Why does this topic sound simpler than it really is
Those who search Botanical Stem Cell Supplements and Multiple Sclerosis are typically trying to link two extremes— the lure of something natural, with the promise (or potential) of activation. That is understandable. MS is a chronic immune-mediated central nervous system disease, and for all those living with it at some point the harder questions start to emerge about repair or remission and whether treatment can do more than slow damage. However this is also a subject, where the language gets slippery really fast. The words fit together though in a way that the science often does not.
What Botanical Stem Cell Supplements usually are
More supplement language than stem cell medicine
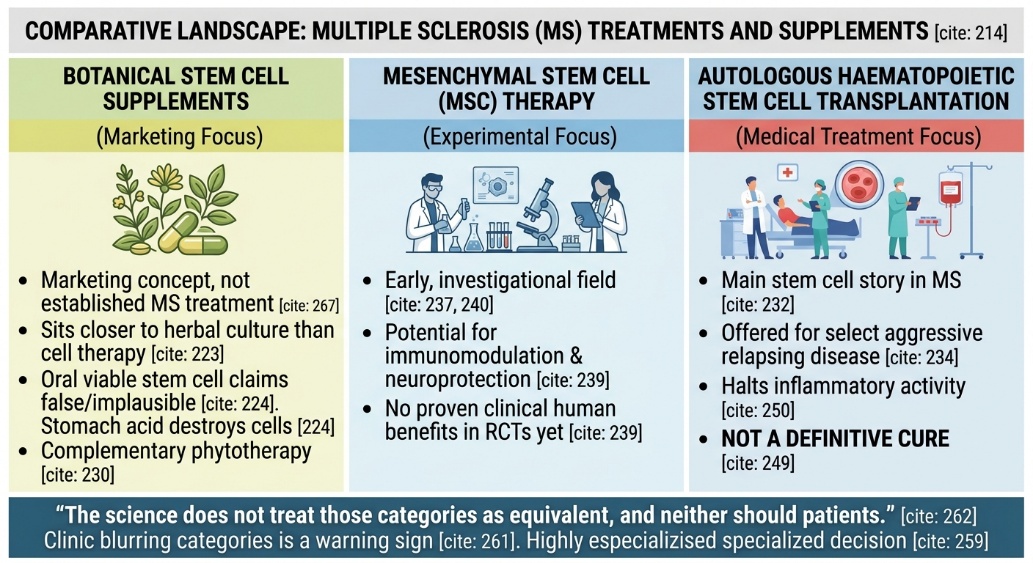
The phrase Botanical Stem Cell Supplements is strong from a marketing point of view, but weak as a formal MS treatment category. In practice, these products usually sit much closer to herbal or plant-derived supplement culture than to real cell therapy. Regulatory warnings make that distinction very clear. Singapore’s Health Sciences Authority says claims that orally consumed products contain active live stem cells are false and not scientifically plausible, because viable stem cells require ultra-cold storage and would be destroyed in the gastrointestinal tract. HSA also states that “contains stem cells” or “a stem cell therapy” claims for oral products are not supported by scientific evidence.
Plant compounds may be biologically interesting, but that is a different conversation
This does not mean that plant-derived compounds are void in MS research. Likewise, literature on herbal therapy and medicinal herbs in MS reveals interest in the anti-inflammatory, antioxidant, and neuroprotective effects of certain higher-order plants (driven by the common perception that botanicals exert some beneficial effect), as well as laboratory-based studies suggesting that some plant extracts may modulate ontogenetic stem cell behavior in experimental models. However, this is a whole lot different than equating a Botanical Stem Cell Supplement to stem cell therapy for MS; one falls into primarily complementary or experimental phytotherapy, and the other cellular medicine.
Where real stem cell therapy for MS actually stands
The main stem cell story in MS is AHSCT, not supplements
When the dialogue moves from drugs to genuine cell-dependent remedy, in best precedence is autologous haematopoietic stem mobile transplantation (AHSCT). For this reason, AHSCT can be offered in clinical practice when done in select patients with MS based on a 2025 ECTRIMS–EBMT consensus update that considers the use of conditioning regimens for aggressive and highly active relapsing disease. In more clinical language, what the Cleveland Clinic nicely illustrates here is that the most likely candidates for benefit were younger people who are still ambulatory and have had relatively recent, highly inflammatory relapsing MS despite treatment with a high-efficacy disease-modifying therapy. Which is a tiny group, not everyone with MS.
Mesenchymal stem cells are still a different, earlier field
The other substantial class is mesenchymal stem cell (MSC) therapy, but the evidence for that one is much less definitive. While reviews from as recently at 2025 characterize MSCs as new fonts of hope, given their immunomodulatory and neuroprotective potential, they also make it clear that there are still no firm clinical benefits in humans yet proven by randomized controlled trials—and that the principal limitation remains efficacy rather than enthusiasm. Moreover, the 2025 systematic review of MSC clinical trials for MS presents such therapy in a similar light as effective yet still investigational rather than established care.
What the “success rate of stem cell therapy for MS” really means
It depends completely on which therapy and which patient
This is what people most care about, but also the easiest to misinterpret. There is not one number for the success rate of stem cell therapy MS, partly because there are such disparate interventions and patient groups involved in what we now refer to as «stem cell therapy» for MS. ECTRIMS: Data from 5-Year Single-Center Protocol of AHSCT for Selected Relapsing-Remitting MS Show Progression-Free Survival Rate of 95% and Overall Survival Rate was 98%. Among other observations, real-world evidence cited by the UK MS Society has shown that 87% of people were flare-free after four years following HSCT. Those are solid numbers but in selected patients and for controlled outcomes such as relapse control or progression-free survival not a blanket claim to cure.
Even strong outcomes are not the same as a cure
This is where honest writing matters. ECTRIMS patient education explicitly says there is currently no suggestion that anyone has been definitively cured of MS, even when some people achieve long periods of “no evidence of disease activity” or remission-like outcomes. The wording is important. In MS, the best AHSCT results are often about halting inflammatory activity in the right patients, not erasing the disease from existence.
Progressive MS changes the picture substantially
The success-rate discussion also becomes less impressive when disease biology changes. Cleveland Clinic notes that AHSCT has modest or no efficacy in preventing or reversing progressive disability worsening when there has not been recent relapse or MRI inflammatory activity, and the MS Society notes that HSCT in active progressive MS is usually something to consider preferably in a clinical trial. So any article that gives one neat “success rate of stem cell therapy for MS” without separating relapsing disease from progressive disease is making the field sound simpler than it is.
What this means for a clinic decision
A credible clinic should sound more like a transplant center than a supplement brand
This is probably the most practical point in the whole article. A trustworthy MS stem cell program should be able to explain which therapy is being offered, why that therapy fits that exact MS phenotype, what the risks are, what the follow-up plan looks like, and whether the treatment is part of a regulated clinical pathway. Cleveland Clinic repeatedly frames stem cell treatment for MS as a highly specialized decision, not a generic wellness service, and FDA says stem cell products generally require approval, with currently approved stem cell products in the U.S. limited to cord-blood-derived blood-forming cells for hematopoietic disorders, not MS treatment.
The biggest red flag is false equivalence
If a clinic blurs Botanical Stem Cell Supplements, herbal support, MSC infusions, and AHSCT into one reassuring story, that is not sophistication — it is usually a warning sign. The science does not treat those categories as equivalent, and neither should patients. Supplements may or may not have a place in supportive care discussions. AHSCT may have a role in carefully selected relapsing MS. MSC therapy remains investigational. Those three things should never be sold as though they all carry the same weight.
The most honest conclusion
The cleanest way to say it is this: Botanical Stem Cell Supplements are mostly a supplement-marketing idea, not an established MS stem cell treatment. The real stem cell discussion in Multiple Sclerosis is about AHSCT for selected relapsing disease and, more experimentally, MSC therapy. And when people ask about the success rate of stem cell therapy for MS, the most truthful answer is that the best results belong to carefully selected AHSCT patients — not to supplements, not to every form of MS, and not to a universal cure narrative. The science is real, but it still rewards precision more than hope-driven shortcuts.