A stroke is not only a medical emergency at the moment. *It is often the beginning of a much longer story. The National Institute of Neurological Disorders and Stroke says stroke happens when blood flow to the brain is blocked or a blood vessel bursts, and that it can cause permanent brain damage within minutes or hours. It is also a leading cause of adult disability, with common long-term effects including weakness or paralysis, speech problems, cognitive changes, emotional problems, and pain.
That is exactly why search interest around stem cell therapy in Thailand and stroke keeps growing. Many patients do not start by looking for experimental options. They start with survival, rehabilitation, and the hope of getting back some part of the life they had before. Recovery is fastest in the initial period after a stroke, but organizations focused on stroke recovery note that improvement can continue for months and even years. For some patients, it may take longer, so we want to promote the search for new methods, including cell-based therapy.
If you read the papers instead of the marketing, the first thing is that cell therapy experience in stroke is still being built, not finished. What makes stem cells different from other cells is their ability to renew and differentiate; for stroke researchers, it is not that they erase brain damage with miraculous effects, but rather that they may assist repair along several routes. Welfare, look at the cloud that describes which might play a part in its role in neuroprotection, immunomodulation, and angiogenesis, which brings a particular idea to back up the patient self-repair environment within the brain after ischemia-induced injury.
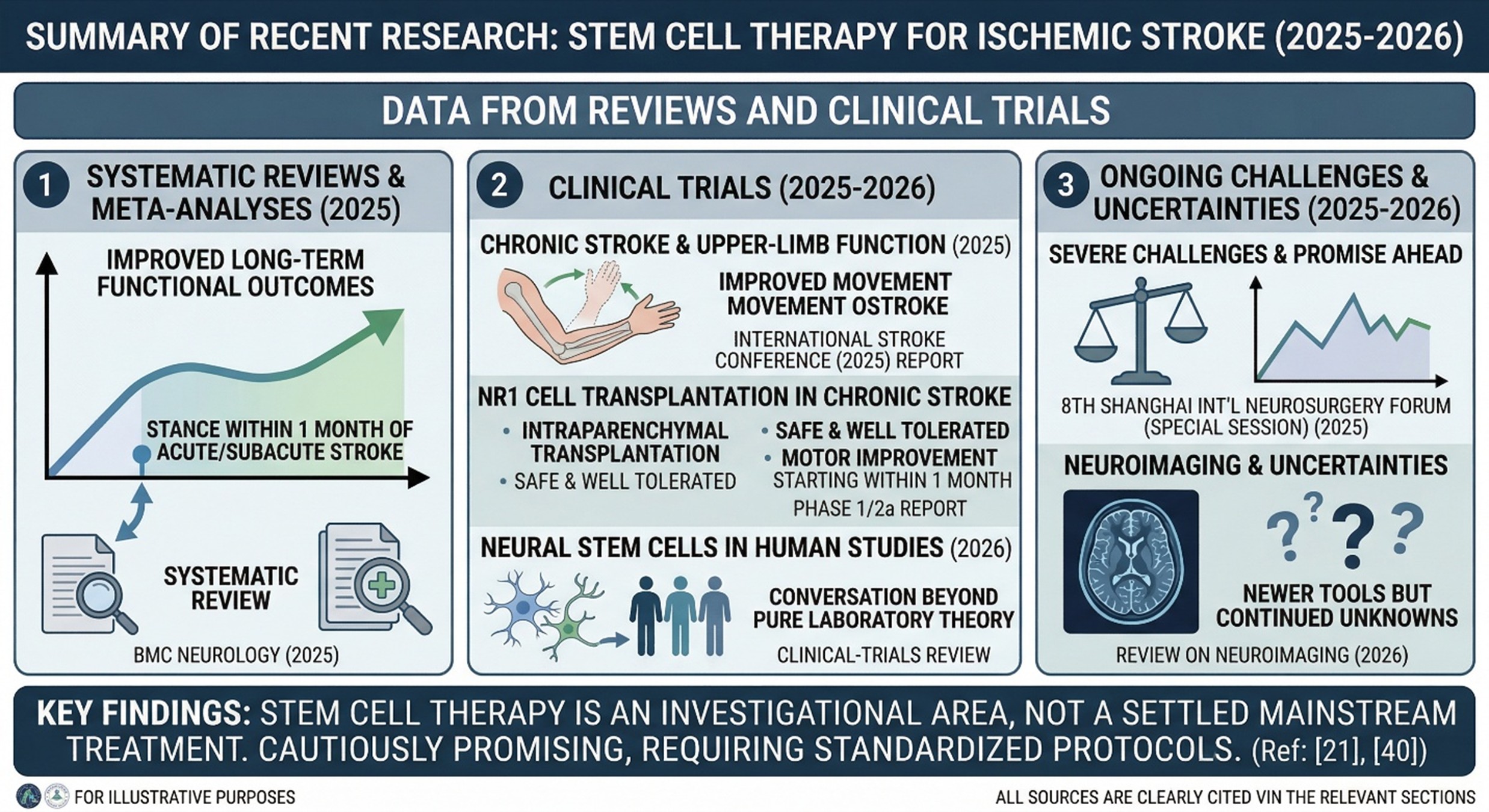
The recent literature is actually more interesting than many people expect. A 2025 systematic review and network meta-analysis in BMC Neurology evaluated stem cell therapy for ischemic stroke, while another 2025 review reported that stem cell therapy within 1 month of acute or subacute ischemic stroke appeared safe and was associated with significantly improved long-term functional outcomes. A 2026 clinical-trials review also shows that the field now includes multiple human studies using neural stem cells and related cell types, which means the conversation has clearly moved beyond pure laboratory theory.
At the same time, the papers do not tell a miracle story. They tell a mixed one. A 2025 review of stem cell therapies for stroke, featured in participant presentations at the 8th Shanghai International Neurosurgery Forum (Special Session), suggested that there is great promise ahead but also severe or ongoing challenges. A 2026 review on neuroimaging and stroke stem cell therapy pointed out much of what newer tools in the field value, however, it also highlighted many uncertainties.
In plain language, the direction of travel is encouraging. Nevertheless, a universally accepted protocol for all types of strokes and their time windows, as well as for every patient profile, has not been agreed upon. So far, carrying out research on cell transplant therapy has been quite productive. For example, a report from the 2025 International Stroke Conference, from a session on intracerebral neural stem cell transplantation in patients with chronic stroke, found that the procedure was safe and resulted in improved upper-limb function. As a first-in-human experience, an accompanying phase 1/2a report on NR1 cells in chronic stroke in the same year showed that intraparenchymal transplantation was safe and well tolerated, with motor improvement starting within 1 month, according to its results. These are exactly the kinds of findings that make stem cell therapy Thailand a compelling search phrase for people looking beyond standard rehabilitation.
Still, the most honest way to describe the current cell therapy experience after stroke is “cautiously promising.” Clinical studies vary in stroke stage, delivery route, cell type, dose, and endpoints. Some focus on chronic stroke and motor recovery. Others look at acute or subacute ischemic stroke. Some measure imaging changes, some track functional scales, and some emphasize safety above all else. That variation is exactly why reading one positive headline is not enough. The papers suggest real scientific momentum, but they do not yet support the idea that cell therapy has become a routine, standardized answer for stroke recovery.
This is also where reality matters more than enthusiasm. FDA warnings continue to stress that regenerative medicine products, including stem cells and exosomes, are widely marketed with claims that go beyond current approval and evidence. The FDA says patients should avoid unapproved products from human cells or tissues marketed for treatment or cure of a wide range of conditions, and it has also stated that there are currently no FDA-approved exosome products. Serious adverse events have been reported with unapproved regenerative products.
So, where does that leave someone searching for stem cell therapy in Thailand for stroke? In the most realistic place possible: interested, hopeful, but careful. The research base is stronger than it was a few years ago. The idea is no longer speculative as it once was. There are human trials, meta-analyses, conference reports, and increasingly sophisticated reviews. But the field is still developing, and the best interpretation of today’s evidence is that stem cell therapy for stroke is a serious investigational area, not a settled mainstream treatment.
For readers who want the shortest, truthful summary, it is this: stroke recovery is long, uneven, and deeply personal. Standard emergency care and rehabilitation still matter most. But if you are reading about stem cell therapy in Thailand or trying to understand the real cell therapy experience, the papers are worth paying attention to. They show enough progress to justify real interest, but not enough certainty to justify overpromising. That combination is exactly what makes this one of the most-watched areas in modern stroke research.
FAQ: Stem Cell Therapy In Thailand, Stroke, and Cell Therapy Experience
Is stem cell therapy for stroke already a standard treatment?
Not at this point. Current studies and reviews show growing evidence and some encouraging results, but stem cell therapy for stroke is still best described as investigational rather than a fully standardized routine treatment.
What does “cell therapy experience” really mean in stroke research?
In the research setting, cell therapy experience usually refers to what clinicians and investigators have observed across trials: safety, motor outcomes, functional recovery, imaging changes, and how patients respond over time. It is not one single treatment model, because different trials use different cells, routes, and timelines.
How long does stroke recovery take in stem cell studies?
The papers do not support a simple fixed timeline. Stroke recovery often continues for months or years, and clinical studies of cell therapy commonly measure outcomes over extended follow-up rather than promising rapid change.
Are all regenerative products for stroke approved?
No. FDA warnings make clear that many regenerative medicine products are marketed without approval, and the agency has also stated that there are no FDA-approved exosome products.
Why are people so interested in stem cell therapy in Thailand for stroke?
Because stroke can leave long-term disability, the idea of a therapy that might support brain repair is naturally compelling. The interest is understandable; the key is to separate real research progress from claims that go beyond the evidence.