Sensorineural hearing loss (SNHL) is an important unmet medical need due to the limited natural regenerative potential of mammalian cochlear hair cells and auditory neurons. There is interest in other types of MSCs, such as umbilical cord mesenchymal stromal cells (UC-MSCs), because they are relatively easy to obtain and expand, and remain biologically active due to immunomodulatory signaling and paracrine activity. In otology, however, the field is quite nascent. The small number of human studies currently available focus not on standardized whole-cell UC-MSC protocols, but in general on heterogeneous cell products such as autologous cord blood-derived mononuclear cells and early case reports using UC-MSC-derived extracellular vesicles (EV). MSC SNHL preclinical studies are highly disparate regarding dosing and administration routes, pointing to an absence of a consensus dose. Thus, current evidence supports characterizing UC-MSC therapy for hearing loss as investigational, with dosage considered a research variable rather than established clinical standard.
- Introduction
The active battle against hearing loss is most often focused through the lens of SNHL, which is characterized by damage to cochlear hair cells, supporting cells, synapses and spiral ganglion neurons. While stem cell-based approaches are considered biologically appealing, recent reviews outline ongoing translational hurdles such as engraftment, cochlear incorporation, delivery limitations and immune problems/safety. Among stem cell sources, mesenchymal stromal cells are often raised as they can be more easily obtained and expanded than other potential cell types and their secreted trophic, anti-inflammatory, and immunomodulatory properties have been the subjects of extensive investigation.
That said, as befits a journal discussion here, I think mechanism can be disentangled from proof. Current literature substantiates a rationale for UC-MSC based approaches in hearing loss, but not yet a standardized clinical treatment paradigm. In practice, “UC-MSC therapy for hearing loss” refers not to one validated, dose-defined approaches but rather a translational research space that includes whole-cell approaches, secretome concepts and UC-MSC-derived EV strategies.
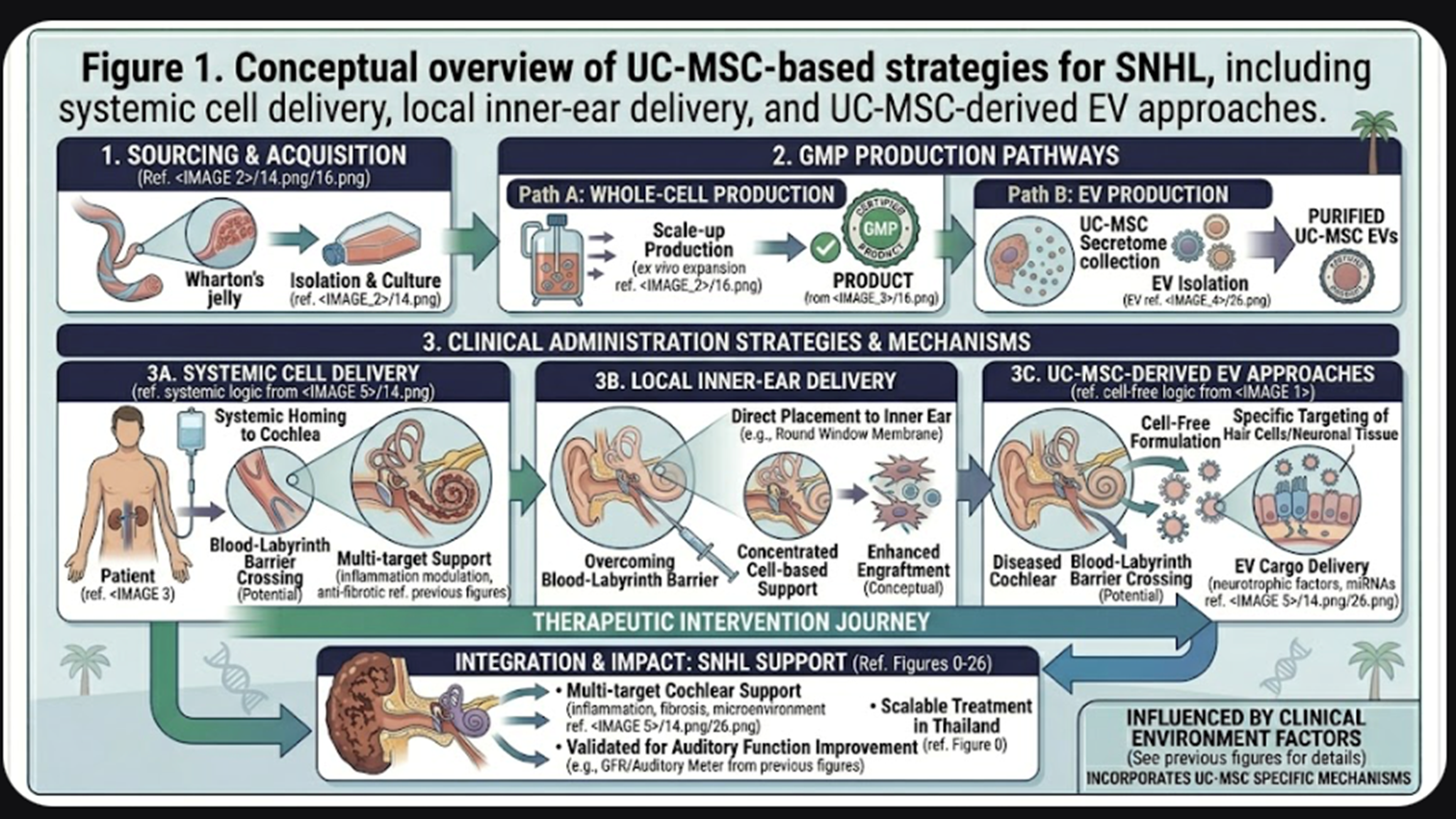
- Why UC-MSCs Are Being Considered for Hearing Loss
This is another reason for why UC-MSCs are being investigated; they may affect the damaged inner ear through paracrine signaling instead of replacing cells that were lost. Reviews for stem cell therapy for hearing loss emphasize the role of MSCs in modulating inflammation, secreting trophic factors and cytoprotection that may promote the survival of auditory cells in hostile cochlear conditions. Notably, this mechanism-based interest is most relevant in acquired SNHL settings including ototoxic injury, noise injury, inflammatory inner-ear damage and peri-implant traumatic injuries.
Some of the most mechanistic support comes from studies of preclinical UC-MSC-derived EV. In a cisplatin mouse model, UC-MSC exosomes improved hearing outcomes and diminished outer hair cell loss, also altering cochlear gene-expression patterns in ways expected for tissue protection and repair. In related ex vivo studies, MSC-derived exosomes diminished cisplatin-induced toxicity in auditory hair cells suggesting that some of the phenomenon known to span organ and species type may be mediated per vesicle cargo secreted.
- Current Evidence Base: Stronger in Animals Than in Humans
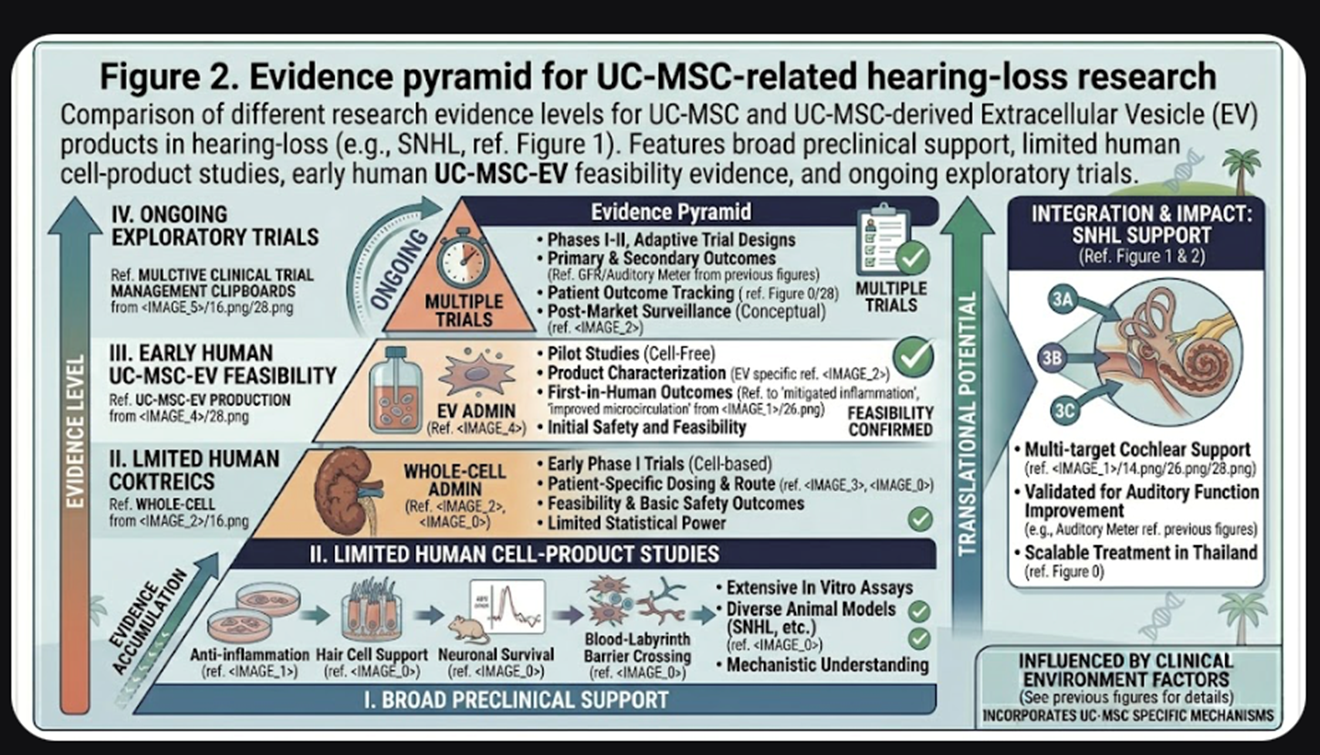
The evidence base for MSCs in hearing loss is still skewed to animal studies. A systematic review of preclinical MSC studies in SNHL7 found 12 animal studies, and reported that MSC-treated groups improved aura brainstem response and distortion product otoacoustic emission outcomes; overall, heterogeneity was high with many unclear risk-of-bias domains. Importantly for the discussion of doses, the review noted that MSC doses varied from 4 × 10^3 to 1 × 10^7 cells with differing tissue sources and delivery routes in each included study.
Human evidence is much thinner. A narrative review from 2025 on stem cells and restoration of hearing concluded that the field remains limited by concerns regarding immune rejection, limited survival of implanted cells, challenges with integration, and the complexity of regulatory issues despite promising preclinical data. The same review is reflective of the general state of the literature: human otology data still remains scarce, and no UC-MSC whole-cell treatment regimen could be found within the SNHL literature that can really be classified as standard care.
- What Human Data Actually Exist
The most famous such cell experiment in humans is not a cultured UC-MSC study, but a phase 1 trial of autologous umbilical cord blood mononuclear cells in children with acquired SNHL. In that study, 11 children were given intravenous autologous cord blood mononuclear cells at a dose of 5 to 30 million cells/kg; there were no reported adverse effects and some participants showed reductions in auditory brainstem response thresholds. This is significant because it shows that hearing-loss cell therapy has tested in humans, but cannot be described as a validated UC-MSC dose. It used a different cell product and had a different biological composition compared to culture-expanded UC-MSCs.
The most explicit human evidence published, to date, regarding the application of a UC-derived mesenchymal product is the first documented intracochlear use of allogeneic UC-MSC-derived EVs (MSCEVs) in a 55-year-old male patient undergoing cochlear implantation for treatment of Ménière’s disease. The report stated the GMP produced suspension of 1.03 × 10^11 UC-MSC-EVs/mL and concluded feasibility of the procedure, but safety and efficacy require controlled clinical trials which were explicitly mentioned. This is again important translational evidence, but it is not a standardized therapeutic dose for hearing loss.
Furthermore, at least one recently registered clinical trial will specifically evaluate the use of intratympanic small extracellular vesicles derived from mesenchymal stem cells for severe and profound sudden SNHL; thus, this research is still in an exploratory stage and testing locally delivered vesicle products rather than an established whole-cell UC-MSC protocol.
Conclusion
Ultimately, UC-MSC therapies for hearing loss is a scientifically stimulating, but clinically underdeveloped research area. Preclinical work of protective and reparative effects in animal and explant models provides the strongest basis, although human evidence remains sparse and heterogeneous. The most prudent and currently defensible conclusion is that dosage for UC-MSC therapy in hearing loss is investigational, rather than standardized. For a journal-style audience, it is therefore more appropriate to emphasize class of product, route and context of the study rather than present a single recommended dose.

