Why does this topic keep drawing attention
When people search for stem cell therapy Thailand for Chronic Kidney Disease, they are usually not looking for abstract biology. They are looking for something that sounds more hopeful than “slow the decline and monitor closely.” That reaction makes sense. CKD means the kidneys are damaged or structurally abnormal and can no longer filter blood the way they should, and the most common adult causes are still diabetes and high blood pressure.
That is also why the keyword stem cell cases feels so compelling. Published case reports, small pilot studies, and early clinical trials can sound like the beginning of a breakthrough. Sometimes they really do show an important signal. But in kidney medicine, the harder question is whether those signals are strong, reproducible, and durable enough to change routine care. Right now, the answer is still more cautious than many websites suggest.
What Chronic Kidney Disease actually is
CKD is not one disease, but one long biologic process
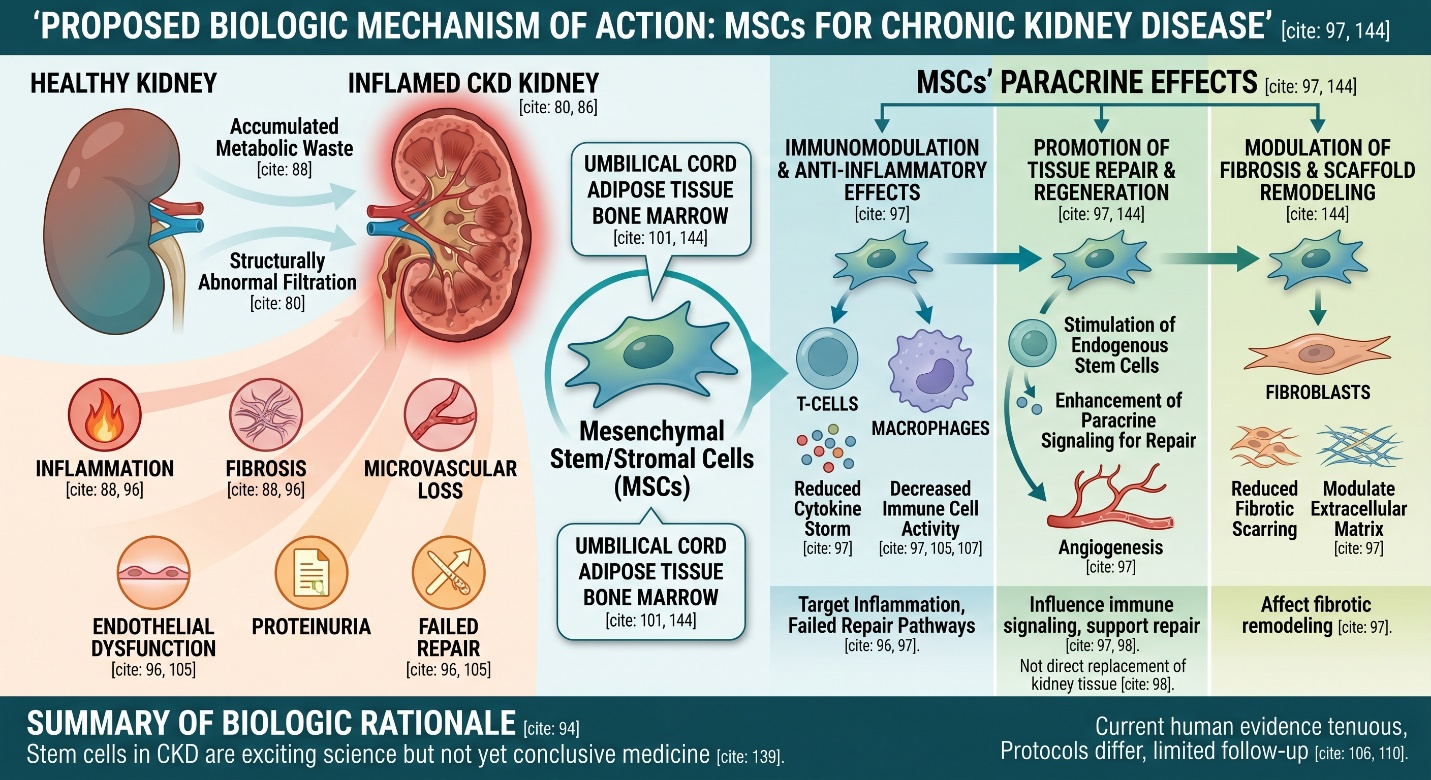
A lot of people talk about CKD as if it were a single condition. It is not. CKD is a broad category that includes different causes, different rates of progression, and different combinations of inflammation, fibrosis, metabolic stress, and vascular injury. NIDDK notes that many people do not know they have CKD until it is advanced, which is one reason the disease often feels silent until it starts taking a larger toll.
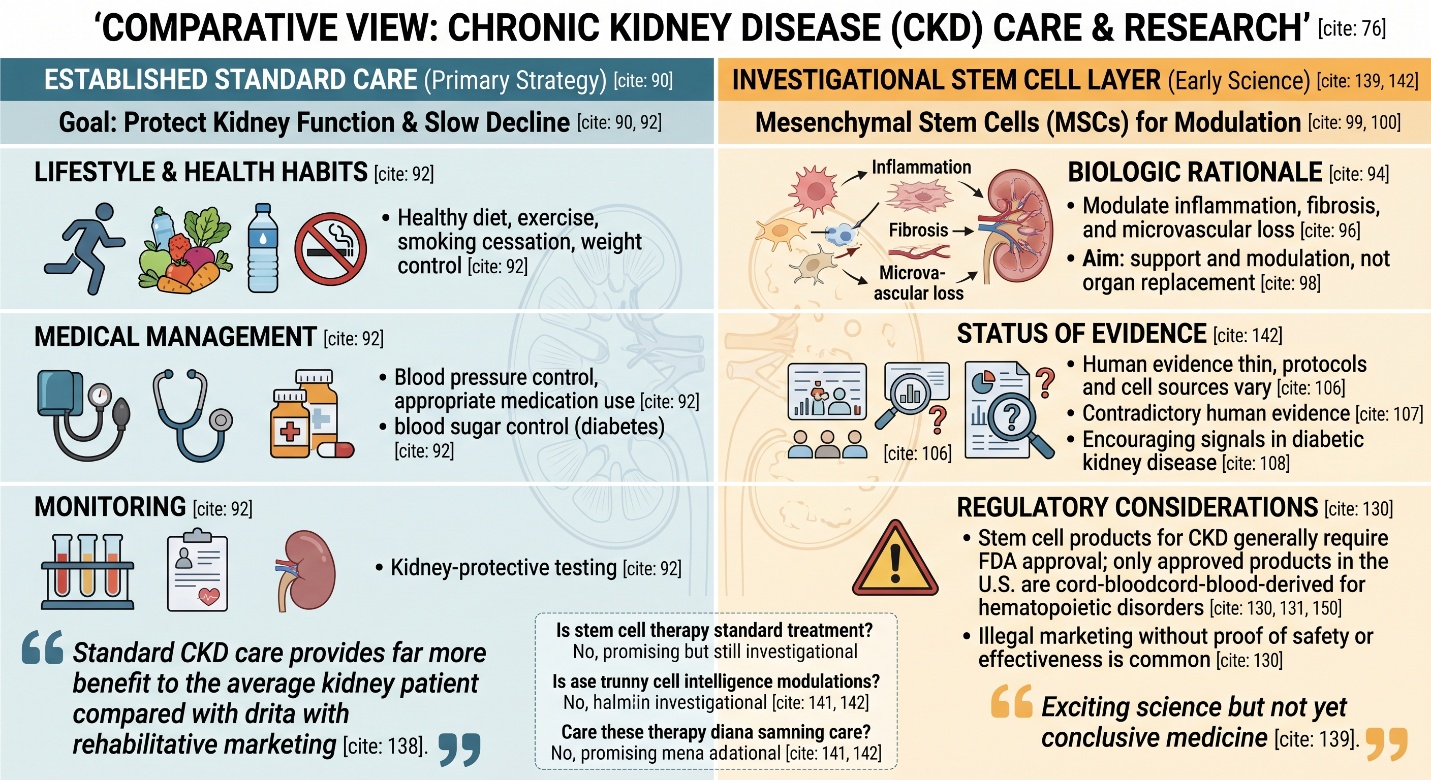
Standard care still has one main goal: protect kidney function for as long as possible
This point matters because regenerative language can sometimes make standard care sound unambitious, when it is actually central. NIDDK emphasizes blood pressure control, healthy habits, testing, and kidney-protective management, while KDIGO’s 2024 patient takeaways stress healthy diet, exercise, smoking cessation, weight control, and appropriate medication use as ways to slow CKD progression and reduce risks such as kidney failure, heart attack, stroke, and heart failure.
Why stem cells entered the CKD conversation at all
The biologic rationale is real
Stem cells became part of CKD research because chronic kidney disease is not just about loss of filtration. It is also about inflammation, fibrosis, endothelial dysfunction, microvascular loss, and failed repair. Recent reviews describe mesenchymal stem cells as attractive because they may influence immune signaling, paracrine repair pathways, angiogenesis, and fibrotic remodeling rather than simply “turning into new kidney tissue.” That is an important distinction. The field is interested in support and modulation of injury pathways, not only direct organ replacement.
Most clinical interest is still centered on MSC-type therapies
If you strip away the marketing language, the current human CKD literature is mostly about mesenchymal stem/stromal cells, not a wide range of interchangeable “stem cell treatments.” A 2025 global clinical landscape review reported growing worldwide interest and a rising number of CKD stem cell trials, largely focused on MSC-based strategies. That does not prove success, but it does show the field is active enough to deserve serious attention.
What published stem cell cases and early studies actually show
The signal is interesting, but it is still early
Here is where the article needs its heart. Good signals in kidney functions, inflammation and proteinuria or safety are frequently found when published stem cell cases and small clinical studies of CKD. But even recent reviews keep saying the same warning: human evidence is still thin, protocols differ, cell sources vary too and numerous studies are so small they cannot support wide-reaching clinical conclusions. A 2025 review specifically identified that, although in animal CKD models MSCs were the superior choice for improving inflammation and renal function, human evidence was contradictory.
Diabetic kidney disease has some of the better clinical data, but even that is not final
A systematic review and meta-analysis published in 2025, examining the effects of stem cell therapy for diabetic kidney disease with all outcomes as categorical variables noted that nearly every renal outcome analyzed by any one study showed improvement among only a portion of people assigned to treatment groups compared with placebo. Even in that more optimistic subset of the literature, familiar issues like small trial numbers, heterogeneity between protocols and limited follow-up beyond a year persist. Put another way, some of the best kidney data are still not standard-of-care routine evidence.
“Stem cell cases” should never be confused with proof
This part is easy to miss because case reports sound vivid. A case can be memorable, but it is still only one data point. In CKD, published case reports and small uncontrolled series can be useful for showing feasibility or generating hypotheses, but they do not establish that a therapy reliably works across a real patient population. That is exactly why systematic reviews and larger controlled studies matter more than testimonials or isolated dramatic stories.
Where “stem cell therapy Thailand” fits into the discussion
A search term is not a standard of evidence
The phrase stem cell therapy Thailand is a real-world search behavior, not a scientific category. People often use it because they are comparing access, cost, travel, and hope all at once. But the better question is not simply where treatment is offered. The better question is what exactly is being offered, under what regulatory framework, with what cell type, and supported by what published evidence. That is especially important in CKD, where many interventions are still investigational.
A credible clinic should sound more like nephrology than like branding
A comprehensive CKD clinic conversation should discuss cause of kidney disease, stage and prognosis, proteinuria (once for each new chronic or progressive condition), blood pressure management (if indicated to manage risk progression), diabetes control when relevant, cardiovascular risk factors plan(s) already in the picture: dialysis vs transplant planning. When stem cell therapy is involved, the clinic should be able to state precisely what that cell product comprises, how it will travel through the body (route), in case of adverse events if needed any form of monitoring follows up length and reason for patient as a reasonable candidate. The more the language smells of a detour around nephrology, so to speak, the least trustworthy it invariably becomes.
Why caution still matters
FDA is very clear about unapproved stem cell products
The FDA states that stem cell products are regulated and generally require FDA approval, and it warns that many regenerative medicine products are illegally marketed without adequate proof of safety or effectiveness. The same FDA consumer alert explains that, in the U.S., the only currently approved stem cell products are blood-forming stem cells from cord blood for disorders of the hematopoietic system, not for kidney disease. That does not invalidate CKD research. It simply means availability and scientific legitimacy are not the same thing.
The most honest conclusion
The truth is far more useful than the sales pitch. Stem cell therapy Thailand does not live in the world of dream; Chronic Kidney Disease lives there, and so should stem cell research. The biologic rationale exists, the trial numbers are increasing for autologous and allogeneic approaches, and while most published stem cell cases have been small there is sufficient signal in several early clinical studies to continue investigation. Sadly, though a lot of momentum is still in its infancy and the human evidence remains tenuous at best standard CKD care provides far more benefit to the average kidney patient compared with rehabilitative marketing. Hence, we conclude our study as follows: stem cell therapy in CKD is exciting science but not yet conclusive medicine.
Frequently Asked Questions
1) Is stem cell therapy already a standard treatment for Chronic Kidney Disease?
No. Current reviews describe stem cell therapy for CKD as promising but still investigational, with human evidence still considered inconclusive or incomplete.
2) What type of stem cells are most often studied in CKD?
Most current CKD clinical research focuses on mesenchymal stem/stromal cells, usually because of their immunomodulatory and paracrine effects rather than direct kidney replacement.
3) Do published stem cell cases prove the treatment works?
Not by themselves. Case reports and small pilot studies can be useful for showing feasibility or early signals, but they do not provide the same level of evidence as larger controlled trials or strong meta-analyses.
4) Is there any subset of kidney disease where the data look more encouraging?
Yes. Diabetic kidney disease currently has some of the more encouraging published clinical data, including a 2025 meta-analysis reporting improved renal outcomes in analyzed studies, though long-term certainty is still lacking.
5) Are stem cell products approved for CKD treatment?
The FDA says stem cell products generally require approval, and the only currently approved stem cell products in the U.S. are cord-blood-derived blood-forming stem cells for hematopoietic disorders, not for kidney disease.