I’m treating “Stoke” here as stroke, because that is the medically relevant context for stem cell treatment and the surrounding research.
Why stroke keeps coming up in stem cell conversations
Stroke is a medical emergency, which occurs due either to block of the (blood)Supply to/inside brain or bleeding in or around it. However, stroke is most often ischemic rather than hemorrhagic and the leading cause of long-term disability. That one fact alone helps to explain why many people searching for a stem cell clinic after stroke are typically seeking something beyond conventional rehabilitation. They seek to restore function rather than compensate for loss.
That hope is not irrational. Modern stroke care can save brain tissue in the acute phase with treatments such as thrombolysis and mechanical thrombectomy in appropriate patients, but many people are still left with lasting weakness, language difficulty, imbalance, or cognitive change. Stem cell research entered this space because it asks a different question: after the emergency phase is over, can brain repair be biologically supported in a more active way?
The biology of stem cells, in plain language
What stem cells actually are
Biologically, stem cells can be characterized by two key aspects: they have the ability to self-renew and differentiate into more specialized cell phenotypes. According to NIH, The National Institutes of Health defines a stem cell as having an extraordinary potential to develop into many different kinds of cells in the body. This is this basic biology that gives them such regenerative medicine potential.
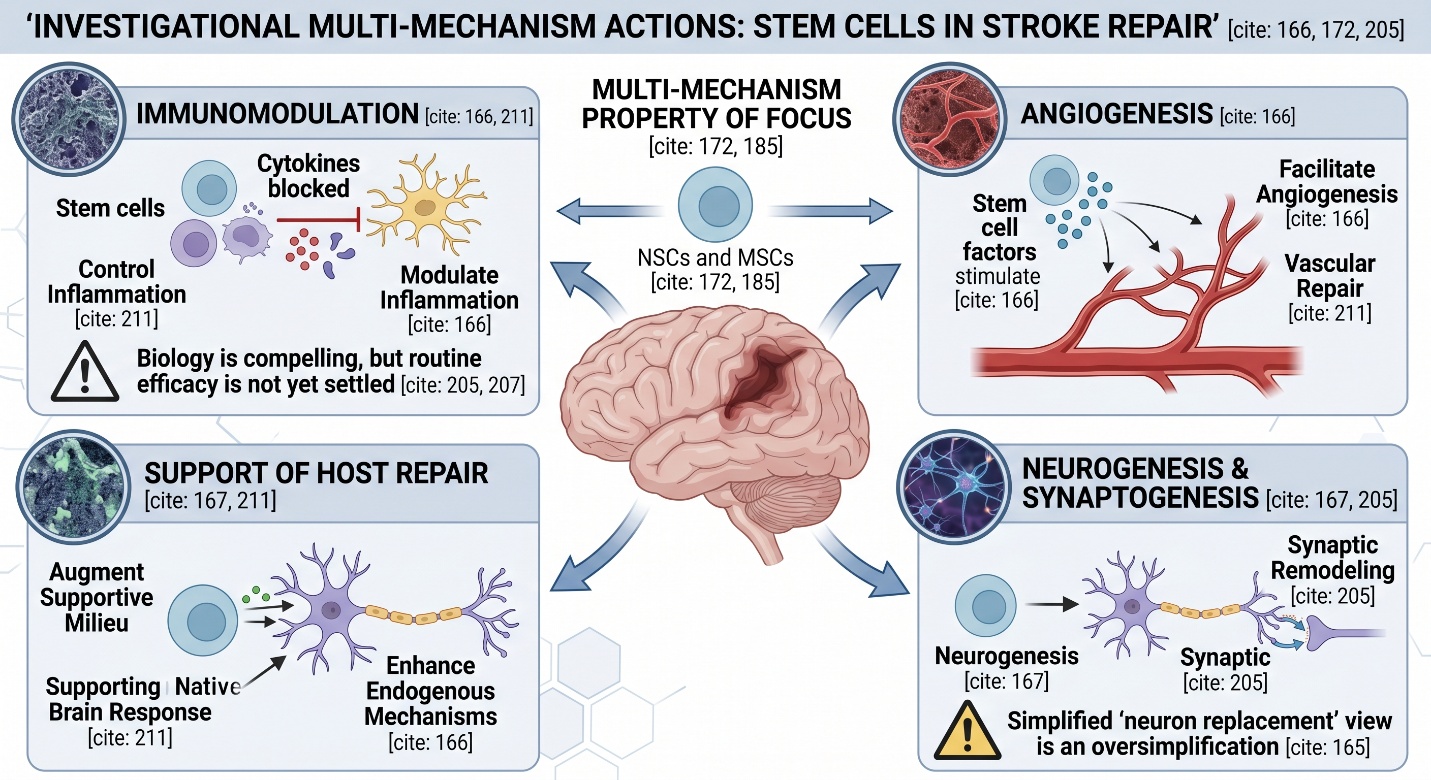
However, stroke research is not solely based on the idea that stem cells can become “neurons.” That is a common oversimplification. Stem cells are frequently investigated in the contemporary stroke literature as they may work through paracrine signaling and immunomodulation — they can modulate inflammation, facilitate angiogenesis, enhance endogenous reparative mechanisms and augment a supportive milieu even after direct cell replacement is constrained. When reviewing 2025 papers, recent articles refer to mechanisms of immunomodulation, angiogenesis, neurogenesis and synaptogenesis as well as support of host repair.
Why that biology matters in stroke
Brain stroke damages the brain in layers. The immediate loss is of tissue, and this triggers a second cascade which encompasses inflammation, excitotoxicity as well as vascular dysfunction (and remodeling). This is why stem cell research for stroke lacks a simple solution like replacing the dead tissue one cell at a time. It comes down to whether you can encourage the post-stroke brain into a more amenable state for repair. 2025 and 2026 reviews described the various neural stem cells as well as mesenchymal stem cells being a major focus of study precisely for this multi-mechanism property.
What a stem cell clinic should and should not imply
A clinic offering treatment is not the same thing as a treatment being established
This is where the phrase stem cell clinic needs more discipline than it usually gets. The FDA states that regenerative medicine therapies have not been approved to treat neurological disorders such as stroke, and it also warns that many stem cell products are marketed before they are shown to be safe or effective. FDA consumer guidance adds that, in the United States, the only FDA-approved stem cell products are cord-blood-derived blood-forming cells for hematopoietic disorders, not for stroke or other neurologic conditions.
That does not make the field fake. It means the field is still investigational. A credible clinic conversation about stroke should therefore sound more like a trial discussion than a miracle promise. It should explain which cell type is being used, by what route, in what stage of stroke recovery, with what published evidence, and with what follow-up plan. If those details are vague, the confidence is usually outrunning the science.
What the clinical evidence in stroke actually shows
The field has moved beyond theory
This part is important. Stem cell therapy for stroke is no longer only an animal-lab idea. A 2025 review of clinical trials and advanced MRI in ischemic stroke describes multiple human trials across different stages of stroke, and a 2026 review of clinical trials in stroke and spinal cord injury similarly notes that neural stem cells and related cell types are already being tested clinically. That means the research question is serious enough to have reached real patients.
But the efficacy picture is still mixed
The strongest honest summary right now is not “it works” and not “it failed.” It is “the results are still mixed.” A 2025 Scientific Reports systematic review and meta-analysis of randomized controlled trials in acute and subacute ischemic stroke concluded that the efficacy of stem cell therapy in long-term functional outcomes remains unclear. A separate 2025 network meta-analysis also evaluated different stem cell types and emphasized that comparative efficacy and safety are still being worked out.
That is exactly where many articles lose honesty. Small positive studies, encouraging imaging signals, or functional gains in selected groups do matter. But they do not yet add up to a mature, clinic-ready standard of care. The current literature still struggles with differences in cell type, timing, dose, route of administration, stroke subtype, and outcome measures.
Why timing and patient selection matter so much
Stroke is not one biologic moment
One reason the evidence stays hard to interpret is that “stroke” is actually several treatment windows, not one. Acute ischemic stroke, subacute stroke, chronic post-stroke disability, ischemic stroke, and hemorrhagic stroke are not identical research problems. A 2025 review focused specifically on the idea that “time is brain” for cell therapies, which reflects how strongly treatment timing may influence mechanism and outcome.
This matters for readers because a therapy aimed at acute neuroprotection is not the same as one aimed at later remodeling or rehabilitation support. It also means that even a genuinely promising stem cell approach might help only certain kinds of stroke patients at certain stages of recovery. That is one reason a good article about a stem cell clinic and stroke should sound narrower, not broader, as it becomes more scientific.
Where the field is strongest right now
Mechanistic promise is stronger than routine clinical certainty
If you step back and read the better reviews side by side, a pattern appears. The biology of stem cells in stroke is compelling: anti-inflammatory effects, support of angiogenesis, neurogenesis, synaptic remodeling, and recovery of the post-stroke microenvironment all make biologic sense. The clinical field is active. Safety signals in many early studies are encouraging. But routine efficacy is not yet settled enough for standard clinical endorsement. That is probably the most accurate place to stand in 2026.
The most honest conclusion
If we want to be as nice as possible, this is the cleanest way of saying it — stem cells matter in stroke not because of solid marketing but because there really exists biology. Stem cell biology provides insight into why researchers continue to revisit this domain—not just for applications in replacement but also inflammation control, vascular repair and supporting native brain response. However, when people cross the divide from science to a stem-cell clinic they enter an area where evidence is still emerging and regulatory caution remains important. Personalized stem cell therapy for stroke is encouraging translational medicine at this time, but not yet standard of care.