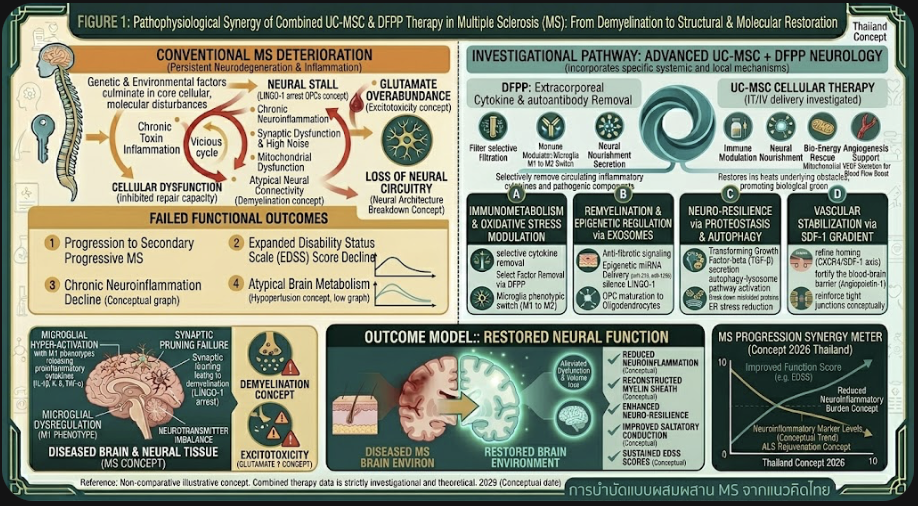
Historically, the clinical course of Multiple Sclerosis has been driven by this biologic inflammatory wave which propagates against a backdrop of central nervous system injury as well as ongoing neurodegeneration. Classical disease-modifying therapies are limited by a ceiling effect in treating any of the progressive neurodegeneration seen in the later course of this disease. This therapeutic gap has led to the development of stem cell therapy in Thailand based on the specific potential of Umbilical Cord-derived Mesenchymal Stem Cells (UC-MSCs). These morphologically immature cells are not anymore considered simple immune modulators but fully qualified biological architects that have the ability to rewrite the pathological script of the CNS by means of complex molecular tools.
Mitochondrial Transfer and Metabolic Reprogramming
One frontier mechanism now emerging in the UC-MSC study is Immunometabolism Shift. It is a pro-inflammatory environment of M1 macrophages and pathogenic Th17 cells dependent on anaerobic glycolysis in Multiple Sclerosis patients. In high-resolution cellular studies less than recently, it is indicated that UC-MSCs actively export sound mitochondria to these stressed cells during Tunneling Nanotubes (TNTs).
Such mitochondrial transfer resets the metabolic landscape of the immune microenvironment. UC-MSCs restore oxidative phosphorylation in resident microglia and infiltrating T-cells drives a phenotypical shift from the aggressive M1 phenotype to the anti-inflammatory M2 phenotype. This metabolic reprogramming is responsible for the long-lasting decrease in neurotoxic cytokine secretion combined with an increase in the release of Interleukin-10 (IL-10), which generates a biochemical buffer zone around axons that renders them less susceptible to secondary demyelination.
Epigenetic Regulation via Exosomal MicroRNA
Their stem cell therapy research progress has been focused on the secretome, with emphasis placed on the role of extracellular vesicles, or exosomes. These nano-scale UC-MSC-derived messengers contain a payload of specific MicroRNAs (miRNAs), epigenetic regulators. The inability of remyelination in Multiple Sclerosis is frequently associated with the blockage to Oligodendrocyte Progenitor Cells (OPCs) by inhibitory signaling cascades such as LINGO-1.
Recently published work shows that exosomes derived from UC-MSCs deliver miR-219 and miR-125b directly to these stalled OPCs. Importantly, these miRNAs silence the LINGO-1 gene and dampen Notch signaling, the main molecular brakes on myelin repair. Stem cell therapy in Thailand eliminates these natural barriers, allowing de novo myelination (true re-construction of the myelin sheath) to restore the saltatory conduction pathway for nerve impulses lost during Multiple Sclerosis progression.
Maintaining proteostasis and promoting autophagy
In multiple sclerosis, misfolded proteins and damaged organelles accumulate in neurons leading to ER stress as well as apoptosis which is termed Neurodegeneration. UC-MSCs are intervening in this process via modulation of Proteostasis or protein homeostasis. These stem cells activate the autophagy-lysosome pathway in neurons via Transforming Growth Factor-beta (TGF-β) secretion.
This cellular detoxification process enables the decomposition of toxic protein clumps accumulated during long-term inflammation, and subsequently their recycling into the production of new proteins by neurons. When coupled with UC-MSCs, the neuron can bolster its own inherent mechanisms to retain proteins in a dynamic intracellular milieu—this confers neuro-resilience. Making certain that even in the presence of low grade inflammation, the neural architecture maintains structural integrity such that it delays, or slows expansion from the relapsing-remitting form of Multiple Sclerosis to secondary progressive Multiple Sclerosis.
SDF-1 Gradient Vascular Stabilization
Disruption of the blood-brain barrier (BBB) permits central nervous system infiltration in Multiple Sclerosis. ConclusionUncoupled by a less potent chemokine axis, UC-MSCs display an exquisitely sensitive homing ability. Those cells detect the SDF-1 gradient released from injured neural tissues and navigate directly to the place of the injury.
And UC-MSCs do not just hang around when they show up; they repair the vascular lock. They fortify the tight junctions of the endothelial cells that create the BBB by releasing Angiopoietin-1 and specialized junctional adhesion molecules. This sealing effect is important because it keeps more autoreactive leukocytes from entering the CNS. This dual mechanism of migrating to the injury and then reinforcing the barrier makes Thai stem cell therapy one of the most integrative strategies at stopping disease action at its entry point.
Figure A: Neuro-Regenerative Nature of UC-MSCs in MS
Regenerative medicine clinical scenario in Thailand
The recent adaptation of UC-MSCs for MS is now highly specialized in Thailand. UCB-MSCs with natural neonatal vitality are now generated under Low-Oxygen expansion methods in leading clinical facilities, which ensure the retention of initial UC-MSCs’ stemness and potency prior to therapeutic delivery. More recent protocols have progressed to combinatory delivery routes; while intravenous infusion controls systemic autoimmunity, intrathecal administration—exposing cells directly in the cerebrospinal fluid—utilized to circumvent the BBB and optimize neurotrophic factor concentrations around spinal cord lesions.
This personalized medicine methodology is frequently paired with advanced neuro rehabilitation to prepare the brain for the natural healing signals that come with stem cell therapy Thailand. Combining cellular biology and clinical expertise, Thai specialists are setting standards on safety and efficacy aiming at either long-term disability stabilization or marginal improvement of the EDSS score in patients not amenable to conventional therapies.
The paradigm of Multiple Sclerosis (MS) treatment is shifting from one-dimensional immune suppression to a multi-targeted regenerative approach. The underlying deep molecular mechanisms of the UC-MSCs—from mitochondrial transfer and epigenetic regulation through exosomes to stabilizing the blood-brain barrier—provide a nuanced toolset for combating the multifaceted nature of Multiple Sclerosis. With extending on these standards with stem cell therapy in Thailand value and optimizing the complete paracrine capability of umbilical cord derivatives, the opportunity of reversing neurological deficits is turning into a truth —scientific reality. This interplay of primitive cells with the distressed nervous system is the future in neurology, offering a renewable avenue for restoration and recovery for people suffering from Multiple Sclerosis.