UC-MSCs: A Transformative Frontier in Neuroregenerative Therapy for Parkinson’s Disease
Parkinson’s Disease stands as a formidable neurological challenge characterized by a relentless decline in motor function that affects millions worldwide. While conventional pharmacological interventions have long served as the cornerstone of clinical management, they offer only a palliative reprieve by alleviating symptoms without fundamentally altering the progressive trajectory of the disease. The emergence of Umbilical Cord-derived Mesenchymal Stem Cells (UC-MSCs) marks a pivotal shift in this therapeutic landscape. By moving beyond simple neurochemical replacement toward a holistic restoration of the neural microenvironment, UC-MSCs offer a sophisticated strategy to reclaim the physiological balance essential for sustainable motor control.
- The Causes and Severe Impact of Parkinson’s Disease
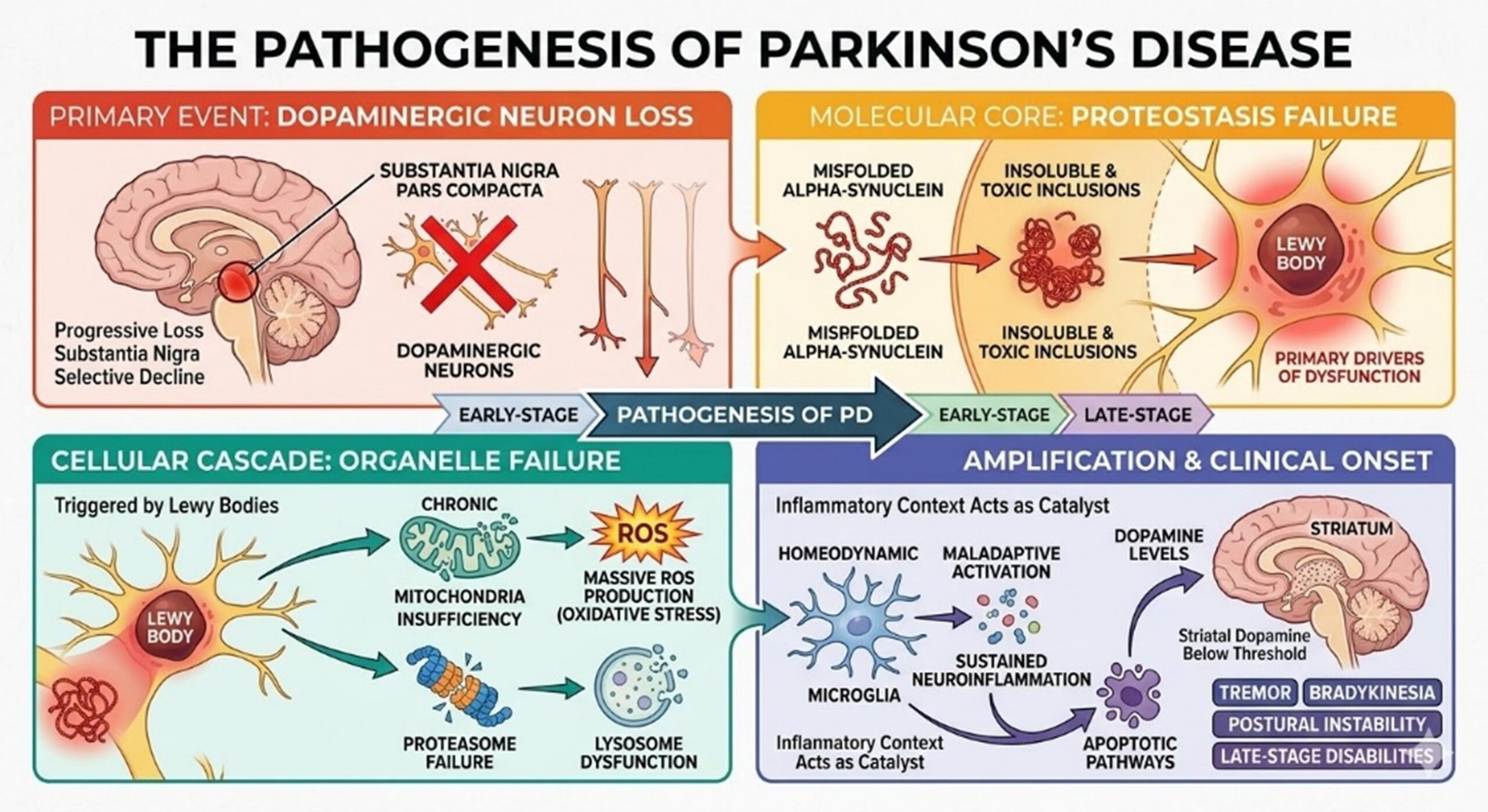
The pathogenesis of Parkinson’s Disease is rooted in the selective and progressive loss of dopaminergic neurons within the Substantia Nigra Pars Compacta. At the molecular core of this decay lies a pathogenic proteostasis failure involving the accumulation of misfolded alpha-synuclein proteins. These proteins aggregate into insoluble and toxic inclusions known as Lewy Bodies, which serve as the primary drivers of intracellular dysfunction in Parkinson’s Disease. Once these aggregates form, they trigger a cascade of organelle failure, most notably within the mitochondria.
This results in a state of chronic bioenergetic insufficiency along with the massive production of reactive oxygen species known as oxidative stress. Compounding this cellular crisis in Parkinson’s Disease is the maladaptive activation of microglia, which precipitates sustained neuroinflammation. This Inflammatory context acts as a catalyst that accelerates neuronal death through apoptotic pathways. As dopamine levels in the striatum plummet below the requisite threshold, the hallmark clinical features of tremor, rigidity, and bradykinesia emerge, eventually culminating in profound postural instability and the complex disabilities associated with late-stage disease. In this context, UC-MSCs are being studied for their ability to interrupt this cycle of decay. (Alushaj, E., et al., 2024.)
- Limitations of Current Clinical Modalities
Traditional therapeutic strategies remain for Parkinson’s Disease centered on dopamine replacement therapy, predominantly through Levodopa, or the surgical modulation of neural circuits via Deep Brain Stimulation. While these tools are invaluable for managing of the Parkinson’s Disease but they are inherently symptomatic in nature. Over time, the therapeutic window narrows, leading to the wearing-off phenomenon and the development of levodopa-induced dyskinesia, which often becomes as debilitating as the disease itself. The absence of a disease-modifying effect in these standard treatments underscores the necessity for biological interventions that can actively preserve and repair neural architecture. The total absence of a disease-modifying effect in these standard treatments underscores the urgent necessity for biological interventions, such as UC-MSCs, that can actively preserve and repair the delicate neural architecture. (Jankovic, J., 2000)
- The Multifaceted Therapeutic Potential of UC-MSCs in Parkinson’s Disease
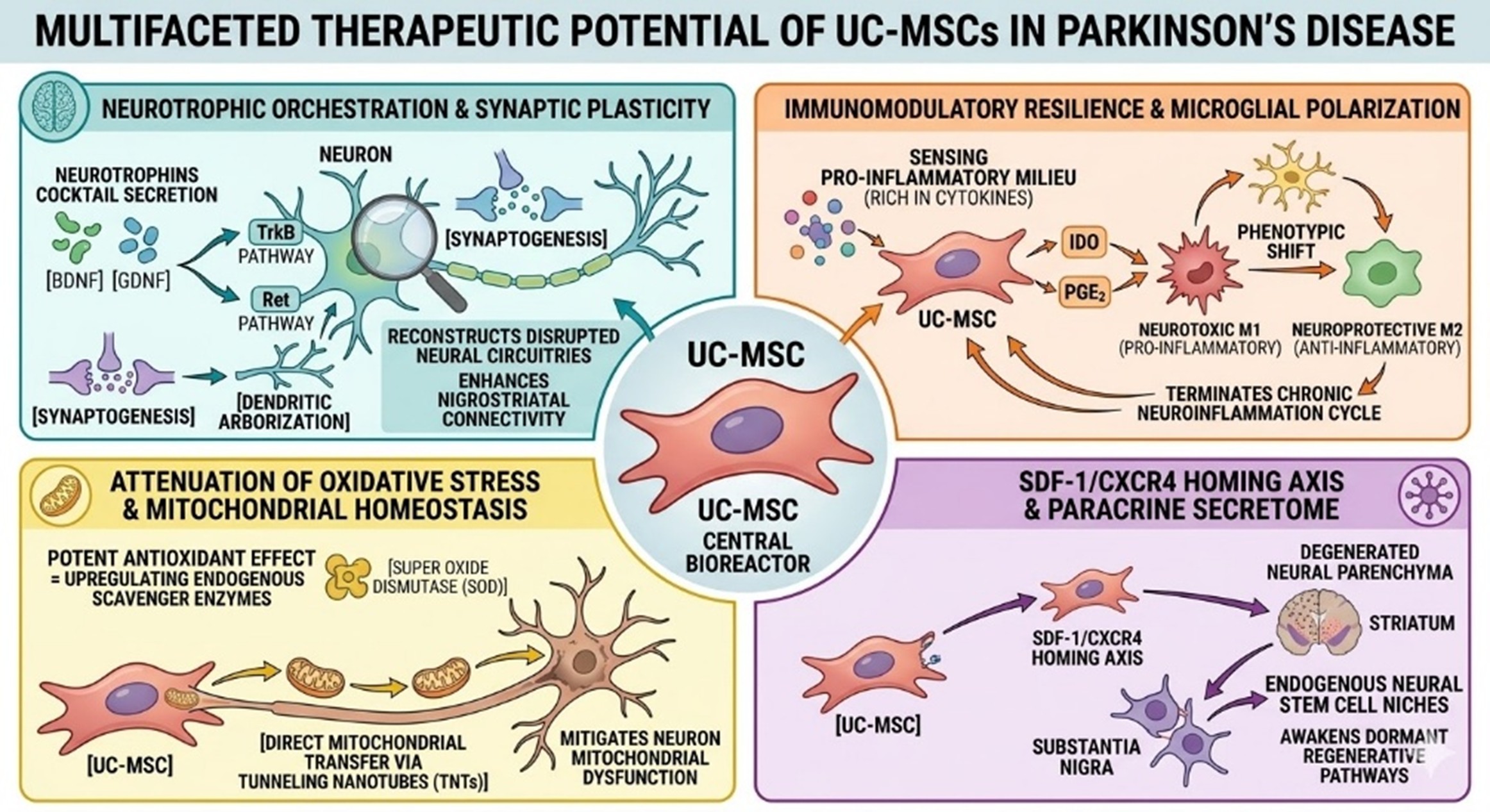
The clinical superiority of UC-MSCs is predicated upon their robust proliferative kinetics and a distinct profile of biological youthfulness characterized by superior immunomodulatory plasticity compared to adult-derived counterparts. Their therapeutic impact on Parkinson’s Disease is a sophisticated physiological orchestration involving:
- Neurotrophic Orchestration and Synaptic Plasticity:
UC-MSCs function as potent bioreactors secreting a broad spectrum of neurotrophins cocktails including Brain-derived Neurotrophic Factor and Glial Cell-derived Neurotrophic Factor. These secretomes activate the TrkB and Ret receptor tyrosine kinase pathways which are critical for the metabolic maintenance of dopaminergic neurons in Parkinson’s Disease. This facilitation of synaptogenesis and dendritic arborization effectively reconstructs disrupted neural circuitries and enhances the functional connectivity of the nigrostriatal pathway.
- Immunomodulatory Resilience and Microglial Polarization:
These cells possess a profound capacity for adaptive immunomodulation. Upon sensing a pro-inflammatory milieu rich in cytokines, UC-MSCs respond by secreting Indoleamine 2,3-dioxygenase and Prostaglandin E2. These mediators induce a phenotypic shift in resident microglia from the neurotoxic M1 (pro-inflammatory) state to the neuroprotective M2 (anti-inflammatory) phenotype. This reconfiguration of the innate immune response effectively terminates the cycle of chronic neuroinflammation typical of Parkinson’s Disease. (Cui, et al,. 2025)
- Attenuation of Oxidative Stress and Mitochondrial Homeostasis:
UC-MSCs exert a potent antioxidant effect by upregulating endogenous scavenger enzymes like Superoxide Dismutase. Beyond chemical neutralization, UC-MSCs can engage in direct mitochondrial transfer via tunneling nanotubes. By donating healthy mitochondria to bioenergetically compromised neurons in Parkinson’s Disease, UC-MSCs directly mitigate mitochondrial dysfunction.
- The SDF-1/CXCR4 Homing Axis and Paracrine Secretome:
UC-MSCs is governed by the SDF-1/CXCR4 signaling axis, which guides these cells toward the degenerated neural parenchyma associated with Parkinson’s Disease. Once localized, UC-MSCs utilize paracrine signaling to communicate with the host’s endogenous neural stem cell niches, awakening dormant regenerative pathways. (Wang, F., et al,.2010).
- UC-MSCs Research and Thailand’s Strategic Position in the Global Future
Thailand has cultivated a sophisticated ecosystem for regenerative medicine characterized by stringent adherence to international Good Manufacturing Practice standards in UC-MSCs processing. Current clinical research is increasingly focused on the next generation of cell-free therapy, specifically Exosomes.
These UC-MSCs-derived extracellular vesicles encapsulate the therapeutic essence of the stem cell by carrying proteins and RNA directly to the target site. This process bypasses the immunological and logistical complexities associated with live cell transplantation in Parkinson’s Disease. This shift toward precision nanomedicine aims to establish a new gold standard for adjunctive therapy in Thailand, ensuring that neuroprotective interventions are both accessible and highly effective.
In Conclusion, the clinical integration of UC-MSCs represents a quantum leap in the management of Parkinson’s Disease that transcends the binary of symptom versus cure. By addressing the underlying pathophysiology through immune modulation, oxidative stress reduction, and trophic support, UC-MSCs offer a comprehensive framework for turning the tide from neurodegeneration toward neurorestoration. This biological revolution holds the promise of not only enhancing longevity but also restoring the fundamental freedom of movement and dignity to those living with the disease.

