Why this keyword combination matters
The term “Botanical Stem Cell Supplements” is popular due to the fact that it includes two notions people already want to buy into: plant-based products are gentler, and stem cells represent the future of medicine. In Parkinson’s disease, this pairing can sound particularly promising. This disease, like heart attacks and arteriosclerosis, is long-term. It is also progressive–year after year goes by with no hope for improvement–and emotionally demanding for patients and families. However, science is scarcely close to what this phrase, so well-chosen from a marketing standpoint, suggests. Parkinson’s disease is a neurodegenerative brain disorder characterized by involuntary movements, rigidity, difficulty in balancing, and a progressive loss of dopamine-producing neurons. Treatment is still at a symptomatic management level, centered on the use of levodopa-based drugs, along with other medicines, plus rehabilitation. In the early stages, soon after diagnosis, when symptoms can be controlled with medication and some degree of normalcy restored to life, from time to time, deep-brain stimulation may also come into play for patients whose symptoms are harder to cope with.
That is exactly why the topic needs more than a simple yes-or-no answer. There are really two separate conversations hiding inside this keyword set. One is about Botanical Stem Cell Supplements and whether plant-derived products have any meaningful role in Parkinson’s disease. The other is about stem cell Parkinson’s disease research, which refers to real cell-based therapies being studied in clinical trials. Those are not the same thing, and treating them as if they were interchangeable is where a lot of online content starts to drift away from the truth.
- It is mostly a marketing phrase, not a recognized Parkinson treatment category
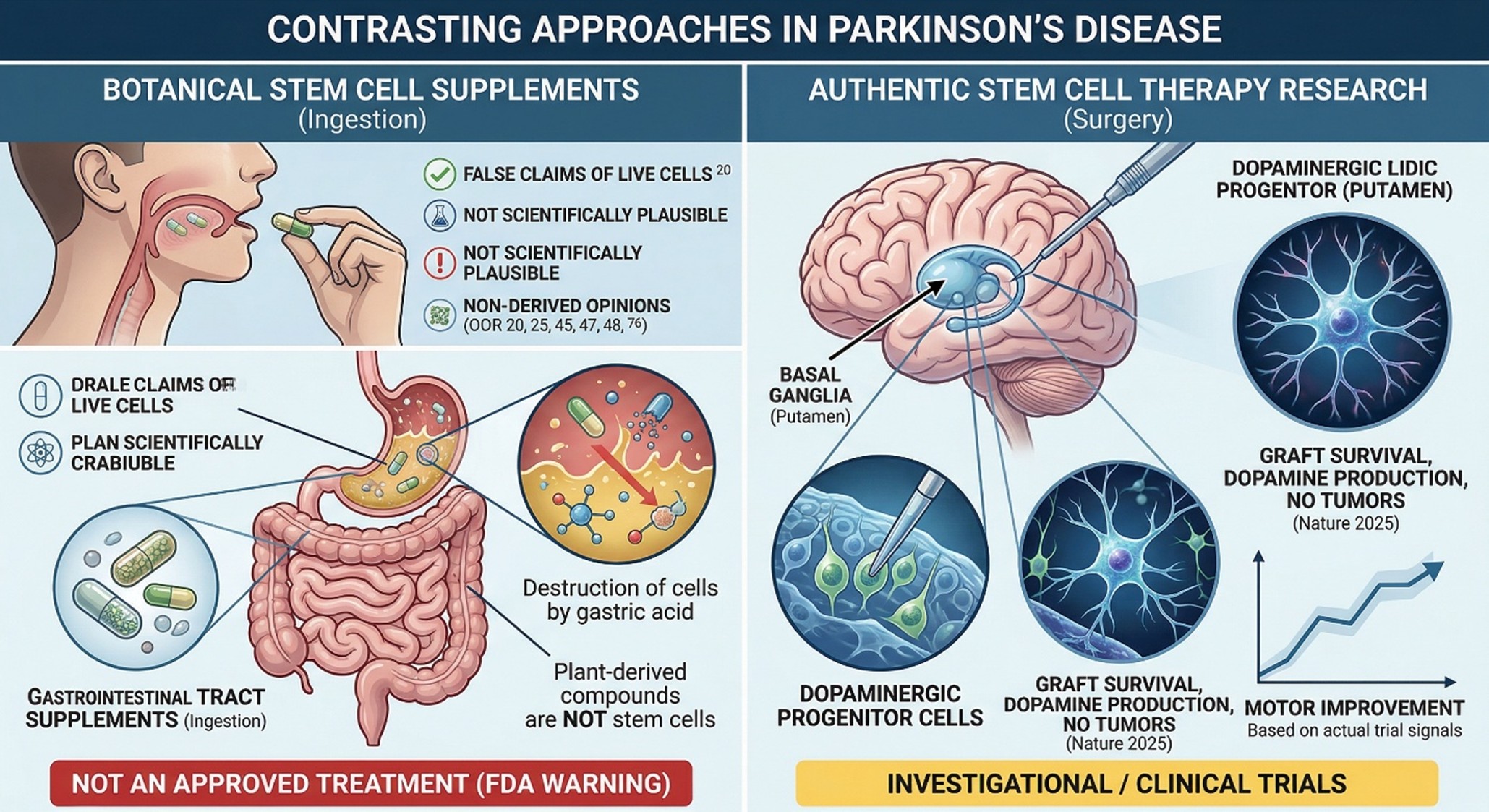
From a scientific standpoint, Botanical Stem Cell Supplements is not a standard therapeutic category in Parkinson’s guidelines or NIH educational material. In practice, the phrase often refers to botanical extracts, plant cell cultures, phytochemical blends, or supplements marketed with “stem cell” language layered on top. That does not automatically make every such product useless, but it does mean the wording itself should be approached carefully. Regulatory guidance from Singapore’s Health Sciences Authority explicitly states that claims that oral supplements contain active live stem cells are false and not scientifically plausible, noting that active stem cells require ultra-cold storage and would be destroyed in the gastrointestinal tract if swallowed.
That distinction matters because people often read “stem cell” in supplement branding and imagine genuine regenerative medicine. But real stem cell-based therapeutic products are not ordinary capsules. They are complex biologic interventions that generally require controlled manufacturing, clinical testing, and delivery methods that preserve biological activity. The HSA guidance is useful here because it cuts through the fog: supplements and authentic regenerative medicine products are fundamentally different things.
- “Natural” does not mean simple, and it definitely does not mean interaction-free
There is another reason this topic deserves care. Many people assume botanical products are inherently safe, especially when compared with prescription medicine. NCCIH warns that natural products can contain pharmacologically active compounds, may interact with prescription drugs, and may also carry side effects and safety risks. The Parkinson’s Foundation makes a similar point more specifically for PD, noting that the science around supplements, herbs, and botanicals remains limited and inconclusive, and that over-the-counter ingredients and dosages can vary greatly.
Where botanicals may still have a real place in the conversation
- Parkinson‘s research does look at plant-derived compounds, but mostly as supportive or exploratory science
This is where the article needs nuance. Just because ” botanical stem cell supplements “ is an imprecise phrase does not mean plant-derived compounds are irrelevant to Parkinson’s research. Reviews continue to examine natural products and phytochemicals for antioxidant, anti-inflammatory, mitochondrial, and neuroprotective effects in neurodegenerative disease, including Parkinson’s. But most of that literature is still exploratory, mechanistic, or preclinical rather than definitive clinical proof of disease modification in humans.
One good example of this is Mucuna pruriens, which is sometimes discussed in Parkinson’s disease circles since it naturally contains levodopa as a part of its seeds. The Parkinson’s Foundation narrates that some people have been trying it as a replacement for standard levodopa, but they also caution that safety and long-term side-effects are still not known. This is one area where it is well to remember: just because a plant is of real significance in biochemistry does not automatically translate into clinically available medicines whose effects and toxicity have been checked and evaluated.
- The human problem is often not the idea, but the gap between the idea and the product
This is what makes the supplement market so difficult to read. A molecule may look promising in cell studies or animal work, yet the final commercial product may still have uncertain dosing, inconsistent composition, poor bioavailability, or unclear interaction risks. That is one reason Parkinson’s Foundation guidance emphasizes involving the care team before adding supplements, especially when someone is already taking levodopa or other neurologic medications. It is also why Botanical Stem Cell Supplements can sound more advanced than the evidence really is.
What stem cell Parkinson’s disease research actually looks like
- This is a real scientific field, and it has become more clinically serious in 2025
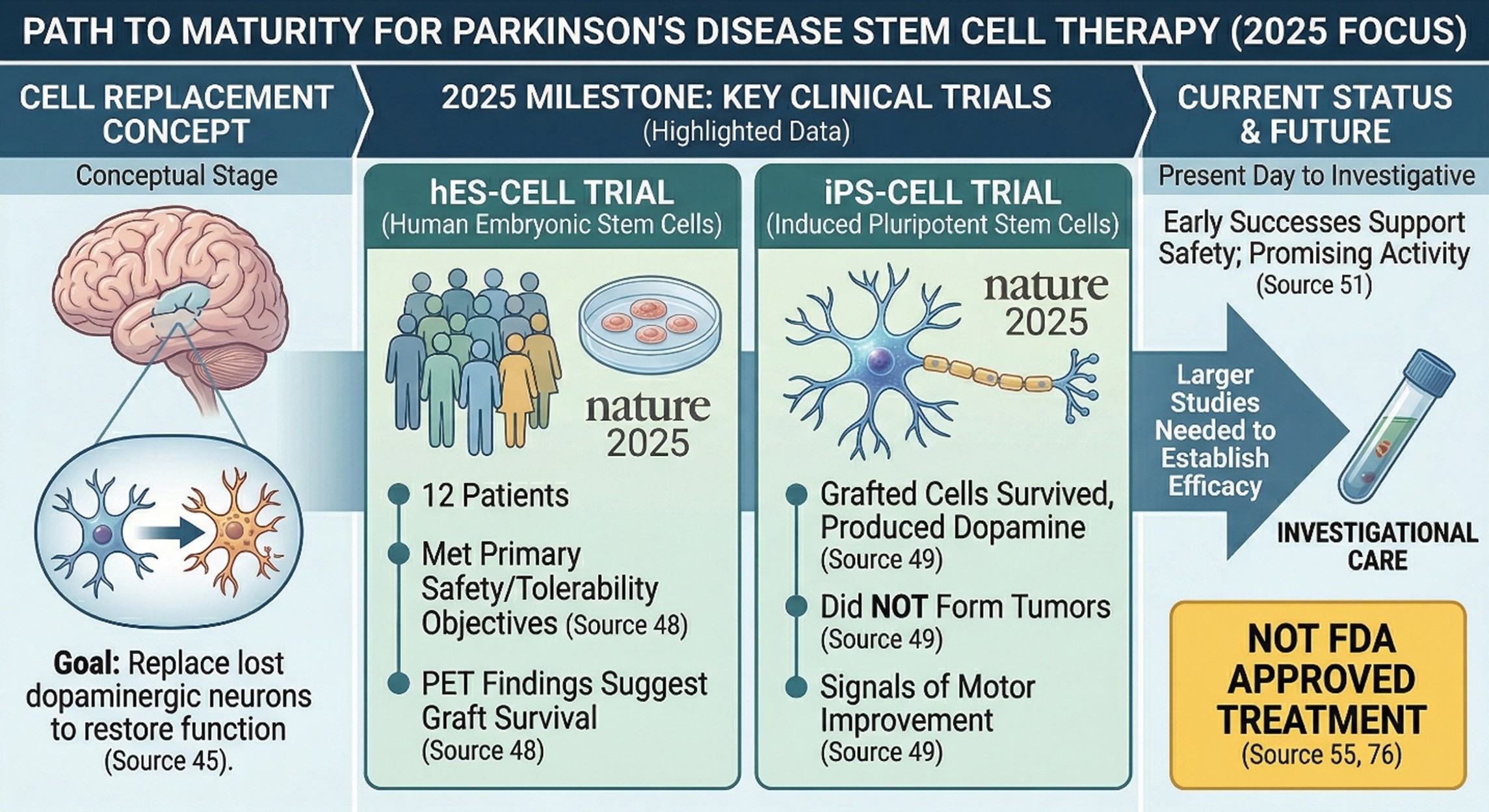
Unlike the supplement phrase, stem cell Parkinson’s disease research is a genuine and fast-moving clinical area. The central idea is biologically straightforward: Parkinson’s disease involves loss of dopaminergic neurons, so researchers are trying to replace or restore that missing neuronal function using cell-based approaches. Two major 2025 papers in Nature reported early-phase clinical trial results for dopaminergic progenitor cells derived from human embryonic stem cells and induced pluripotent stem cells. In the hES-cell trial, twelve patients received bilateral putamina grafts; the study met its primary safety and tolerability objectives at one year, with no adverse events related to the cell product, and PET findings suggested graft survival. In the iPS-cell study, grafted cells survived, produced dopamine, did not form tumors, and the small cohort showed signals of motor improvement.
That is a meaningful development. It moves the field beyond abstract hope and into actual human trial evidence. But the language still has to stay disciplined. Nature’s coverage of these trials described them as early-phase successes that support safety and suggest promising activity, while also making clear that larger studies are needed to establish efficacy. In other words, the field has matured, but it has not finished maturing.
- None of that means stem cell therapy is standard Parkinson’s care today
This is the point that would be most beneficial for your loved ones to listen to clearly. The FDA says that stem cell products are normally subject to approval, and it has not approved any product to deal with neurologic disorders like Parkinson’s disease. The FDA is also warning patients that they might be misled into using yet-unapproved, unsafe products. So, even if the body of statistical proof shows that stem cell Parkinson’s disease therapy is a dripping reality, this does not give wide approval for any clinic or product that uses stem cell vocabulary.
How to think about Botanical Stem Cell Supplements and Parkinson’s without getting lost
- The supplement question and the regenerative medicine question are different
For someone living with Parkinson’s, these are really two different decisions. The supplement decision is about whether a botanical or nutraceutical may support symptoms, sleep, constipation, mood, or general well-being, while also considering interaction risks and weak evidence. The regenerative medicine decision is about whether cell-based therapy might someday replace lost dopaminergic function or modify disease biology more directly. One conversation is about supportive care. The other is about experimental restorative neurology. They should not be merged just because both happen to sound modern.
- The most honest bottom line
The most honest reading of the current evidence is this: Botanical Stem Cell Supplements is a strong SEO phrase, but not a clearly defined Parkinson treatment category in mainstream medicine. Botanicals may matter in Parkinson research, and some compounds are biologically interesting, but clinical evidence remains limited, and product quality varies. By contrast, stem cell Parkinson’s disease research is real, increasingly credible, and newly strengthened by 2025 early-phase human trial data. Even so, it remains investigational, not routine care. That is the layered truth: the supplement side is often overstated, the cell-therapy side is genuinely advancing, and neither should be discussed without precision.
FAQ
- Are Botanical Stem Cell Supplements proven to treat Parkinson’s?
No. That phrase is not a recognized standard treatment category in Parkinson’s medicine, and evidence for most botanical supplements in PD remains limited or inconclusive.
- Can botanical products still matter in Parkinson care?
Sometimes, but mainly in a supportive or exploratory sense. Some botanicals and natural compounds are being studied for neuroprotective or symptom-related effects, but stronger human evidence is still needed.
- Is stem cell Parkinson’s disease treatment already approved?
Not as a standard approved treatment for Parkinson’s disease. The FDA says stem cell products have not been approved for neurological disorders such as Parkinson’s disease.
- Why is stem cell Parkinson’s disease research getting more attention now?
Because 2025 early-phase clinical trials reported that dopaminergic progenitor cell grafts appeared feasible and met important early safety goals, with signals that justify larger efficacy studies.