Type 1 diabetes mellitus (T1DM), also known as Type 1 diabetes or T1D, is a form of chronic autoimmune disease that destroys the insulin-producing pancreatic beta cells. With the decline of functional beta-cells, you are no longer able to produce enough insulin, impairing glucose homeostasis (balance) and so long-term insulin therapy is critical for survival.
Recently, stem cells treatment Thailand have raised more and more attention in regenerative medicine research such as UC-MSCs for the study of autoimmune and metabolic disorders. Designated UC-MSCs have been inspected immunomodulatory, anti-inflammatory, paracrine and tissue-supportive effects in Type 1 diabetes. The current evidence is early, and not yet grounds for saying that any of the approaches are a cure; however, there may be benefits in some populations, such as preservation of residual beta-cell function (in terms of better C-peptide levels and less insulin requirement). According to a 2025 systematic review and meta-analysis after umbilical cord MSC: (u) treatment indicated clinical improvement in HbA1c, insulin requirements, and C-peptide; however it was noted long-term safety/efficacy needs confirmation with larger multicenter clinical trials.
The Introduction type 1 diabetes is more than a blood sugar?
Type 1 diabetes is most commonly considered a disease of high blood sugar, but it is in fact an autoimmune disease. The immune system erroneously destroys the pancreatic beta cells that produce insulin. Over time, the loss of beta cells increases to the point that very little or no insulin is made by the pancreas and there becomes a lack of medication-free options to treat it which means lifelong permanent reliance on treatment with insulin itself.
The standard management for Type 1 diabetes includes initiation of insulin therapy, glucose monitoring, diet planning and exercise; prevention of acidosis and other complications. These tools are crucial to stay alive. However, they do not truly halt the autoimmune process or revert a fully functional beta cell mass. This is why regenerative medicine has emerged as a scientific interest where researchers are currently inquiring whether cell-based therapies such as UC-MSCs might counteract the immune dysregulation, inflammation and microenvironment surrounding the pancreas.
Stem cell therapy Thailand for Type 1 diabetes treatment should be carefully positioned in terms of search engine optimisation NOT as a definitive CURE or replacement of insulin but an investigative and supportive regenerative approach.
A Regenerative Approach to Type 1 Diabetes
2.1 Auto Immune Destruction of Beta Cells
For this reason, the essential biological defect leading to Type 1 diabetes is that of immune-mediated beta-cell destruction. Beta Kejadian in the pancreatic curve, responsible for producing insulin. Chronic hyperglycemia and metabolic instability develop due to the loss of insulin production when these cells are attacked by the immune system.
2.2 Loss of Insulin Production
The entire body is affected because insulin is what allows glucose to enter cells and be utilized for energy. In absence of adequate fasting insulin replacement, blood glucose can rise to hazardous levels. With this in mind any discussion regarding regenerative approaches using UC-MSCs for Type 1 diabetes would need to emphasize that insulin is still the gold standard/first line therapy.
2.3 Rationale for Studying UC-MSCs
Human umbilical cord mesenchymal stem cells became the model of interest, because they are an easily accessible cell source with a robust proliferative capacity, and they implicated less ethical debate than other potential sources for embryonic stem cells which is broadly analyzed in basic and clinical research on diabetes. A review in 2023 suggested potential roles in diabetes and diabetic sequelae, but with the caveat of difficulty in clinical translation.
Type 1 Diabetes and Stem Cell Therapy
Stem cell therapy for Type 1 diabetes broadly encompasses research examining the experimental use of stem cells or products from these cells in supporting immune regulation, tissue repair, and preservation of pancreatic function. For UC-MSCs, the focus is not about directly replacing beta cells. On the contrary, a number of researchers are convinced that UC-MSCs mainly function by mediating biological signaling, regulating immunity and inflammatory pathway changes, secreted extracellular vesicles (EVs), and inducing microenvironmental alterations.
This frames stem cell therapy Thailand with UC-MSCs for Type 1 diabetes as an area at the frontier of regenerative medicine. While it could be applicable for educating patients, consulting on research and future potential therapies but should not be advertised as highly reliable cure.
Categorized Action Mechanisms of UC-MSCs in Type 1 Diabetes
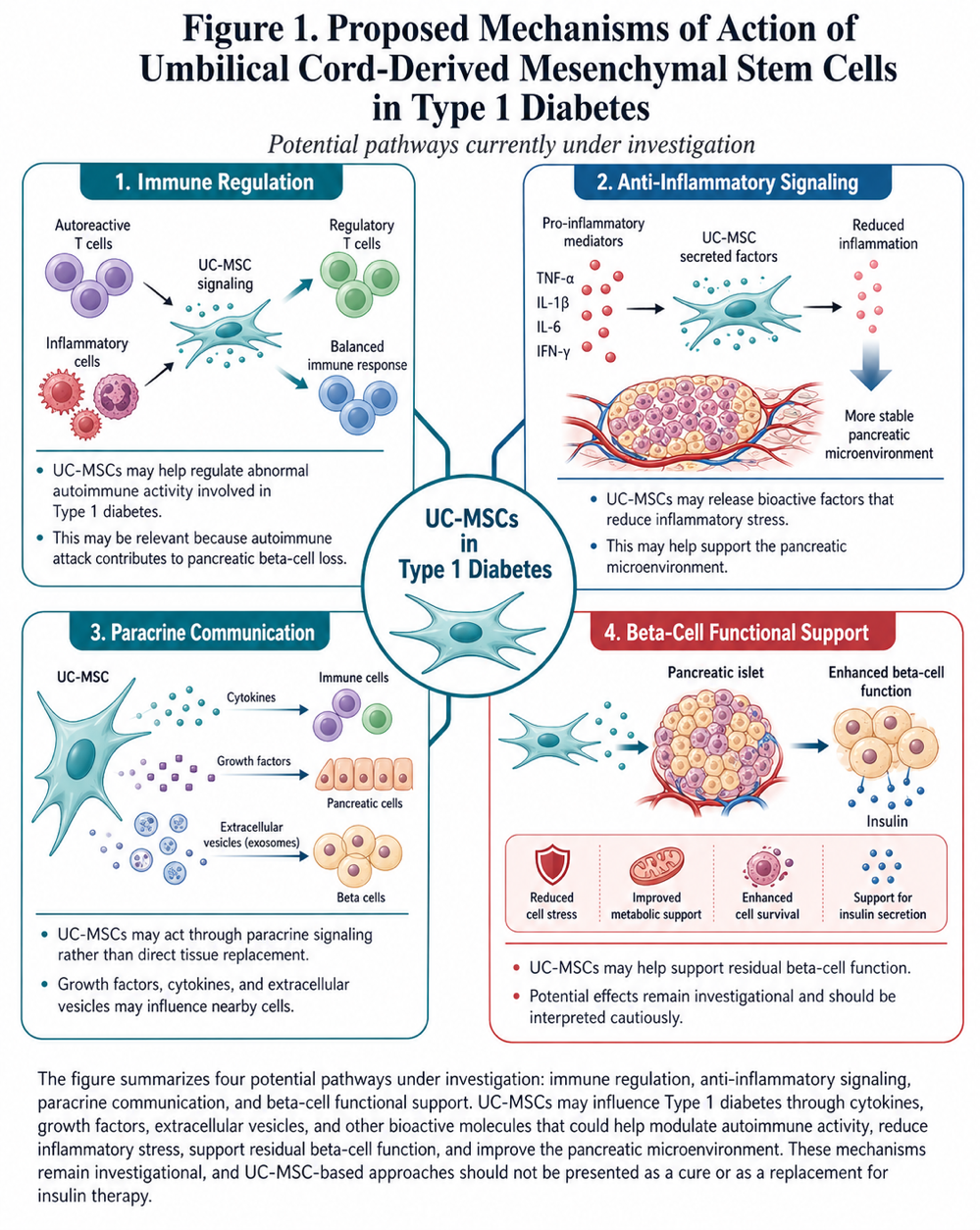
4.1 Immune Regulation
UC-MSCs may modulate the pathogenic immune responses associated with Type 1 diabetes. Due to the autoimmune nature of T1D, a mechanism specifically targeting the immune system has been proposed as one of the most essential approaches in this condition. Interaction studies of MSCs with T cells, B cells, macrophages, dendritic cells and regulatory T cells have been performed. In a 2025 review, it was stated that Type 1 diabetes MSCs act via several immune mechanisms, although clinical trials were not abundant.
4.2 Anti-Inflammatory Effects
One of the keys in the autoimmune context around pancreatic islet beta cells is inflammation. UC-MSCs may secrete bioactive molecules and anti-inflammatory cytokines that counteract inflammatory stress. An experimental autoimmune diabetes model also indicates that UC-MSC transplantation can suppress inflammation and immune attack, delaying the onset of hyperglycemia.
4.3 Paracrine Signaling
Unlike the damage-remote properties of UC-MSCs with a permanent replacement of the damaged pancreatic tissue, they may act through paracrine signaling. That is, they secrete growth factors, cytokines, extracellular vesicles and other substances that affect surrounding immune and pancreatic cells. Mesenchymal stem cells derived from human umbilical cord MSCs may help control diabetes due to their potential for tissue signaling, immune regulation, and the mobilization of pathways underlying repair responses in diabetic tissues.
4.4 Beta-Cell Protection and Functional Augmentation
This has made UC-MSC a promising avenue for Type-1 diabetes research, with one of the primary aims of research being about preservation of residual beta-cell function — so that total regeneration is not an expectation. As a surrogate for endogenous insulin production, C-peptide has been commonly used in. Although some clinical reviews and meta-analyses have indicated that umbilical cord MSC-based approaches may enhance C-peptide levels, minimize insulin requirement in selectively chosen patients, more extensive larger trails are required to determine the conclusiveness of most recent findings.
Current Clinical Evidence
5.1 Human Findings Are Fairly Limited But Promising
Phase I studies in Type 1 diabetes and this clinical pipelines include various other stem cell sources such as bone marrow-derived MSCs or umbilical cord (UC)-derived MSC. The increasing interest in using MSCs for this novel clinical application led to the initiation of a phase I/II randomized placebo-controlled clinical trial to assess autologous bone marrow-derived MSCs in newly diagnosed Type 1 diabetes; which is specifically geared towards both safety and efficacy.
5.2 Evidence of UC-MSC should be taken with caution
In a 2025 systematic review and meta-analysis with umbilical cord MSCs on Type 1 and Type 2 diabetes, the results were found to reduce HbA1c and insulin needs while raising C-peptide levels. Still, the authors stressed that multicenter clinical trials remain necessary to validate long-term efficacy and safety.
5.3 Research Is Not Yet Standardized
A significant limitation within this area is the heterogeneous nature of the study design. Studies differed regarding cell source, dose, timing, route of administration, disease stage at treatment initiation, patient age and residual beta-cell function as well as follow-up period. These differences mean that the results cannot be effortlessly generalized to all patients with Type 1 diabetes.
| Patient Factor | Why It Matters |
| Duration of Type 1 diabetes | Newly diagnosed patients may still have more residual beta-cell function |
| C-peptide level | Helps estimate remaining endogenous insulin production |
| HbA1c and glucose variability | Shows current metabolic control |
| Insulin requirement | May help track treatment response over time |
| Autoantibody status | Supports autoimmune diagnosis and disease characterization |
| History of diabetic complications | Important for safety and treatment planning |
| Infection screening | Essential before any cell-based intervention |
| Endocrinologist supervision | Necessary because insulin remains standard care |
FAQ Section
Can you cure Type 1 diabetes with UC-MSCs?
This would help persuade diabetic Type 1 in pairs that UC-MSCs cure T1D, you may say. Although UC-MSCs seem a promising and relevant tool for immune regulation, inflammation control, and beta-cell support especially when combined with immunosuppressants, further trials in larger cohorts are required before this technique will become an established treatment.
Can UC-MSCs replace insulin?
No; in general, after autoimmune beta-cell destruction most patients with type 1 diabetes do not produce sufficient endogenous insulin and require exogenous insulin administration. Never stop insulin without contacting a regional authority doctor.
Why are UC-MSCs being researched for Type 1 diabetes?
Since UC-MSCs may modulate immune activity, inflammation, paracrine signalling and the pancreatic microenvironment they are of interest for research. These mechanisms may be important for maintaining residual β-cell function.
What is required recommendations before UC-MSCs model selection?
Key indicators include fasting glucose, c-peptide, insulin substitution requirement, auto-antibody status, kidney and liver function tests as well as screening for infections and diabetes complications.
Stem cell therapy thailand for Type 1 diabetes?
Type 1 diabetes related programs are consultable in some regenerative medicine centers in Thailand. People need to select providers who utilize physician-led screening, clear cell sourcing, and careful safety standards leading to realistic expectations.
Figure 1: Proposed mechanisms of action of umbilical cord-derived mesenchymal stem cells in Type 1 diabetes. The figure summarizes four potential pathways currently under investigation: immune regulation, anti-inflammatory signaling, paracrine communication, and beta-cell functional support. UC-MSCs may influence Type 1 diabetes through the release of cytokines, growth factors, extracellular vesicles, and other bioactive molecules that could help modulate autoimmune activity, reduce inflammatory stress, support residual beta-cell function, and improve the pancreatic microenvironment. These mechanisms remain investigational, and current evidence should be interpreted cautiously. UC-MSC-based approaches should not be presented as a cure or as a replacement for insulin therapy.