There is a trend in the clinical management of complex neurodevelopmental conditions to go beyond symptom-oriented treatments and engage with interventions that alter key biological pathways. Over the last several decades, research into the co-occurrence of Autism Spectrum Disorder (ASD) and Attention-Deficit/Hyperactivity Disorder (ADHD) has become an increasingly active area of investigation in light of this overlap that further complicates cognition، behavior, and sensory processing. In some cases, however, recent scientific studies have suggested that this overlap could lead to chronic neuroinflammatory activity and changes in immune signalling. In this context, umbilical cord-derived mesenchymal stem cells (UC-MSCs) have gained interest as a possible cell supportive strategy due to their immunomodulatory and neuro-supportive properties. Advanced regenerative medicine centers in Thailand are providing a contribution to this field through structured clinical programs investigating whether UC-MSC-based interventions can balance biological dysregulation underlying neurodevelopmental dysfunction.
- Biological Complexity in the Autism–ADHD Overlap
To appreciate the rationale for studying UC-MSCs in this context, one needs to consider the biological complexity underpinning the overlap between ASD and ADHD. There is an increasing amount of research focusing on how microglial activation, chronic inflammatory signaling and altered synaptic development affect different populations of children with neurodevelopmental difficulties. Then microglia are the resident immune cells in brain, and when they become persistently activated upon their exposure to inflammatory signals (Avignone et al., 2008), Microglia release pro-inflammatory mediators like IL-6, TNF-alpha- which are involved in regulating the signaling pathways inside of the brain and shaping of neural network.
This changed inflammatory milieu might disrupt synaptic pruning, the natural developmental process by which the brain fine-tunes and optimizes neural networks. Then, if there’s a less organized structure on the synapse level, children may respond with greater degrees of sensorimotor persistence (sensitivity to sensory information), attention dysregulation or impulsivity and social processing inhibitors. Within this framework, UC-MSC-based treatment is being studied for its potential not just in reducing symptoms (via immune and inflammatory mechanism) but also as a means to affect the microenvironment that may underlie brain function.
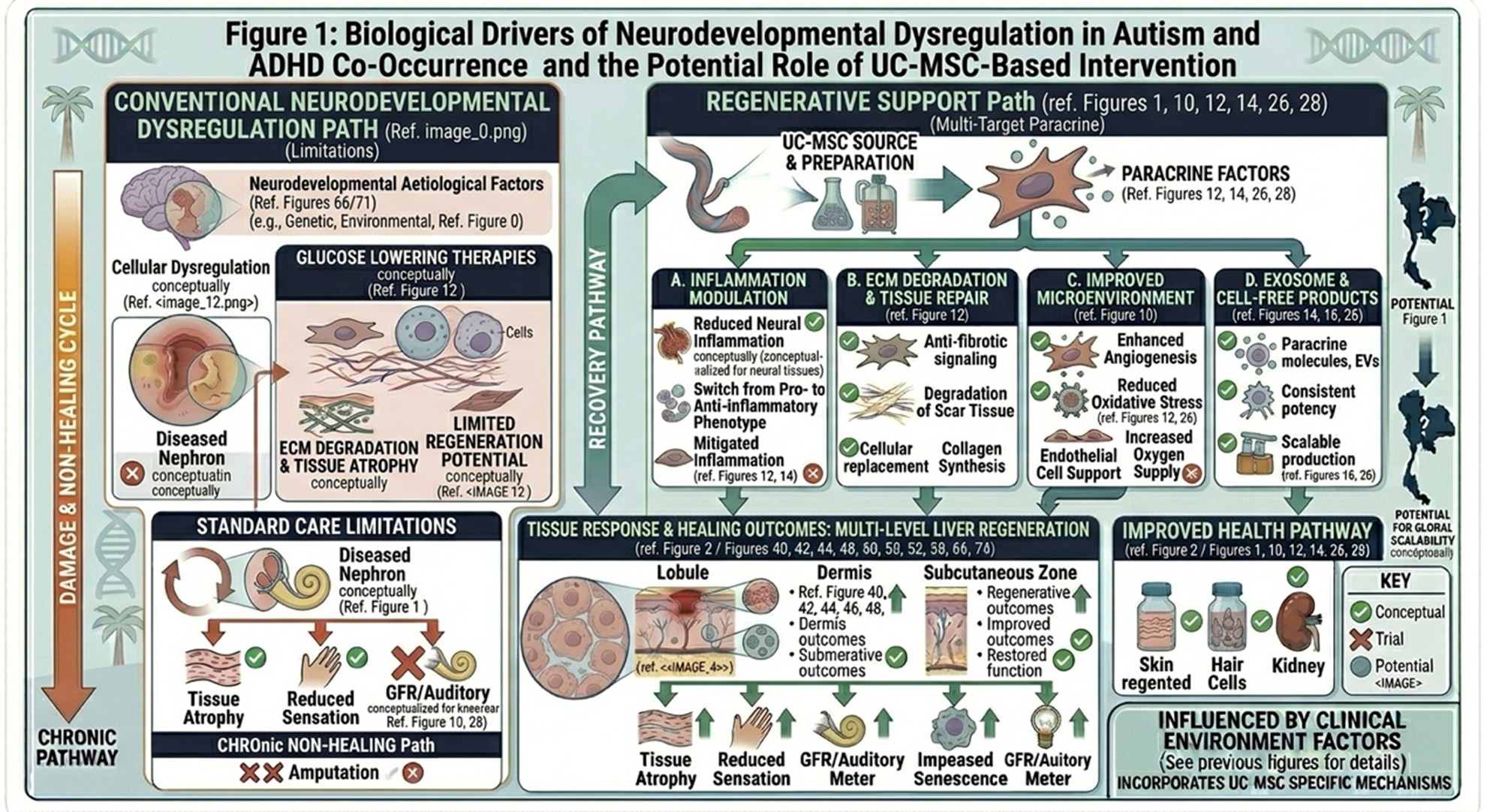
- Paracrine Activity and Immune Regulation
UC-MSCs are being investigated in neurodevelopmental medicine for a core reason: their paracrine signaling capability. Rather than acting mainly through the direct replacement of neural cell types, these cells are thought to mediate a large part of their function by releasing bioactive factors capable of modulating inflammation and IL-10 homeostasis, as well as driving tissue signals. This substance can be secreted cytokine, factor related to growth and extracellular vesicle and is called secretome.
One candidate pathway is increased regulatory T-cell activation. UC-MSCs may promote the immune response to shift away from an excessive inflammatory activation state to a more balanced, healthy immune response. This type of immune modulation may help subjugate over active inflammatory pathways which helps living organisms better combat the biologic stressors associated with sensory overstimulation, lack of focused concentration or behavioral dysregulation in selected patients. In this manner, UC-MSC-based therapy is presented as a biological adjunct that could augment developmental therapies rather than displacing them.
- Exosomal Communication and Molecular Influence
A second aspect open to feeding into the future direction is MSC-derived exosomes, nanosized vesicles that can include proteins, lipids and regulatory RNA. These vesicles are of increasing interest because they can be part of a cellular signaling machinery at a distance and, under certain conditions, may communicate with the central nervous system in clinically relevant ways. In arguably the most common mechanism discussed, exosomal signaling by UC-MSCs is proposed to help modulate inflammatory pathways and improve the cellular environment of neural tissue in neurodevelopmental research.
This has contributed to the notion that UC-MSCs could act as a mode of a living biological signaling system, exerting effects beyond their time of administration. Instead of immediate structural rearrangement, the conceptual advantage is in slowly adjusting internal ecology—potentially promoting optimal washout processes, energy utilization efficiency and more harmonious cell-cell communication over time. For families facing ASD & ADHD overlap, this type of model can be considered different than more overtly symptom-suppressive proposals, because it does attempt to get at underlying biological contributors in a more integrative way.
- Neurotrophic Support and Synaptic Plasticity
In addition to immunomodulation, UC-MSCs are under study for their potential use in promoting neuroplasticity One pathway that is often invoked in the literature is stem-cell signaling regulating Brain-Derived Neurotrophic Factor (BDNF), a molecule thought to influence neuronal wellbeing, synaptic development and learning-related plasticity. In children with neurodevelopmental challenges altered BDNF signaling has been suggested as one element that could influence connectivity and adaptive neural development.
Also, via indirect signaling mechanisms, UC-MSCs may promote microenvironmental conditions that are more conducive to optimal synaptic transmission and increased neural plasticity. In the context of a structured clinical setting, such biological support is most frequently combined with rehabilitation, behavioral therapy or developmental training, under the premise that a more balanced neural environment could enhance ongoing therapeutic work. The larger aim is not to assert a cure but to facilitate brain function in ways that potentially enhance attention, regulation, learning and engagement.
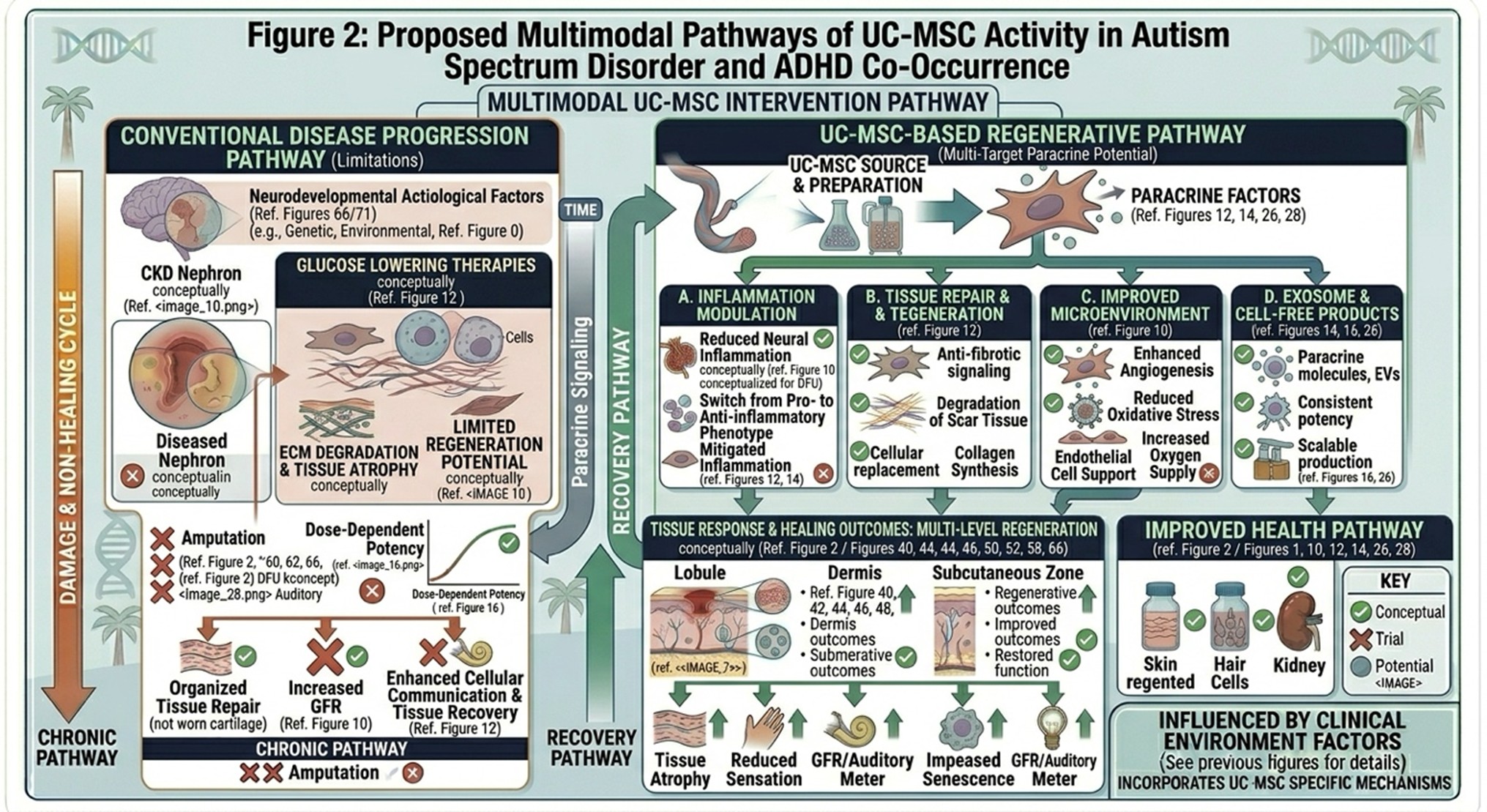
- Clinical and Translational Considerations in Thailand
Thailand has quickly become a region of interest for many international families seeking advanced supportive options, due to its emerging regenerative medicine infrastructure and the experience of its hospitals in caring for international patients. Clinics working in this area often tout standards for cell preparation, maintaining viability and purpose-built treatment workflows. Umbilical cord derived MSCs tend to receive positive attention due to the fact that they come from neonatal tissue, resulting in a younger cellular profile and greater biological activity compared with many other adult-derived sources.
Some Thai programs also take a wider systems approach to neurodevelopmental care by recognizing the role of factors such as gut-brain axis, immune burden and systemic inflammation. This points to a growing belief that neurological and behavioral disorders may not only be influenced by the brain itself, but also by gut permeability, microbial equilibrium or imbalance therein, and immune signaling around the body. Within this framework, UC-MSCs can be seen as part of an immunoregulatory strategy that is broader in scope than the isolated intervention.
- Concluding Perspective
UC-MSC-based approaches in the support of children with ASD and ADHD overlap are an emerging field of regenerative medicine. The potential impact of these cells lies in their ability to modulate neuroinflammation, favor synaptic plasticity and create a more neuroprotective niche for the brain. This is particularly important in cases where standard treatments improve certain skills and behavior, but families are also seeking strategies that may directly address underlying physiological stress.
Responsible discussion is important as well, of course. The UC-MSC-based therapy should be presented as a supportive, emerging clinical intervention rather than an assured definitive solution. The most plausible way forward looks like prudent patient selection combined with realistic expectations, diligent medical oversight, and incorporation of evidence-based developmental care. For families considering advanced care options, the advantage to Thailand in that balanced approach is now gaining recognition as a destination where clinical experience, regenerative protocols and multidisciplinary support can be focused together and comprehensively assembled.

