- Clinical Context of ALS and the Search for Advanced Supportive Care
Amyotrophic lateral sclerosis (ALS) is a lethal neurodegenerative disorder that is characterized by the progressive degeneration of upper and lower motor neurons. As these nerve cells slowly die off, patients may have progressively worsening weakness and loss of muscle control that make it difficult for them to do such things as walk, talk and swallow. Eventually, the muscles that help with breathing can’t work. And because it isn’t a disease, one but rather an entire spectrum of diseases, and one that stops and starts in no particular order, it turns into an extremely heavy-duty physical, emotional and practical burden for patients — and their families.
And despite tremendous advances in treating the nervous system, ALS remains one of modern medicine’s most complex diseases. The larger portrait of what treatment looks like today, with the exception of broad strokes, swings away from cure and toward slowing progression where it makes sense; managing side effects while sustaining quality of life. That’s why so many patients over and above families are seeking other adjuncts including regenerative medicine and cell-based therapy in a late-phase clinical program context.
- Pathophysiological Basis of ALS
The biology of ALS is complicated and no single mechanism can fully account for it all. It has been proposed by multiple independent studies that disease progression may be driven by several overlapping processes. These feature abnormal protein accumulation, oxidative stress, mitochondrial impairment, neuroinflammation, glutamate-associated excitotoxicity, and the progressive loss of motor neurons.
In a subset of patients, genetic mutations like those in SOD1 and related pathways associated with disease have been implicated in mismanagement of proteins. Proteins like TDP-43 are also frequently mentioned in ALS studies due to their link with neuronal malfunction. At the same time, elevated extracellular glutamate can hyperstimulate neurons to allow excessive calcium influx into cells leading to injury. This process leads motor neurons to become more susceptible and progress to apoptosis.
Clinically, this neural degeneration manifests as progressive muscle wasting and weakness. The more the signalling from brain, spinal cord and skeletal muscles diminishes, that is when voluntary motor control starts to fade. Depending on the pattern of disease onset, symptoms may develop asymmetrically and can involve the bulbar, cervical, thoracic or lumbosacral regions.
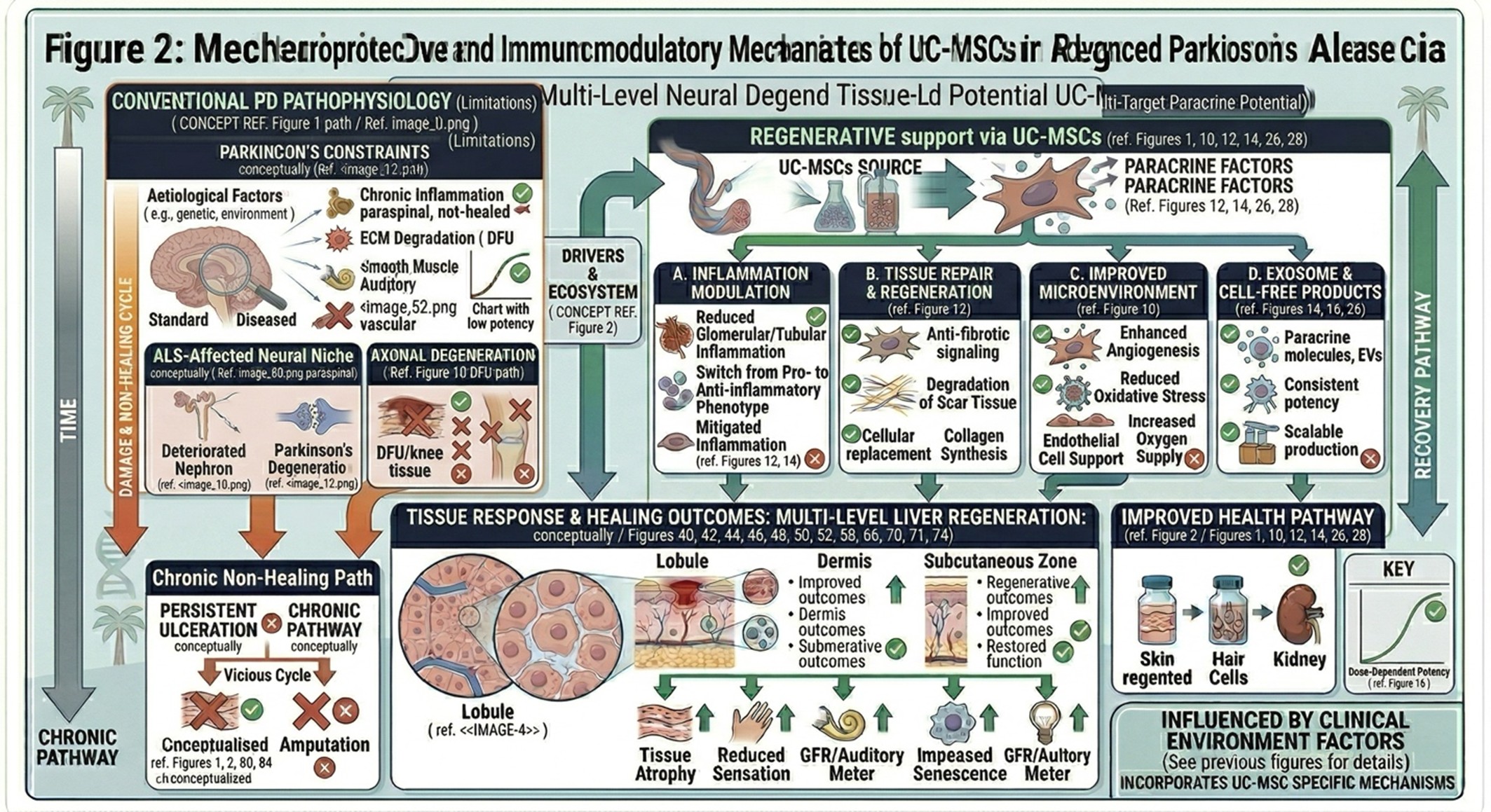
- Limitations of Conventional ALS Pharmacotherapy
Drug therapy for ALS, as it stands today, has limited reach. Theorial inhibitors such as riluzole have been used to moderately slow disease progression through glutamatergic signaling–related effects, half of which can be detected in electrophysiological studies. Edaravone, an antioxidant that can potentially attenuate injury related to oxidative stress, has been applied in a subset of patients as well.
Although these therapies might offer some benefit in specific situations, they do not reverse already established motor neuron loss or completely prevent long-term deterioration. Their role is mostly supportive, and more so as a disease-modifying agent than restoration. Consequently, patients can further suffer from progressive weakness and loss of independence, as well as requiring more supportive care with conventional therapy.
This clinical gap has led to interest in investigational strategies that not only target the symptoms, but more directly modulate the nervous system biology.
- Biological Rationale for Stem Cell–Based Approaches in ALS
For instance, in regenerative medicine, stem cell-based therapy is studied by contributing to damaged neural tissue as a result of modulatory processes beyond regular pharmacology. In ALS, the largest amount of interest has been directed towards mesenchymal stromal/stem cells (MSCs), such as umbilical cord-derived MSCs (UC-MSC), due to their purported immunomodulatory and trophic signaling properties.
These cells are not regarded as replacements in place of the missing motorneurons. Instead, they might function as a reservoir of biologically active mediators that promote neuronal survival and protect against inflammatory stress and tissue environment. When administered, mesenchymal cells can also migrate toward injured tissues and deliver their effect in a paracrine manner through secretion of factors like brain-derived neurotrophic factor (BDNF) or vascular endothelial growth factor (VEGF).
Other therapies based on MSC are also currently underway exploring their ability to regulate microglial activation and other inflammatory pathways. Like alarmins, by contributing to redirecting the local environment from a strongly neurotoxic regime these cells may establish a more protective microenvironment for damaged neural tissue. And that’s partly why stem cell–based treatment remains an area of scientific interest in ALS research.
- Regenerative Medicine Development in Thailand
The field of regenerative medicine in Thailand has become increasingly, both visible and accessible, due to the growth of clinical infrastructure, laboratory capability and translational biotechnology interests. Within the stem cell field, this has taken form in specialized centers with focus on cell preparation, clinical oversight and protocol-based treatment models.
In patients seeking advanced supportive care, Thailand is also often mentioned as a potential destination with the prevalence of regenerative medicine programs in organized medical settings. Such programs will not only depend on the existence of future cell-based therapies, but also the standards for laboratory processing as well as patient-, safety- and realistic outcomes-oriented monitoring.
While stem cell research in ALS is still at a relatively precoclinical stage of development, the options to investigate this type of regenerative medicine in Thailand are consistent with ongoing interest (worldwide) into interventional strategies that may offer some benefit for devestating conditions where management options remain limited.
- Concluding Perspective
Despite the availability of highly-effective therapies for controlling disease symptoms, there is still an unmet need for novel therapeutics that specifically target underlying biological complexity and motor neuron degeneration. While traditional pharmacologic therapies are required and effective, their limitations have resulted in a growing scientific interest in regenerative and immunomodulatory cell-based therapies.
One stem cell–based approach that is being studied involves mesenchymal stromal cells, which have been shown to support neuronal survival, modulate the inflammatory injury, and enhance the microenvironment surrounding damaged nerves. At the moment, these therapies must be considered supportive and experimental if not yet proven cures. Still, they are a major advance in the ongoing search for improved overall treatment of people with ALS.
Future management of ALS in well-structured clinical settings will probably be a growing partnership between classic neurology, supportive rehabilitation and the judicious use of regenerative treatments to maintain both function and quality of life for as long as possible.

