Why does this topic need a more careful conversation?
Anyone searching for the best stem cell clinic for Psoriasis is usually not looking for abstract immunology. They are looking for relief from a disease that can itch, crack, flare, affect sleep, change how skin looks, and wear people down over time. Psoriasis is a chronic, inflammatory, multisystem disease, and standard medical sources are still very clear on one central point: it can be managed, but it is not considered cured.
That is exactly why stem cell language becomes so attractive. When a disease is chronic and frustrating, “regenerative” ideas sound emotionally larger than creams, tablets, or injections. But in psoriasis, the gap between biologic promise and established treatment is still real. The current evidence is interesting enough to discuss seriously, but not mature enough to blur into clinic-style certainty.
What psoriasis treatment already looks like without stem cells
Standard care is broader and more effective than many people assume
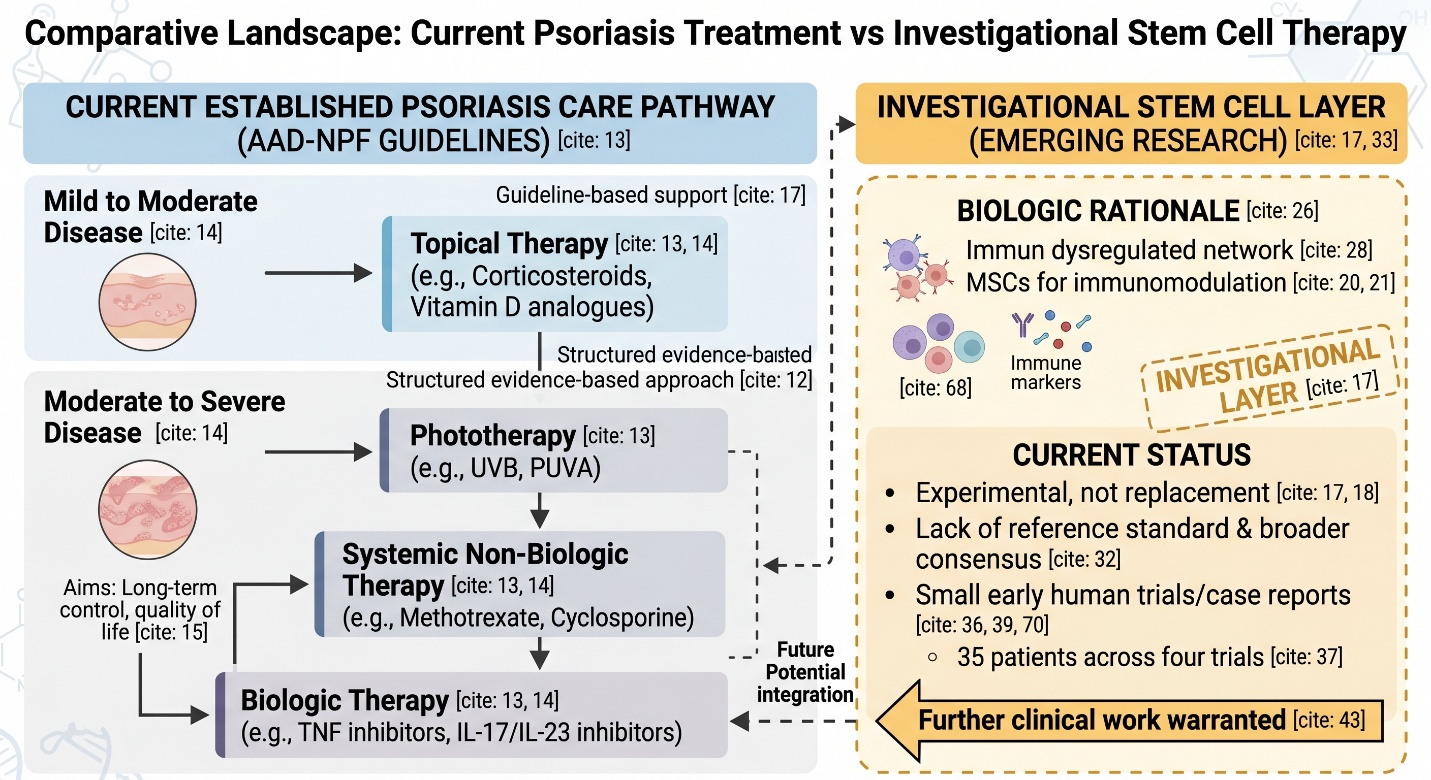
Before stem cells even enter the discussion, it helps to remember that psoriasis already has a structured evidence-based treatment pathway. The AAD–NPF guideline set covers topical therapy, phototherapy, systemic non-biologic therapy, biologics, and management of comorbidities. The same guidance notes that many patients with mild to moderate disease can be managed with topical agents, while moderate to severe disease may require systemic non-biologic drugs or biologics. NICE likewise frames psoriasis care around long-term disease control and quality of life.
That matters because a lot of “best stem cell clinic” language quietly suggests that stem cells sit above conventional psoriasis care. Right now, the evidence does not support that framing. In real dermatology, stem cells are still an investigational layer, not a replacement for the therapies that already have guideline-level support.
What do “specialized stem cells” usually mean in psoriasis
In practice, the main specialized stem cells under study are MSCs
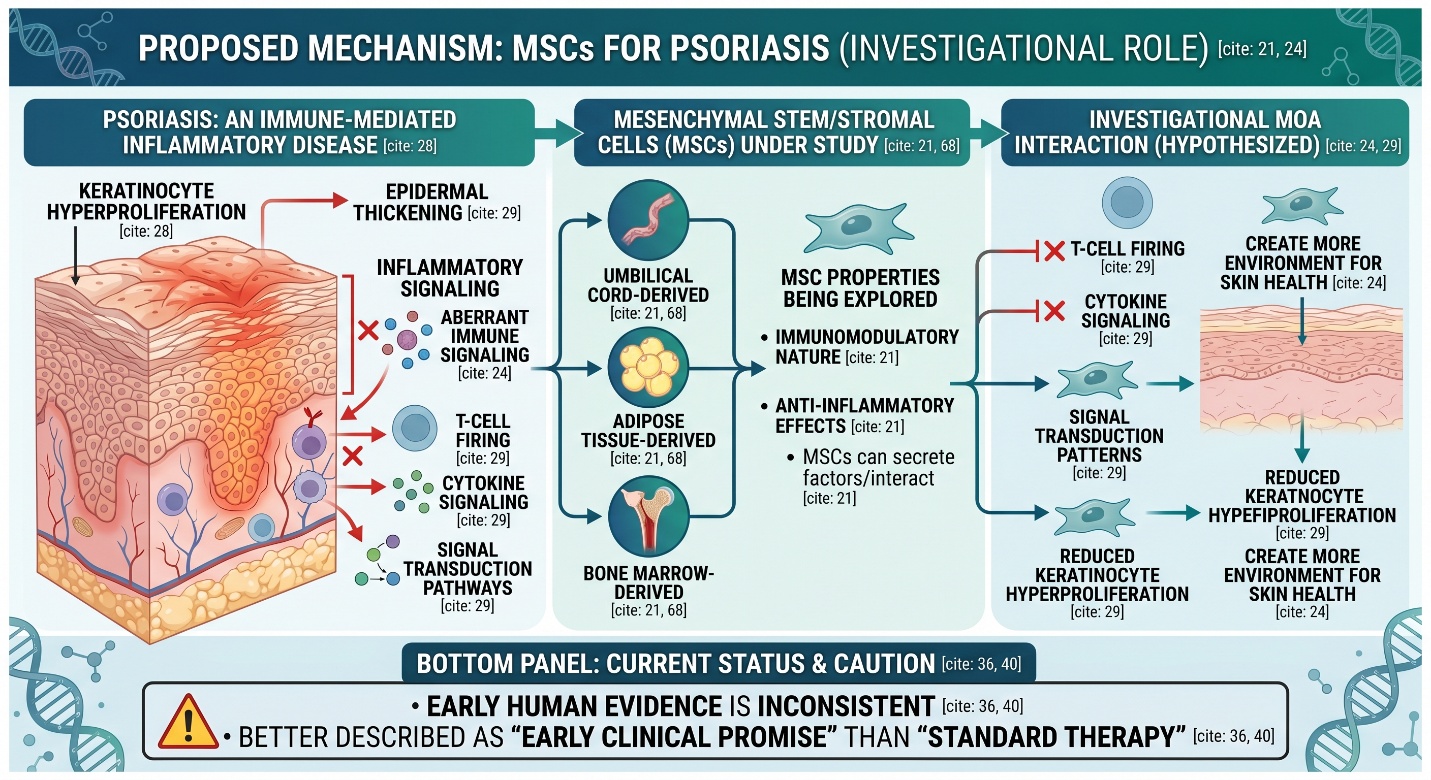
This even sounds broad, but within psoriasis often indicates MSCs (mesenchymal stem/stromal cells) rather than a diverse variety of unrelated cell types. MSCs from umbilical cord, adipose tissue, and bone marrow were described as main cell sources currently being explored in recent 2025 reviews because of their immunomodulatory nature and anti-inflammatory effects.
This is a relevant distinction because psoriasis, like certain chronic-reclusive diseases, is not actually about missing cells. An immune-mediated inflammatory disease. Hence, the specific interest in stem cells is not that great a deal in replacing skin by one cell here and there. Ultimately, the question comes down to whether MSCs can modulate inflammatory pathways and aberrant immune signaling as well as create a more conducive environment for skin health.
Why did stem cells become part of the psoriasis conversation at all
The biologic rationale is real
There is a Tangible scientific purpose for why this industry exists. Psoriasis is a chronic immune-mediated disease characterized by keratinocyte hyperproliferation and inflammatory signaling, with much recent psoriasis research still describing it in terms of an immuodysregulated network. Synchronization MSCs are studied because veteran T-cell firing, cytokinacyclic signal transduction pathways ego pattern epidermal thickening advisable more wide to the inflammatory behavior significant with standard psoriasis molecular biology.
But the rationale is not the same as proof
This is where articles often become too eager. A plausible mechanism can justify a research program without proving a treatment is ready for routine care. The 2025 expert consensus on stem cells in psoriasis says directly that the field still lacks a reference standard and broader consensus for clinical application, even while interest and published work are growing. That is a strong sign of promise, but also a strong sign of incompleteness.
What the actual clinical evidence shows
Early human data are small, but not empty
This is probably the best summary of current human evidence: small early trials and case-based clinical reports suggest that MSC-based therapy may be beneficial for some subjects with psoriasis, particularly cases that are severe or difficult to control. There was growing evidence from in vitro, animal, and clinical studies noted as of 2025, while a landmark paper published in Frontiers reported that no serious adverse events were found across four different clinical trials involving 35 patients who received the treatment alongside standard care, with some degree of positive effect shown by its use.
That sounds encouraging, and it is. But it is also important to notice how small that evidence base still is. Four trials and a few dozen patients are enough to justify continued research, but not enough to settle the “best stem cell clinic” question in the way clinic marketing often implies. In psoriasis, the evidence is still better described as early clinical promise than as standard therapy.
Preclinical evidence is stronger than clinical evidence
Another important layer is that preclinical evidence currently looks more robust than human evidence. A 2025 meta-analysis in animal models found that MSC transplantation improved psoriasis-like disease measures such as PASI-style outcomes and epidermal thickness, and concluded that further clinical work is warranted. That is helpful scientifically, but animal-model success is not the same thing as mature patient-level validation.
What “best stem cell clinic” should really mean
The best clinic should sound more cautious than promotional
This is probably the most practical point in the whole article. If someone is seriously comparing the best stem cell clinic for psoriasis, the best sign is not bold language. It is a restraint. A credible clinic should first acknowledge that psoriasis already has established guideline-based therapies, and second explain clearly that stem cell treatment for psoriasis remains investigational. If a clinic sounds more confident than the literature does, that is not a strength.
Regulation matters more than branding
This would be especially pertinent to the FDA’s consumer alert. The FDA states here that stem cell products typically need approval from the agency, and notes also that the only approved U.S. stem cell products are cord-blood-derived blood-forming cells for hematopoietic disorders, and there is no such thing as an FDA-approved exosome product yet! The FDA also says patients could be misled by regenerative products marketed illegally and cautions that unapproved human cells or tissues may carry serious hazards, including reports of serious injuries and deaths.
So in practice, the “best” clinic is not the one that makes psoriasis sound curable by stem cells. It is the one that behaves like real medicine: careful diagnosis, transparent discussion of evidence, clear explanation of risks, and honesty about what is still experimental.
The most honest conclusion
The pitch is better than the truth. Given the immunology of psoriasis, specialized stem cells—mostly umbilical cord-, adipose tissue- or bone marrow-derived MSCs are an important focus area in psoriatic research. While early clinical reports are reassuring, the safety signal thus far may be better than many presume it to be. But the space is still small, heterogeneous, and investigational.
The most truthful, honest answer right now, if someone were to ask you what is the best stem cell clinic for psoriasis is, would not be a name brand. You are trained to answer this: the real best clinic is that one telling you stem cells for psoriasis is hopeful science, and not established standard care.
Frequently Asked Questions
1) Is stem cell therapy already an approved treatment for psoriasis?
There is no FDA-approved stem cell product specifically approved for psoriasis. FDA says stem cell products generally require approval, and current approved stem cell products in the U.S. are limited to blood-forming cells from umbilical cord blood for hematopoietic disorders.
2) What are the “specialized stem cells” usually studied in psoriasis?
Most published psoriasis work focuses on mesenchymal stem/stromal cells (MSCs), especially from umbilical cord, adipose tissue, and bone marrow.
3) Have clinical trials in psoriasis shown anything positive?
Yes, but only at an early level. Recent reviews report small clinical studies with encouraging signals, and one 2025 review noted that across four clinical trials in 35 patients, no serious adverse events were reported.
4) Why are stem cells being studied for psoriasis in the first place?
Because psoriasis is an immune-mediated inflammatory disease, and MSCs may help modulate inflammatory pathways, immune-cell activity, and epidermal overgrowth that are central to the disease.
5) What should I look for if a clinic offers stem cells for psoriasis?
Look for a clinic that clearly explains the treatment is investigational, discusses standard psoriasis therapies first, provides transparent evidence and safety information, and does not market the therapy as though it were already routine or guaranteed. FDA and ISSCR both advise patients to be cautious and to ask detailed questions about evidence, oversight, and risks.