The phrase Botanical Stem Cell Supplements sounds modern, gentle, and almost naturally compatible with chronic autoimmune disease. That is probably why it attracts attention in Lupus searches. On the other hand, lupus is not a one-size-fits-all condition, and “natural” doesn’t always mean safe. It’s a disease for which the public cannot afford to be ahead of prevention or even behind treatment. SLE is a multisystem autoimmune disease, having far-reaching effects on the skin, joints, blood vessels, kidneys, and brain. The present evidence-based care of SLE is built on suppressing immune dysregulation rather than making the body “better” in general terms. EULAR’s 2022 recommendations on hydroxychloroquine in lupus were an unqualified “yes” for a Change. One considers non-urgent surgery after six months of stable treatment because the medicinal benefits of the positive effects do not yet outweigh potential risks, and it’s both healthier — for patients as well as providers — so long as everyone involved can live with a reasonable wait time.
Why Lupus changes the supplement conversation
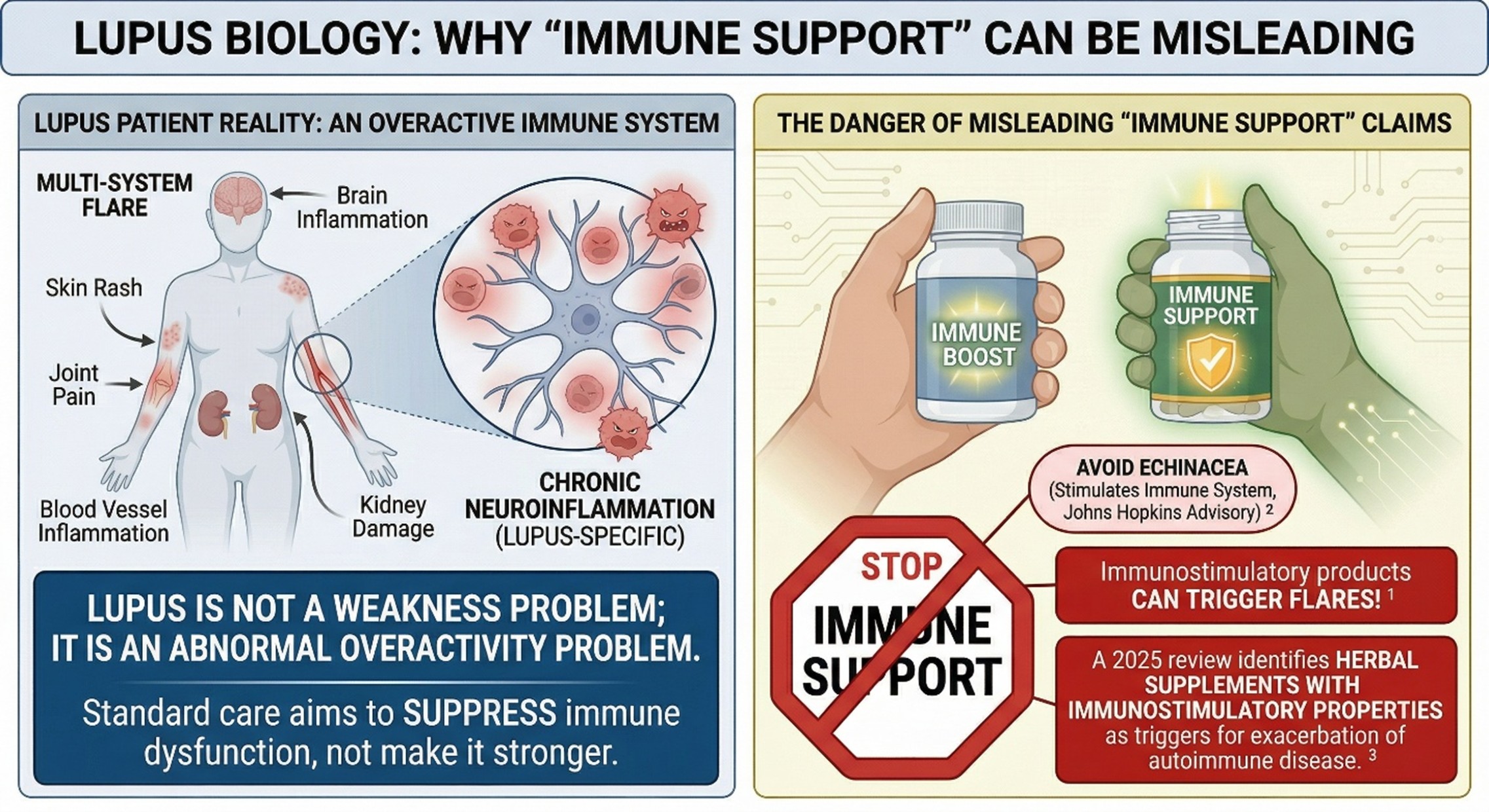
- Lupus is an immune overactivity problem, not an immune weakness problem
This is the first layer that matters. In many consumer wellness spaces, supplements are framed as ways to “support immunity” or “strengthen the body.” In Lupus, that framing can be misleading. Because lupus is driven by abnormal immune activation and loss of tolerance to self-antigens, anything marketed as strongly immunostimulatory deserves extra caution. That is why the supplement conversation in lupus is different from the supplement conversation in general wellness.
- Some herbal products may be the wrong fit for lupus biology
That caution is not theoretical. Johns Hopkins Lupus Center advises people with lupus to avoid echinacea because it may stimulate the immune system and trigger flares, and it also advises avoiding alfalfa sprouts because of compounds associated with increased inflammation in lupus. A 2025 scoping review in Lupus Science & Medicine similarly highlighted that herbal supplements with immunostimulatory properties may trigger or exacerbate autoimmune disease. So when Botanical Stem Cell Supplements are marketed with phrases like “immune support” or “cellular activation,” that language should be read carefully rather than passively accepted.
What “Botanical Stem Cell Supplements” usually mean in practice
- A useful SEO term, but not a defined lupus treatment category
Scientifically, Botanical Stem Cell Supplements is not a standard therapeutic category in lupus guidelines. This is an inference from the literature: published work around “plant stem cells” is concentrated far more in cosmetics, skin applications, plant cell extracts, and phytochemical biology than in recognized lupus treatment pathways. Regulatory sources also warn that supplement marketing often uses the phrase “stem cell” in ways that are not scientifically plausible, especially when products are sold orally as though they contained active, living stem cells.
- Plant-derived compounds are not the same thing as clinical stem cell therapy
There is real science behind plant-derived compounds, but it needs to be described accurately. A 2025 review in Nutrients explains that vitamins, minerals, and phytochemicals can influence mesenchymal stem cell function in experimental settings, including proliferation, differentiation, immunoregulation, and stress responses. That is interesting biology. It does not mean that oral Botanical Stem Cell Supplements act like a proven stem cell treatment for Lupus, and it does not turn a supplement into an established regenerative therapy. The gap between “may influence cell pathways in research models” and “works clinically in lupus” is still very large.
- Why the word “stem cell” in supplements should be handled carefully
One of the clearest regulatory warnings comes from Singapore’s Health Sciences Authority, which states that claims that currently marketed oral supplements contain live stem cells are false and not scientifically plausible, noting that active stem cells require very cold storage and would be destroyed by the digestive tract if swallowed. The same source also notes that genuine regenerative medicine products intended to treat disease are typically delivered as injections or infusions and must undergo rigorous clinical testing. That distinction is crucial for SEO writing: Botanical Stem Cell Supplements and injection stem cell approaches should never be treated as interchangeable ideas.
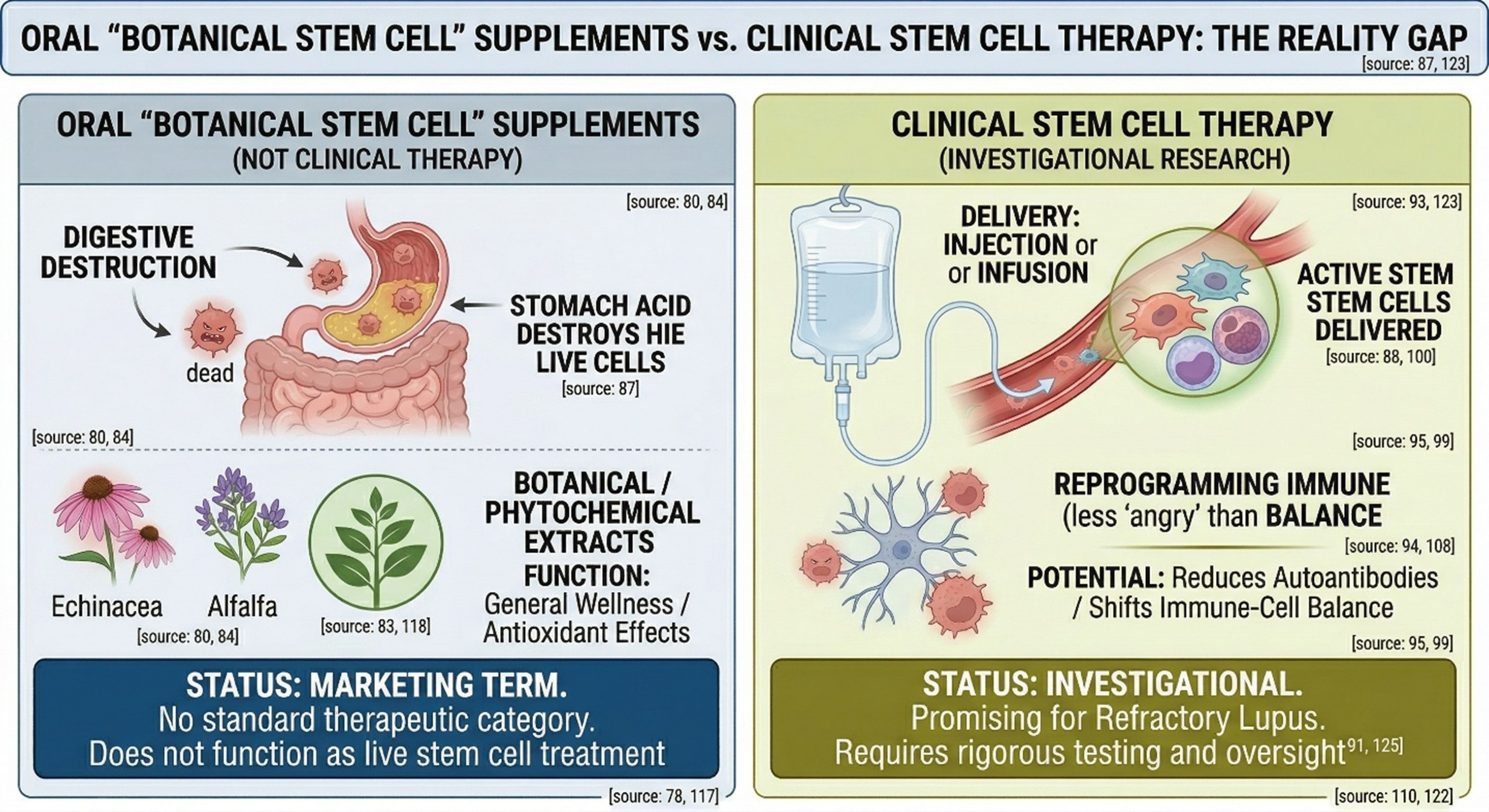
- Injection stem cell research is real, but it is still investigational
Unlike supplement marketing, injection stem cell research in Lupus does exist in the peer-reviewed literature. The main focus has been mesenchymal stromal or stem cells, with some literature also addressing hematopoietic stem cell transplantation in severe disease. A 2024 review reported that mesenchymal stem cell and hematopoietic stem cell approaches were associated with improvements in lupus disease activity and immunological markers in animal models and human studies, including reductions in autoantibodies and shifts in immune-cell balance. The Lupus Foundation of America likewise describes mesenchymal stromal cell therapy as a promising frontier, especially for refractory lupus that does not respond adequately to available treatment.
- Why has this still not become routine lupus care
But at the same time, that literature too is cautious. The 2024 update also speaks further about complications such as repeated infection and disease, yet by the Lupus Foundation standards, stromal-cell performance in and of itself has become a study question rather than one answer. Hence injection stem cell will appear as investigational in lupus rather than routine. Although early results are encouraging, translational medicine still needs to address more difficult questions: which patients, what cell source, what dose, what route of delivery, how lasting a result, and at what price for safety.
Regulatory agencies remain cautious about unapproved cell products
That caution is also reflected by regulators. FDA states that unapproved products made from human cells or tissues may pose serious risks, that many such products have not been reviewed for safety, purity, potency, or effectiveness, and that the agency continues to receive reports of adverse events, including patient deaths, involving unapproved products. FDA’s consumer alert also emphasizes that many stem cell products promoted online are marketed with misleading claims. So even when a phrase like injection stem cell sounds more medically serious than Botanical Stem Cell Supplements, it still needs to be judged by evidence and regulatory status rather than by tone.
How to think about Botanical Stem Cell Supplements and Lupus without oversimplifying either
- The supplement question and the regenerative question are different questions
This is where the article requires its finishing touch. With Lupus, such supplementation questions usually involve alleviation of symptoms, diet, inflammation (medication interaction ), and even which herbs could worsen an autoimmune disease or create more long-term trouble. The regenerative medicine question is another matter. It concerns whether stem cell therapy can safely reprogram the behavior of immune systems, reduce refractive disease processes, or offer protection for organs in patients who previously performed poorly under standard treatment. Both questions are worth asking, but calling a spade a spade can be hard to do with one such shiny phrase like “Precision Medicine”.
- The most honest reading of the evidence
The most honest conclusion is that Botanical Stem Cell Supplements may function mainly as a marketing umbrella unless the product’s ingredients, mechanisms, and claims are described with precision. Plant-derived compounds and nutraceuticals are scientifically interesting, and some may influence inflammatory pathways or cell biology in laboratory settings. But in Lupus, the stronger evidence base still sits with standard rheumatology care, while injection stem cell therapy remains an active but investigational area of research for selected refractory cases. That distinction does not make the field uninteresting. It makes the conversation more trustworthy.
FAQs
- Are Botanical Stem Cell Supplements proven to treat Lupus?
No established lupus guideline currently treats Botanical Stem Cell Supplements as a standard therapy category. The phrase is mostly better understood as marketing language around botanicals, phytochemicals, or plant-derived extracts rather than as a proven lupus treatment pathway.
- Can immune-boosting supplements be a problem in Lupus?
Yes, they can be. Johns Hopkins advises avoiding echinacea in lupus because it may provoke immune activity and flares, and it also advises avoiding alfalfa sprouts. A 2025 review also identified many herbal supplements with immunostimulatory properties that may aggravate autoimmune disease.
- Is injection stem cell therapy the same thing as a stem cell supplement?
No. Regulatory sources make a clear distinction. Oral supplements marketed as containing active stem cells are not scientifically plausible in the way they are often advertised, whereas true cell-based regenerative products are typically administered as injections or infusions and require rigorous testing and oversight.
- Is injection stem cell therapy already standard care for Lupus?
Not at this point. Reviews and patient-focused research organizations describe mesenchymal stromal cell therapy as promising, particularly for refractory lupus, but still investigational rather than routine standard care.