Molecular Bio-Reclamation of the Joint Environment Using UC-MSCs for Knee Cartilage Damage Stem Cell Therapy in Thailand
Biological re-engineering of the joint environment has superseded traditional mechanical stabilization as the primary objective in modern orthopedic science. For patients grappling with the erosion of the articular surface, the emergence of UC-MSCs (Umbilical Cord-derived Mesenchymal Stem Cells) offers a sophisticated alternative to invasive resurfacing surgeries. While conventional treatments often focus on temporary symptomatic masking through corticosteroids or lubricants, the application of knee cartilage damage stem cell therapy addresses the fundamental cellular depletion at the core of the injury. Within the high-tier regenerative facilities found in Thailand, this clinical approach utilizes the innate potency of neonatal cells to trigger a deep-tissue repair sequence that standard orthopedic interventions simply cannot initiate.
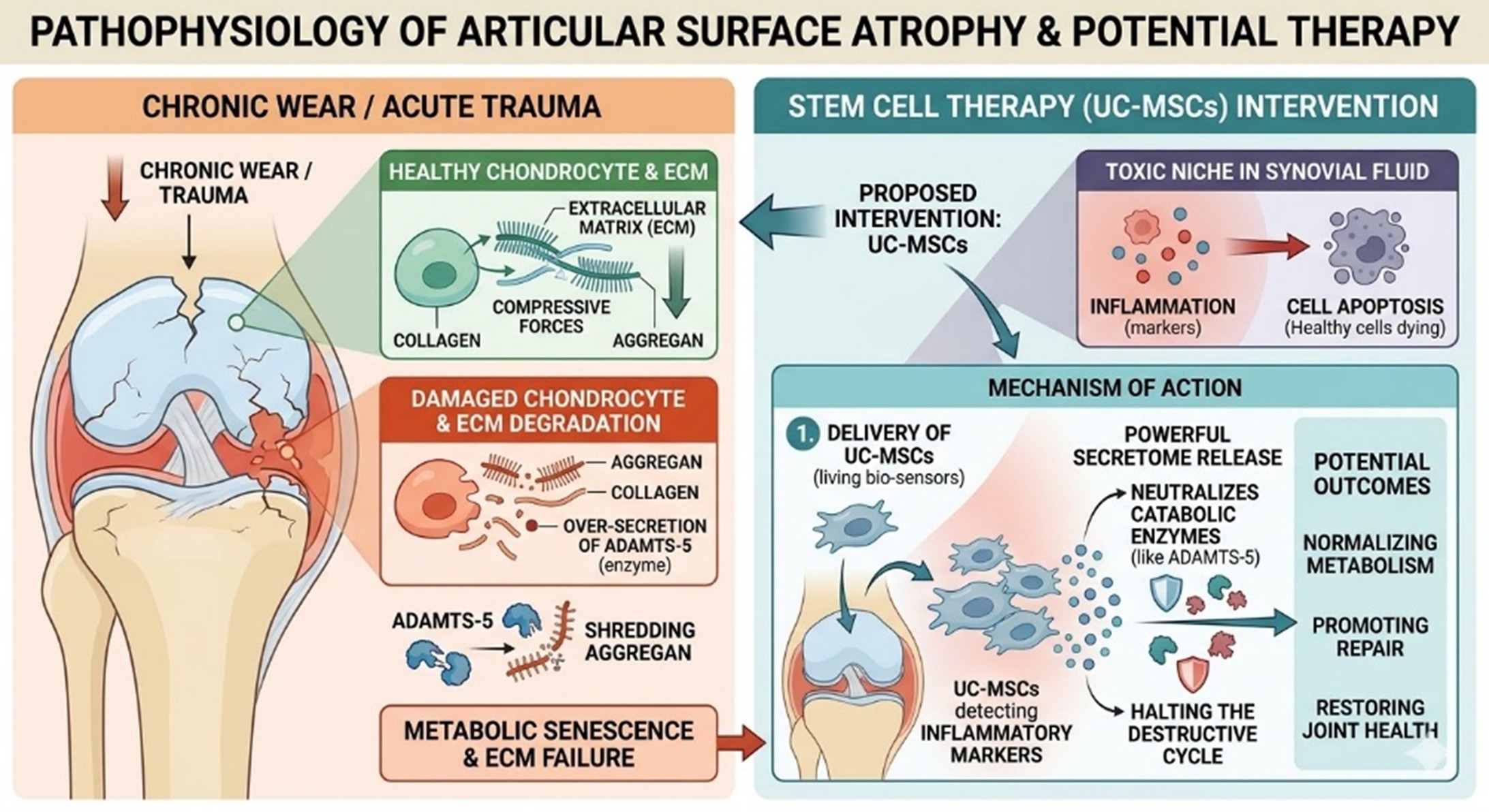
1.The Pathophysiology of Articular Surface Atrophy
Articular cartilage is a unique, avascular tissue characterized by its lack of a direct blood supply, which renders it notoriously incapable of self-repair. In the event of chronic wear or acute trauma, resident chondrocytes enter a state of metabolic senescence, failing to maintain the integrity of the extracellular matrix (ECM). This degradation is primarily driven by the over-secretion of ADAMTS-5, an enzyme that targets and shreds aggrecan—the critical proteoglycan responsible for the cartilage’s ability to withstand compressive forces.
When patients pursue knee cartilage damage stem cell therapy, they are actively addressing this enzymatic imbalance. The persistent inflammation within the synovial fluid creates a toxic niche where healthy cells are progressively forced into apoptosis. Introducing UC-MSCs into this environment provides an immediate biological counter-response. These cells function as living bio-sensors; once delivered into the intra-articular space, they detect inflammatory markers and release a powerful secretome designed to neutralize catabolic enzymes, halting the destructive cycle that defines osteoarthritis and focal cartilage defects.
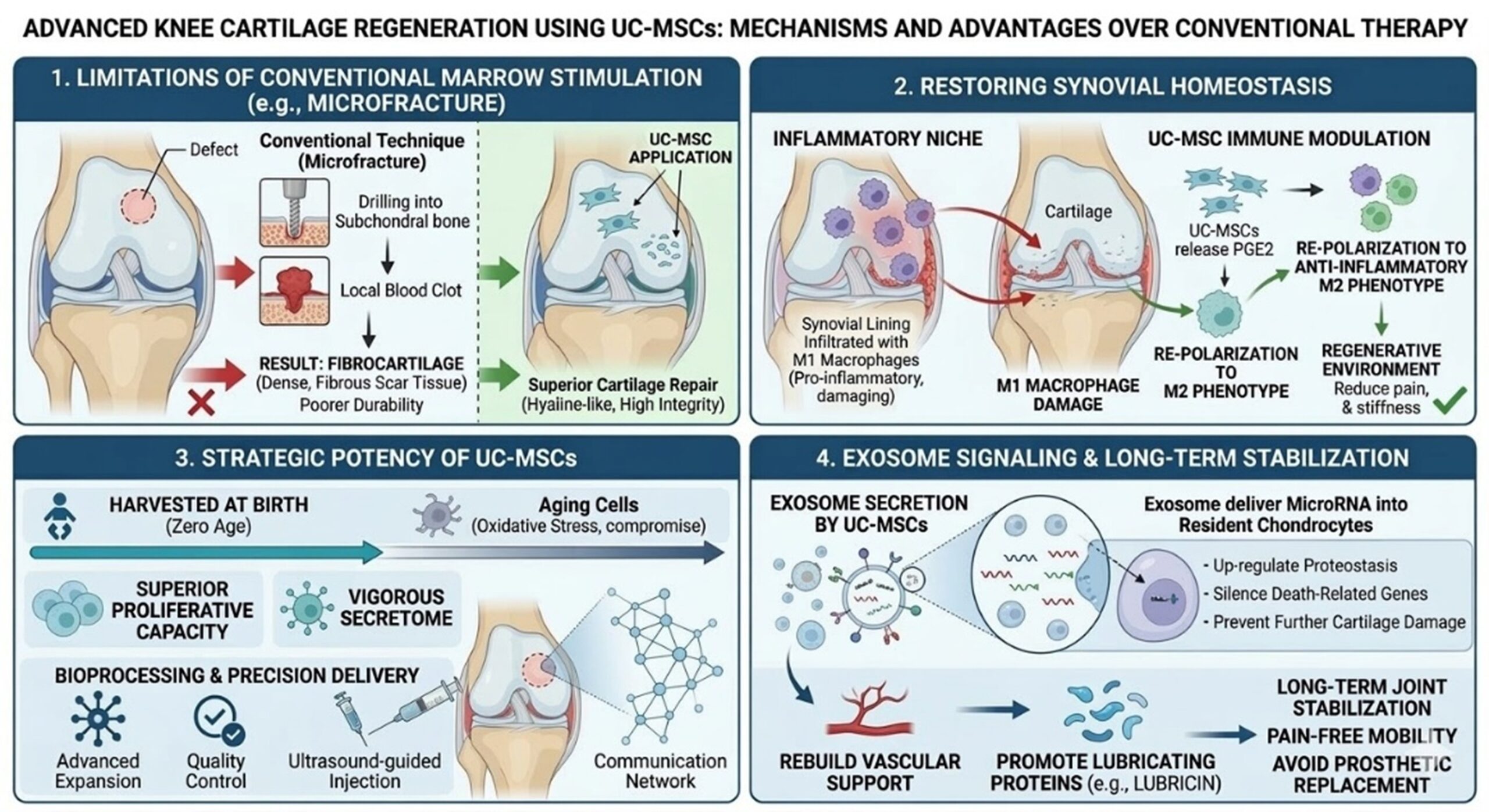
- Limitations of Conventional Marrow Stimulation
A primary pillar of traditional orthopedics is the reliance on mechanical marrow stimulation, such as microfracture, to treat knee cartilage damage. Unlike advanced stem cell therapy, these conventional techniques depend on creating subchondral bone perforations to induce a localized blood clot. While this initiates a healing response, the resulting tissue often lacks the structural integrity of natural cartilage. Through this process, the body recruits local cells compromised by aging, limiting their ability to differentiate into high-quality repair cells. Consequently, these cells primarily produce a dense, fibrous scar tissue known as fibrocartilage. To overcome these failures, the application of UC-MSCs has emerged as a superior alternative, not only by providing fresh cells but also by actively reprogramming the joint’s internal environment.
- Restoring Synovial Homeostasis through Macrophage Reprogramming
Beyond building new tissue, the success of UC-MSCs lies in their ability to stabilize the synovial membrane, a vital factor for long-term joint survival. While conventional surgery ignores the inflammatory toxic niche, this advanced stem cell therapy addresses the knee’s synovial lining, which is often infiltrated with M1-polarized macrophages. These pro-inflammatory cells contribute to further knee cartilage damage, ensuring that even surgical repairs eventually fail. By releasing mediators such as PGE2, UC-MSCs signal the joint’s immune system to re-polarize these cells into the anti-inflammatory M2 phenotype. This immune-modulatory shift explains why patients report reduced pain and stiffness even before the physical cartilage has fully rebuilt, effectively transforming a degradative joint into a regenerative one.
- Strategic Potency of UC-MSCs in the Clinical Context
The choice of UC-MSCs is scientifically prioritized because their superior proliferative capacity is essential for maintaining the homeostasis required in stem cell therapy. Harvested at birth, these UC-MSCs have not been exposed to the oxidative stress that affects a patient’s own bone marrow. This provides a more vigorous secretome, which is essential for overcoming the advanced age of many patients seeking relief from knee cartilage damage. Furthermore, sophisticated bioprocessing ensures that these cells maintain their stemness through advanced expansion. This quality control, combined with precise, ultrasound-guided delivery, ensures that the UC-MSCs reach the areas of highest need, initiating a communication network that lasts far beyond the initial injection. This high-level quality control ensures that UC-MSCs reach the areas of highest need, initiating a communication network that lasts far beyond the initial injection through the release of signaling vesicles.
- Exosome Signaling and Long-Term Matrix Stabilization
The final layer of this deep-dive mechanism involves the secretion of exosomes—nano-vesicles that act as the long-term messengers of the UC-MSCs. Even as the injected cells complete their primary tasks, these exosomes deliver microRNA directly into resident chondrocytes. In the context of stem cell therapy, these instructions silence death-related genes and upregulate proteostasis to prevent further knee cartilage damage. By rebuilding vascular support and promoting lubricating proteins like lubricin, this molecular communication ensures that the joint remains stabilized for the long term. This holistic restoration offers a path toward pain-free mobility without the permanence of a prosthetic replacement, marking a definitive evolution in orthopedic science.
In conclusion, Application of UC-MSCs represents a fundamental advancement in the management of articular health. By targeting the molecular drivers of cartilage degradation and synovial inflammation, this technology provides a grounded, regenerative solution through knee cartilage damage stem cell therapy. The clinical excellence and bioprocessing sophistication available in Thailand ensure that UC-MSCs are utilized at their maximum metabolic potential. Through the restoration of chondrogenic signaling and the modulation of the joint’s immune environment, UC-MSCs provide a scientifically backed hope for long-term mobility and the preservation of natural joint function.

