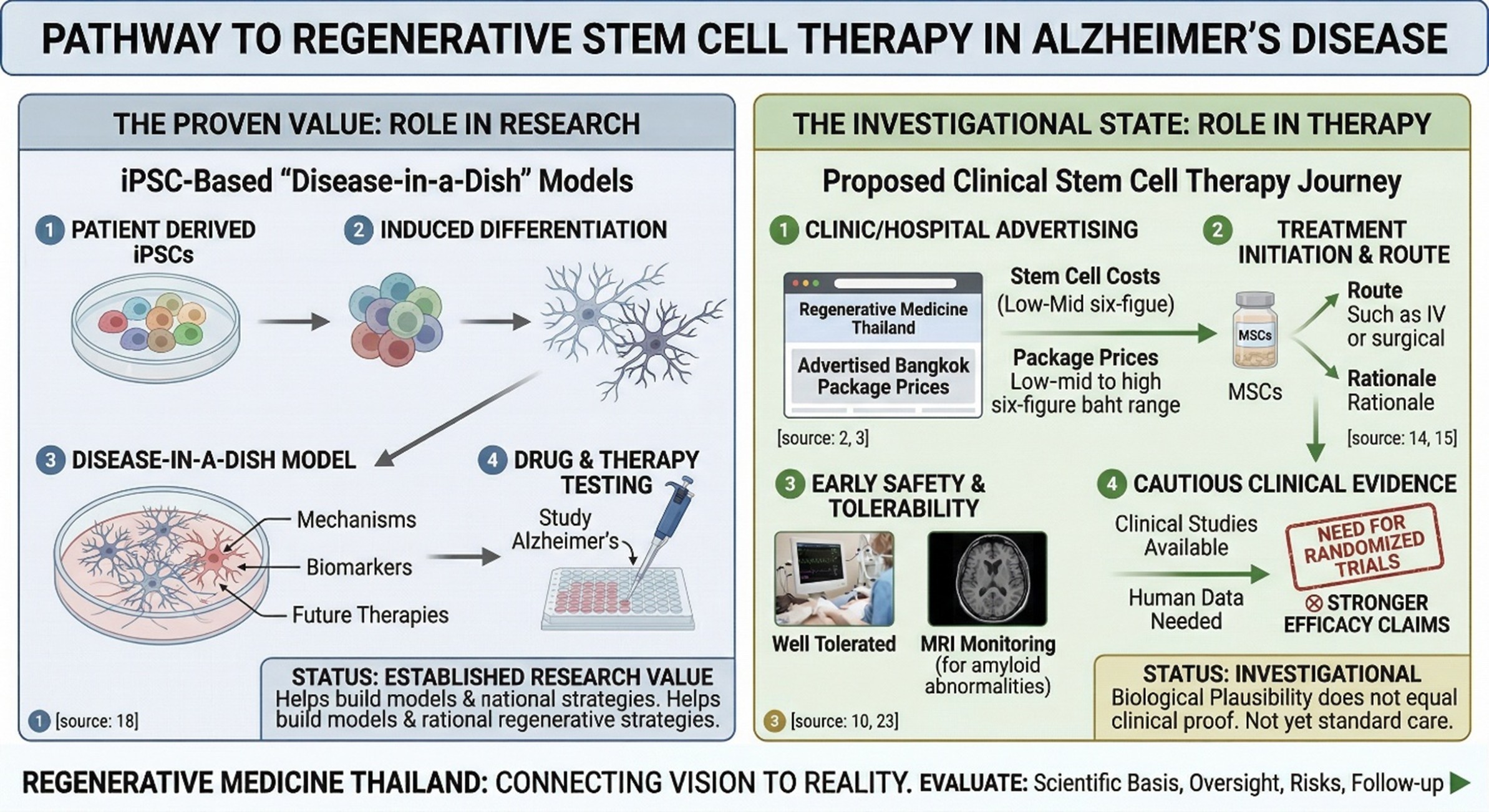
In regenerative medicine Thailand searches, families often look up stem cell costs almost immediately. That is understandable. Publicly advertised Bangkok package prices for Alzheimer ’s-related stem cell programs have been listed in broad bands from the mid-six-figure baht range up toward around seven figures, depending on the provider and protocol. But cost is a poor shortcut for scientific quality. Professional organizations such as ISSCR and CIRM repeatedly warn that stem cell interventions may be marketed at high prices long before they are proven to be safe or effective, which means price alone should never be mistaken for evidence.
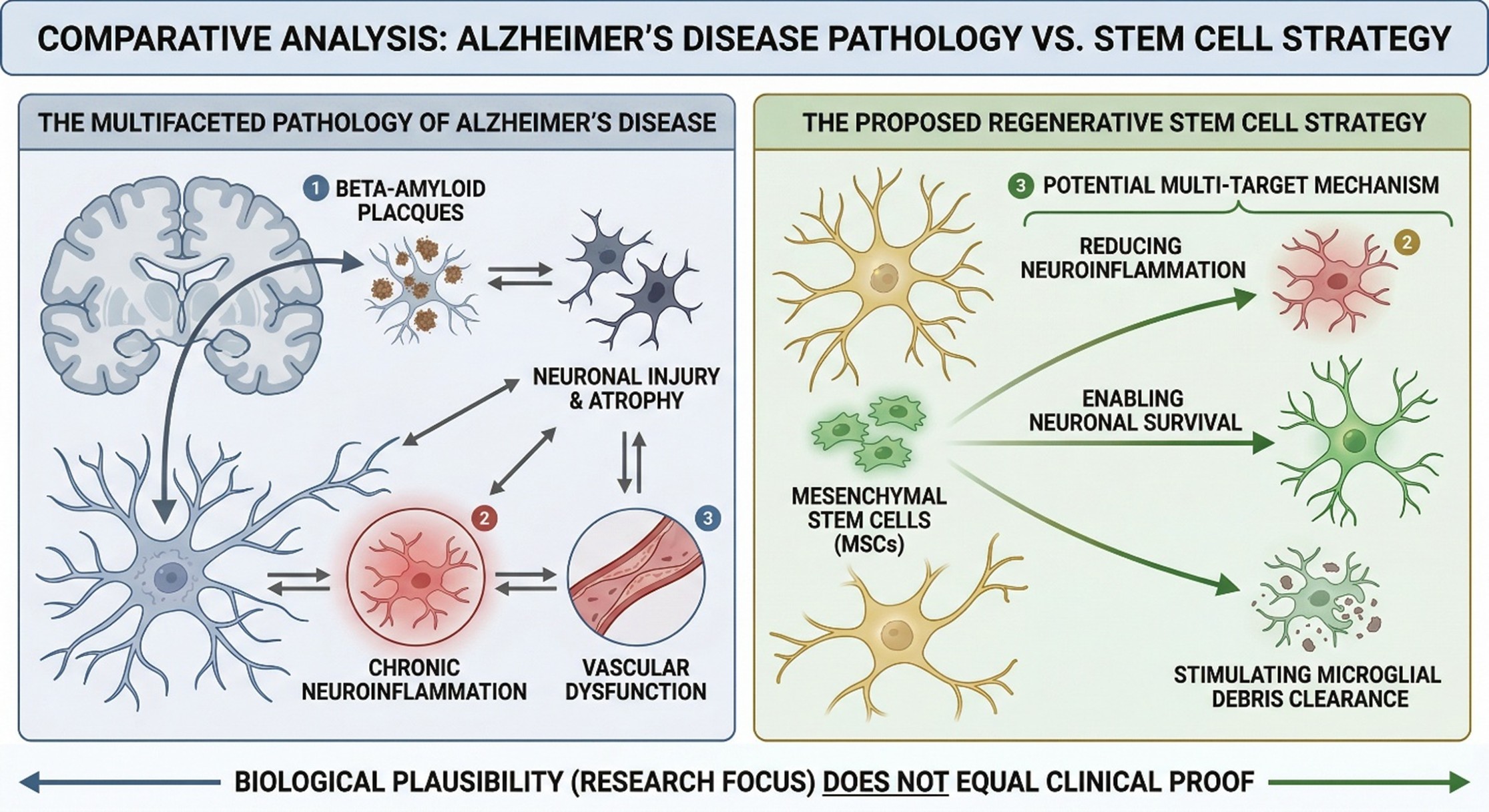
That point matters even more in Alzheimer’s disease, because this is not a simple target for repair. The disease involves progressive injury to neurons, breakdown of connections across brain networks, chronic inflammation, and vascular dysfunction that can further reduce nutrient delivery and impair clearance of toxic proteins. The National Institute on Aging notes that by later stages of Alzheimer’s, brain atrophy becomes widespread, while malfunctioning microglia and astrocytes may contribute to chronic inflammatory damage instead of effective debris clearance. This is one reason regenerative medicine Thailand conversations around Alzheimer’s disease tend to sound so urgent: the biology is devastating, and families are often searching for something more than symptomatic management.
Still, the current therapeutic landscape is more nuanced than many advertisements suggest. The NIA notes that lecanemab and donanemab are FDA-approved immunotherapies for early Alzheimer’s disease, targeting beta-amyloid plaques and requiring MRI monitoring because of risks such as amyloid-related imaging abnormalities. Meanwhile, the 2025 Alzheimer’s drug development pipeline documented 138 therapies across 182 clinical trials, which shows that the field is moving rapidly but also that no single strategy has solved the disease. This is exactly where stem cell-based ideas enter the discussion: not because the disease has already yielded to regenerative medicine, but because conventional and disease-modifying options still leave major unmet needs.
From a scientific point of view, it’s easy to see why people are so excited about using stem cells in Alzheimer’s disease. Researchers are asking whether products of mesenchymal stem cells and comparable sources might help by reducing neuroinflammation (host defense activity), enabling neuronal survival, or stimulating microglial interference–and so forth. Recent reviews point to this area as biologically plausible and increasingly complex, particularly in the context of inflammation, trophic signaling, and resorting to surgery to solve all brain delivery problems. But biologically plausible isn’t the same thing; proof positive that this condition is both verified and treatable comes from where one has written responsibly about regenerative medicine Thailand.
There is also another layer that often gets missed in patient-facing content: stem cells are already deeply useful in Alzheimer’s science even when they are not yet established as routine therapy. NIA’s dementia research materials highlight how scientists use induced pluripotent stem cells to create neurons and “disease-in-a-dish” models that help study Alzheimer’s mechanisms and test future therapies. In other words, stem cells already matter in Alzheimer’s disease research in a very real way. They are helping build the models that may eventually produce better drugs, better biomarkers, and more rational regenerative strategies. That is less dramatic than a promise of a cure, but scientifically, it is one of the strongest and most credible contributions of stem cell biology today.
When we move from laboratory science to actual human treatment, the evidence becomes much more cautious. A 2024 systematic review of cell-based therapies in Alzheimer’s disease concluded that these approaches were generally well tolerated in the available studies, but also stressed the need for carefully planned randomized trials before stronger efficacy claims can be made. A 2024 Frontiers review similarly noted that only a small number of completed clinical studies were available and that more human data are still needed to show both safety and effectiveness. That is an important reality check for anyone comparing stem cell cost across clinics: the research signal is interesting, but the clinical evidence is still early and far from definitive.
Connecting vision to reality in this way is not merely a technical difficulty. It is reflective of the difficulty of the disease itself. Alzheimer’s is more than just replacing one missing cell type. Protein aggregation, synaptic loss, glial dysfunction, blood-brain barrier changes, metabolic stress, vascular injury, and progressive network failure in the brain-all these different problems are involved in one problem to overcome. Reviews of stem cell therapy for Alzheimer’s disease constantly review the situation as promising in part because stem cells might affect more than one of these pathways at the same time, but they also stress that it remains difficult to translate from animal models to benefits in humans. That is why the best academic writing about regenerative medicine Thailand and Alzheimer’s disease should sound measured, not breathless.
Safety and oversight are equally important. The Frontiers review discusses concerns such as immune reactions, tumorigenicity, unintended differentiation, contamination risk, and the broader difficulty of translating stem cell products safely into human neurological care. CIRM’s patient advisory makes a similar point from the public-health side: experimental cell therapies should ideally be studied in rigorous clinical trials with independent review, transparent informed consent, clear reporting of outcomes, and full disclosure of risks and costs. That guidance is especially relevant when families are searching for stem cell cost first and science second. In a disease as serious as Alzheimer’s disease, the order should be reversed.
So, where does that leave regenerative medicine Thailand today for Alzheimer’s disease? In realistic terms, it sits in a space between serious scientific interest and incomplete clinical maturity. Science is not imaginary. There are genuine mechanistic reasons to study mesenchymal stem cells, extracellular vesicles, neural repair strategies, and cell-based delivery systems in Alzheimer’s disease. There are also early human studies suggesting feasibility and tolerability. But none of that currently justifies treating stem cell therapy as established standard care for dementia. The strongest evidence today still supports comprehensive dementia evaluation, evidence-based medical management, caregiver planning, and discussion of approved therapies where appropriate, while regenerative approaches remain investigational.
That is also why stem cell costs deserve only a small place in the conversation. Yes, in Thailand the advertised price range can be broad, and yes, families understandably compare packages. But the harder and more useful questions are these: What exact cell product is being used? What is the route and rationale? What peer-reviewed data support that protocol? How is follow-up handled? What outcomes are being measured, and over what time frame? Those are the questions that make content sound human, because they reflect what real families eventually discover: when the disease is this serious, the price tag is never the whole story.
FAQ
- Is stem cell therapy already a standard treatment for Alzheimer’s disease?
Current reviews describe cell-based therapy for Alzheimer’s disease as promising but still investigational, with limited human trials and a need for stronger randomized evidence. - Why do people search for stem cell costs so often in regenerative medicine Thailand?
Because publicly advertised packages vary widely, and Thailand is often discussed in medical-travel contexts. But professional guidance stresses that families should evaluate the scientific basis, oversight, risks, and follow-up plan rather than use price as a proxy for quality. - What is the strongest current role of stem cells in Alzheimer’s science?
One of the strongest current roles is in research itself: stem cells are already being used to create human neuronal models that help scientists study Alzheimer’s mechanisms and test future therapies. - Can regenerative medicine help with the biology of Alzheimer’s disease at all?
Possibly, which is why the field is active. Researchers are studying whether stem cells or stem-cell-derived products can influence inflammation, neuronal survival, microglial function, and delivery across the blood-brain barrier. But that biological rationale is still ahead of definitive clinical proof.