regimens
- Determine long-term safety and efficacy
Clinical end points and surrogate markers as end points: define patient subgroups most likely to benefit Moreover, whole-cell delivery systems (like exosome-mediated drugs) may provide another mechanism to evoke regenerative signals. Randomized controlled trials with appropriate design will be needed to confirm clinical relevance and inform future therapeutic utilization
Conclusion
Autism Spectrum Disorder is a multifaceted disorder comprised of neurological and immunological components. Existing therapies are mainly aimed at managing the symptoms and providing behavioral support, being unable to target biological processes lying beneath the disease.
Umbilical cord–derived mesenchymal stem cells are a new-type regenerative medicine with the specific advantage of immunomodulatory and paracrine effects. UC-MSC-based approaches may represent a new approach for maintaining neurological function through attenuation of neuroinflammation and modulation of intracellular signaling pathways. These therapies, however, are investigational and their clinical role needs to be studied.

