If one reads enough diabetes materials, one soon finds that while scientific progress is promising, the level of vocabulary on the Internet often precedes evidence. In diabetes written matter as well as speech lived online, that is more true than for a key concept like Botanical Stem Cell Supplements. It sounds up-to-date, scientific, and even a bit futurist. However, once you enter diabetes literature, the picture must be seen in a different light. In conventional diabetes management, supplements are not regarded as an effective means of lowering blood sugar or relieving diabetes in a credible way. The American Diabetes Association concludes that without evidence for deficiency, nonverbal or herbal supplementation is neither required nor recommended-for people with diabetes. It goes further. The U.S. National Center for Complementary and Integrative Health declares there is no reliable proof that herbal remedies can control diabetes or its complications.
That does not mean plants are irrelevant to diabetes research. Far from it. In the literature, plant-derived compounds appear frequently. But the papers usually discuss them as plant extracts, phytochemicals, or bioactive compounds, not as a well-established therapeutic category called Botanical Stem Cell Supplements. A 2022 review of plant-derived bioactive substances in diabetes described medicinal plants and their compounds as interesting because they may influence hyperglycemia, oxidative stress, and metabolic pathways linked to diabetes, although it also noted that mechanistic data remain limited. A 2021 review on plant extracts for type 2 diabetes similarly emphasized anti-inflammatory, antioxidant, and insulin-resistance-related pathways as major reasons plant compounds remain attractive research targets.
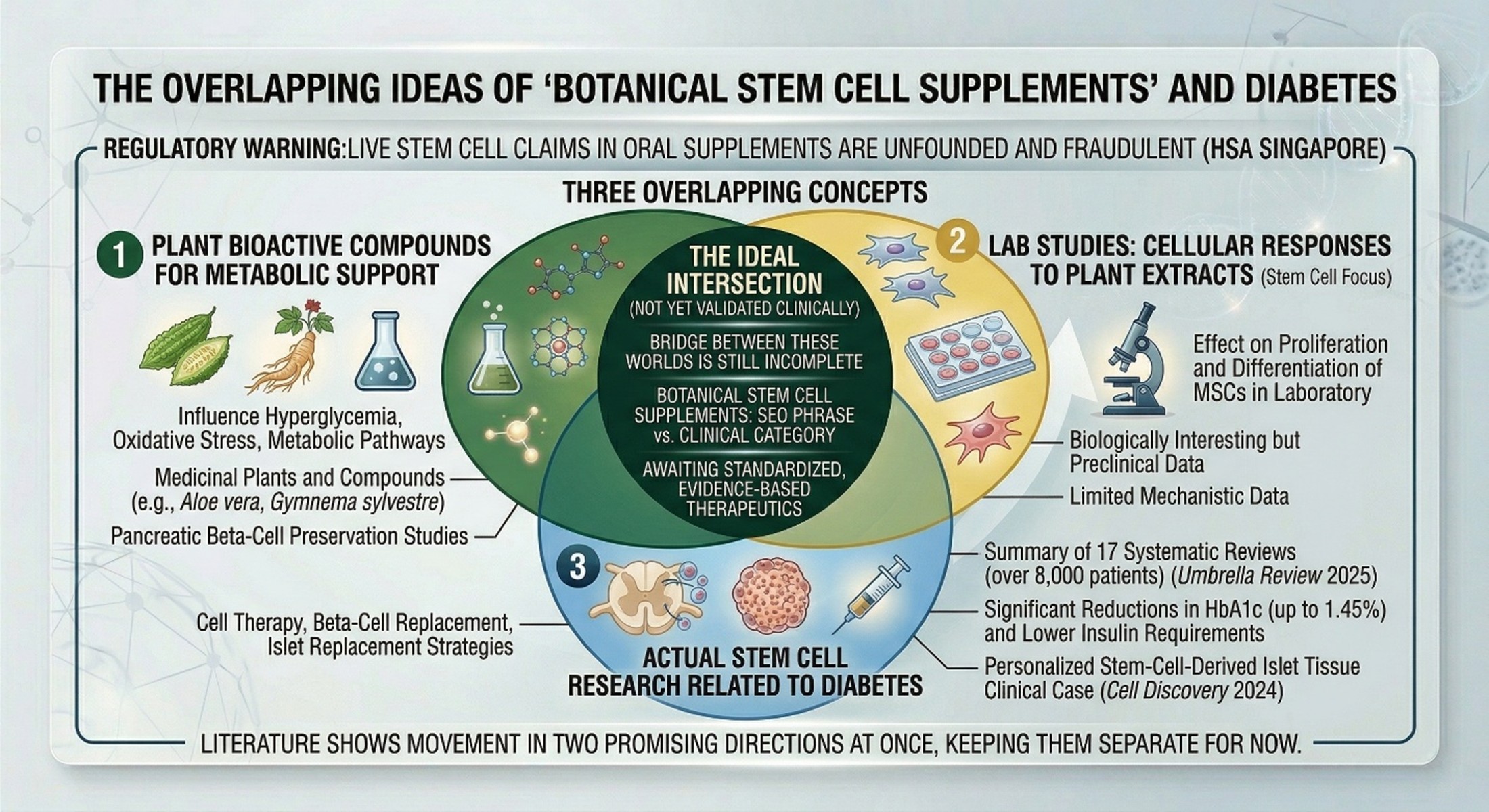
If you keep reading, a second theme appears: some plant-focused papers are especially interested in pancreatic beta-cell preservation. That is where the literature becomes more compelling. In a 2023 systematic review paper on medicinal plants and components for treating pancreatic elevate cells to restore their normal function, it was concluded that eight types of plant and plant-derived compound (Aloe vera, Momordica charantia, Gymnema sylvestre, Panax ginseng, berberine, genistein, naringenin) have good prospects and may support beta-cell functioning, protect beta-cell mass or impact pathways associated with beta cells’ regeneration. A catch, however, soothes ruffled feathers at it serves as the screens applicants from users. Most of the evidence is still preclinical, heavily dependent upon animal models and cell lines and at one remove from humans. It seems, then, that the story defining diabetes research and plants truly is correct, except that these Botanical Stem Cell Supplements have some way to go before they are standardized or evidence-based therapeutics for diabetes.
This is where the keyword becomes tricky in the best possible way for SEO writing. If you think about it, the best way to use Botanical Stem Cell Supplements is just not to pretend as though the phrase represents some guideline-backed diabetes cure. It is therefore stronger and can reduce the likelihood of dishonesty to explain that these words sit at the boundary of three overlapping ideas: on the one hand, plant bioactive compounds for metabolic support; on another level, lab studies into cellular responses when exposed to extracts from plants that have an action on stem cells; with finally actual stem cell research related to diabetes. A 2022 review in Stem Cell Research & Therapy found that plant extracts can affect the proliferation and differentiation of mammalian mesenchymal stem cells in the laboratory, which is biologically interesting. However, that does not amount to proving scientifically that an oral botanical stem cell supplement improves human diabetes outcomes.
This difference becomes even more significant insofar as claims about “stem cell” in supplements quickly become slippery. In a regulatory warning issued by the Singapore Health Sciences Authority, it was stated categorically that claims that live stem cells can be used for improvisational treatment of conditions such as diabetes are unfounded, fraudulent, and scientifically unconvincing. The warning itself was aimed at selling placenta-based supplements rather than any particular botanical diabetes remedy, but the general principle is clear: once ‘stem cell’ enters the world of supplements, rather than making it more difficult to prove, one’s demands for evidence should be higher. No clinical trial data supporting the reasons given in the above warning can be found. For example, it is unwise to suppose that a mere nutrient supplement might supply active adult stem cells into human intestines and thereby cure Diabetes.
If the plant story is one half of the literature, the other half is the field that the papers actually call stem cell research diabetes. This is where things become much more concrete. Here, the work is not about botanical supplements. It is about mesenchymal stem cells, hematopoietic stem cells, pluripotent stem cells, beta-cell replacement, immune modulation, and engineered islet tissue. A 2021 review of stem-cell-based clinical trials for diabetes described stem-cell-derived beta-cell therapy as a way to overcome donor shortage in islet replacement strategies and framed it as a serious therapeutic direction rather than a fringe idea. A 2025 umbrella review of mesenchymal stem cell therapy for diabetes went further, summarizing 17 systematic reviews and meta-analyses representing more than 8,000 patients and reporting improvements in glycemic control, especially in type 2 diabetes, with HbA1c reductions of up to 1.45% and lower insulin requirements in some analyses—while still emphasizing the need for standardized protocols and better long-term data.
The high-profile example came in 2024 when a Cell Discovery paper reported the first human use of personalized endoderm stem-cell-derived islet tissue in a patient suffering from type 2 diabetes and with impaired pancreatic islet function. The paper described substantial improvement over 27 months in glycemic control and put the case as evidence that stemcell–derived islet tissues can restore islet function even late in type 2 diabetes. It was an impressive paper. And that meant it caught the attention of many people. However, it remains a case-level clinical milestone not yet proof that stem cell therapy for diabetes has already become routine care.
So where does that leave a topic like Botanical Stem Cell Supplements and Diabetes? After reading the papers, the honest answer is this: the plant side of the literature is promising mostly in the form of phytochemicals, beta-cell-protective compounds, and metabolic-support mechanisms; the true stem cell research diabetes field is advancing through cell therapy, beta-cell replacement, and mesenchymal stem cell studies; but the bridge between those two worlds is still incomplete. Today, Botanical Stem Cell Supplements is a strong SEO phrase and an intriguing research-adjacent concept, but it is not yet a clearly validated clinical category in diabetes care.
That may actually be the most useful conclusion for a reader. The literature does not say that plants are useless. It does not say stem cells are hype. What it says is more interesting than that. It says Diabetes research is moving in two promising directions at once: one through plant-derived bioactives that may protect metabolism or beta cells, and another through real stem cell research diabetes that aims to restore insulin production or improve glycemic control more directly. The temptation is to collapse both stories into one phrase and call it a breakthrough. The smarter move is to keep them separate for now—and watch very closely as the science catches up


