When people think about diabetes, they usually think about blood sugar, insulin, or long-term complications. But underneath all of that is a more fundamental biological story: diabetes is also a disease of cellular loss, oxidative stress, and cellular dysfunction. In type 1 diabetes, the immune system destroys insulin-producing beta cells in the pancreas. In type 2 diabetes, insulin resistance is part of the picture, but beta-cell dysfunction increasingly appears central to the progression from metabolic strain to overt hyperglycemia. That is why the topic of Natural Cellular Regeneration has become so compelling. It points attention back to the level where the disease actually unfolds: the cell.
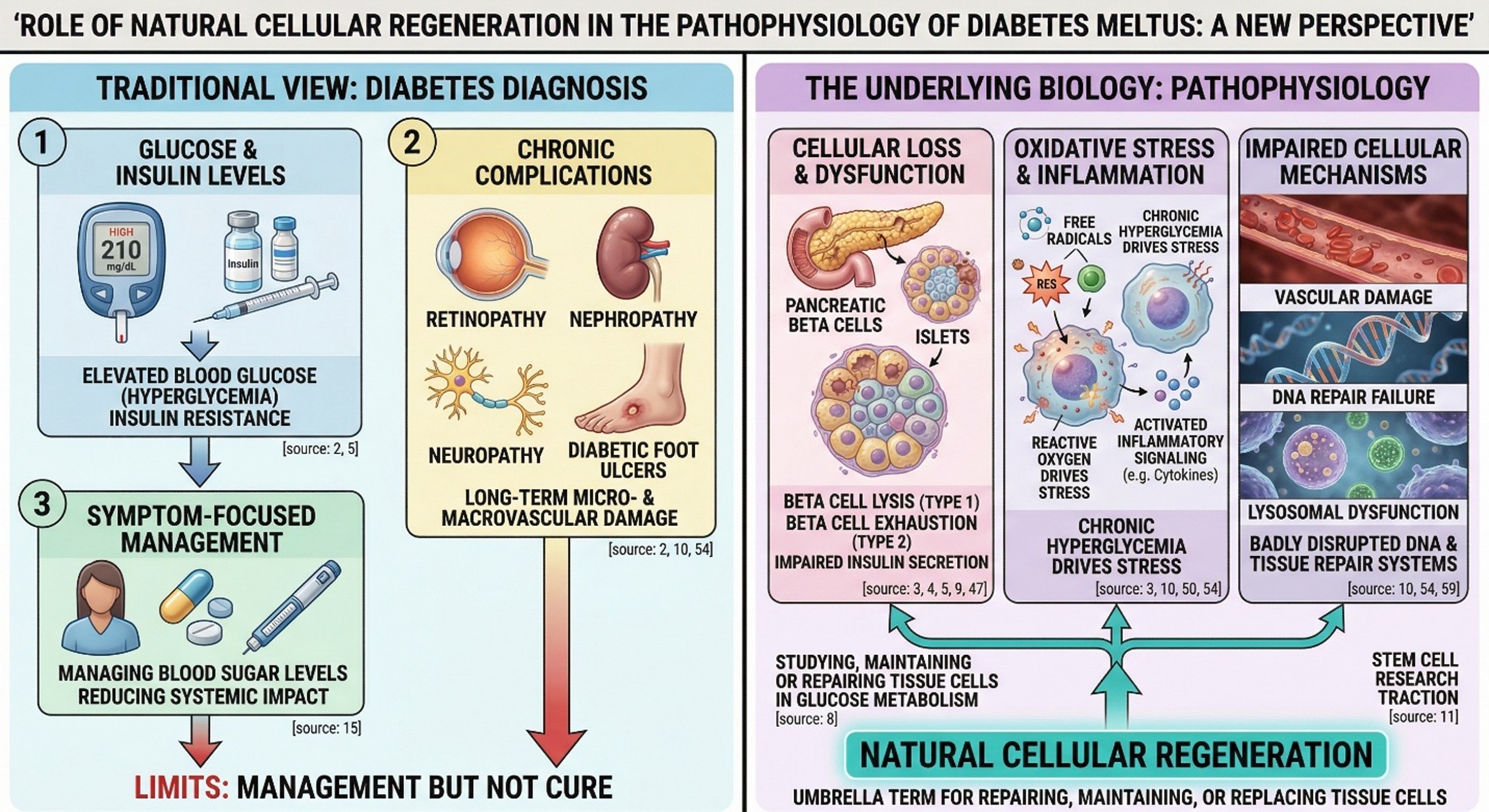
The term “Natural Cellular Regeneration” is applied in this article not as a generalization of something more specific but as an umbrella term for such things as studying, maintaining or repairing (and if necessary, replacing whole) tissue cells involved in glucose metabolism. In diabetes research, this typically means insulin-producing pancreatic beta cells and/or pancreatic islets; in some cases, also the vascular systems (e.g. blood vessels), lysosomal degraded proteins that inhibit new growth of blood vessels, or DNA repair systems come into operation as part and parcel with every episode of chronic hyperglycemia, all these things having been badly disrupted in patients suffering Not ago. This is where diabetes stem cell research has gained scientific traction. The field can no longer exist only as an idea. It’s increasingly centered on translational experiments that ask not whether a malfunctioning system can be managed from outside, topsy-turvy, but whether and how to restore damage metabolic function by natural biological means.
- Why regeneration matters in diabetes
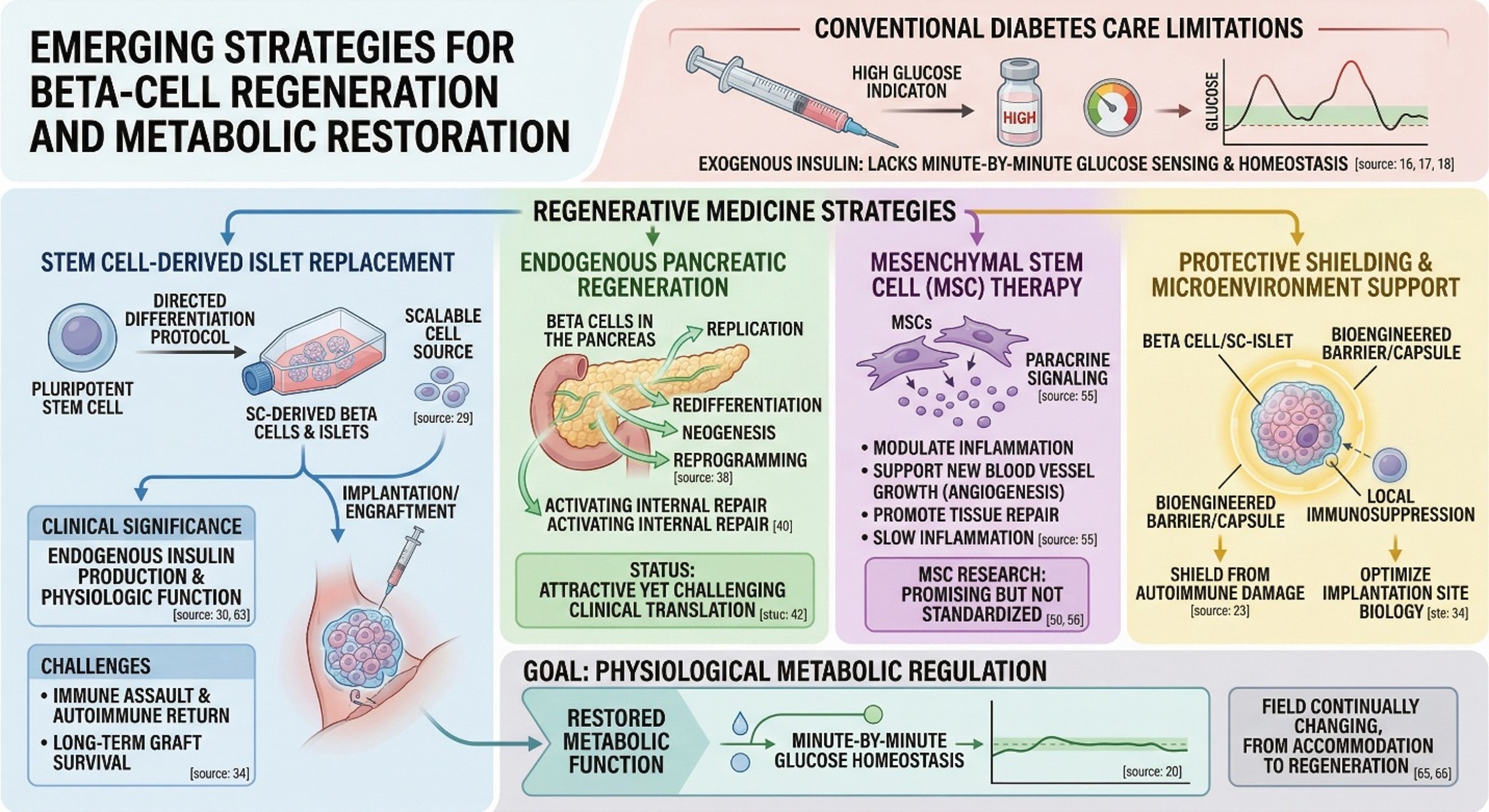
Conventional diabetes care remains essential, and for many people it is life-saving. But even excellent insulin therapy does not fully reproduce the dynamic behavior of living islet tissue. Real pancreatic beta cells do not simply release insulin in a fixed way; they sense glucose, respond to meals, coordinate with neighboring cells, and help maintain glucose homeostasis minute by minute. That is why researchers have continued to pursue regenerative medicine for diabetes. The goal is not only lower glucose values, but restoration of more physiological metabolic regulation.
In return, the article got its first real layer. Diabetes treatment and diabetes recovery is not the same thing. The former refuels after the tank is empty, while the latter tries to take a part of that filling back once again into function. But in practice, this translates into the application of islet cells derived from stem cells, beta-cell regeneration strategies, techniques to shield autoimmune damage from reaching the beta cells and ways of healing up or replacing tissues damaged during complications in diabetes. Each of these technologies is at a different stage of maturity and each deserves careful consideration rather than being rolled together into a great common pledge.
- What is stem cell research for diabetes showing in type 1 diabetes?
Today, the most powerful signal of this kind comes from type 1 diabetes. Over many years islet transplantation from a donor regularly showed that doing so could better metabolic control in selected patients. But this model has always been confined to donors and the need for long-term immunosuppression. Stem cell-derived islet strategies matter because they aim to get around at least part of this difficulty by creating replacement tissue in a more scalable form. A 2024 Nature portfolio article has described recently achieved and clinically meaningful benefits in metabolism, from transplanted stem cell-derived tissue The 2025 New England Journal of Medicine reported that stem cell-derived islets engrafted, made endogenous insulin, and restored physiologic islet function in a small, short-term study. That is a major turning point, but still should be read with temperance. These results do not mean that everyone’s diabetes has been reversed or stem cell therapy is routine care now. The present evidence is early, highly selected and still heavily influenced by major practical barriers. Researchers still have to work their way through immune assault and autoimmune return, implantation site biology, long-term gem survival quality and the clinical burden of scarce necropsy. In other words, stem cell research for diabetes gives encouraging signs, but it is still at a stage where promising results must be bounded and confirmed.
- The deeper question: can the pancreas regenerate from within?
This is where the conversation becomes more interesting. Not all regenerative ambition in diabetes depends on transplantation. Some researchers are trying to understand whether beta cells can be regenerated directly within the pancreas through replication, redifferentiation, neogenesis, or reprogramming. That idea sits very close to what many readers intuitively imagine when they hear Natural Cellular Regeneration. The attraction is obvious: if endogenous repair could be activated in a controlled way, the therapy might become biologically elegant as well as clinically powerful.
But the literature is also very clear that this is one of the hardest parts of the field. A 2024 review on beta-cell regeneration biology described direct pancreatic beta-cell regeneration as attractive, while also emphasizing that clinical translation has proven challenging. That makes this one of the most exciting frontiers in regenerative medicine for diabetes, but not yet one of the most established. Scientifically, it is better described as a serious direction of research than as a settled therapeutic reality.
- What about type 2 diabetes?
Type 2 diabetes adds another layer because the biology is broader. It is not only about insulin resistance. Human studies increasingly place pancreatic beta-cell dysfunction near the center of early disease progression, with inadequate insulin secretion helping drive the transition to hyperglycemia. That matters because it means regenerative thinking is not irrelevant to type 2 diabetes; it simply has to be framed differently.
In type 2 diabetes, stem cell research for diabetes is often explored less as simple beta-cell replacement and more as biologic support: modulation of inflammation, support of residual pancreatic function, improvement in metabolic signaling, and possibly protection of tissue under chronic stress Recent evidence syntheses report improvements in glycemic measures in some studies, including signals in HbA1c C-peptide, and insulin requirements, but they also highlight an important weakness in the literature: heterogeneity Cell source, dose, route of administration, follow-up length, and study design still vary substantially across trials So the field is promising, but not yet standardized enough for broad clinical certainty
- Natural Cellular Regeneration also applies to diabetes
One reason the theme has become so central is that diabetes isn’t just a problem for your pancreas. Long-term hyperglycemia not only affects blood vessels, extracellular matrix decomposition and systemic inflammatory signaling but also subsequent repair of any damage done to these sites by the body’s own system. “That’s why it becomes more and more crucial that rehabilitation medicine discusses problems with diabetic wounds,” said Dr. Infections from any source can be fatal if they happen to occur in a diabetic patient whose immune system is already compromised Review chapters on mesenchymal stem/stromal cells in 2024 and 2025 write that are relevant here as such cells can support new blood vessel growth and lay down its needed collagen, do paracrine signaling that promotes repair while slowing inflammation in general as well as tissue pores formation and repair. “Mesenchymal stem cells remain a hot topic,” said these paragraphs time and again 2025:.At the same time, they consistently emphasize the need for more standardized protocols and better corroboration in larger patient cohorts.
This part of the literature is especially useful because it prevents the whole topic from becoming too narrow. Natural Cellular Regeneration and diabetes is not only about replacing beta cells. It is also about understanding how diabetes weakens the body’s ability to repair itself, and how cell-based strategies might one day strengthen that repair response in specific clinical settings. That broader interpretation makes the keyword more meaningful and makes the article feel more human, because it reflects how the disease behaves in real life: not as a single defect, but as a system-wide burden on tissue resilience.
- Conclusion
The most credible conclusion is a balanced one. Stem cell research for diabetes has clearly moved beyond abstract possibility. For type 1 diabetes, there is now early human evidence that stem cell-derived islet replacement can result in engraftment and endogenous insulin production. The rational is that in type 2 a This literature demonstrates biologically feasible benefits which are increasingly measurable, but with considerable variability and many questions unanswered. This is a field continually changing, not yet changed. Until now, Natural Cellular Regeneration ought not to be seen as an assertion of cure; instead it represents a set of rigorous scientific tools for looking into how diabetes care might evolve over time from accommodation into regeneration.
FAQ
- Is stem cell research for diabetes already a cure for diabetes?
Current studies are promising, especially in selected type 1 diabetes settings, but the field still faces major questions around immune rejection, long-term durability, standardization, and safety. - Does Natural Cellular Regeneration apply only to type 1 diabetes?
The concept is highly relevant to type 1 diabetes because of beta-cell loss, but it also matters in type 2 diabetes, where beta-cell dysfunction and impaired tissue resilience contribute to disease progression and complications. - Why are diabetic wounds often included in regenerative medicine discussions?
Because chronic hyperglycemia can impair angiogenesis, inflammatory balance, collagen deposition, and wound closure, making tissue repair itself part of the diabetes That is why diabetic wound healing is a major focus in cell-based research. - What is the biggest limitation in stem cell research for diabetes right now?
There is not just one limitation. The main barriers include immune protection, graft survival, long-term function, manufacturing consistency, and the absence of standardized large-scale protocols across different study designs.