UC-MSCs Rheumatoid Arthritis stem cell Therapy Thailand advancement
Rheumatoid Arthritis represents a formidable challenge within modern rheumatology, characterized by a chronic, systemic inflammatory state targeting synovial joints. This condition is not merely joint discomfort; it is a progressive autoimmune disorder where immune surveillance mechanisms erroneously attack healthy synovial tissues. The severity is profound, leading to irreversible cartilage destruction, bone erosion, and systemic complications including cardiovascular involvement. For patients, the impact is devastating, frequently resulting in physical disability and loss of functional independence.
While pharmaceutical interventions have advanced, many patients remain non-responsive or suffer from debilitating side effects. This therapeutic gap necessitates exploring innovative regenerative medicine. The emergence of stem cell Therapy Thailand offers a transformative perspective through the application of UC-MSCs. By moving beyond symptom management, these advanced biological therapies aim to reset the immune environment, providing hope for long-term remission where conventional medicines have failed to provide adequate relief.
- Pathophysiological Mechanisms and Clinical Manifestations
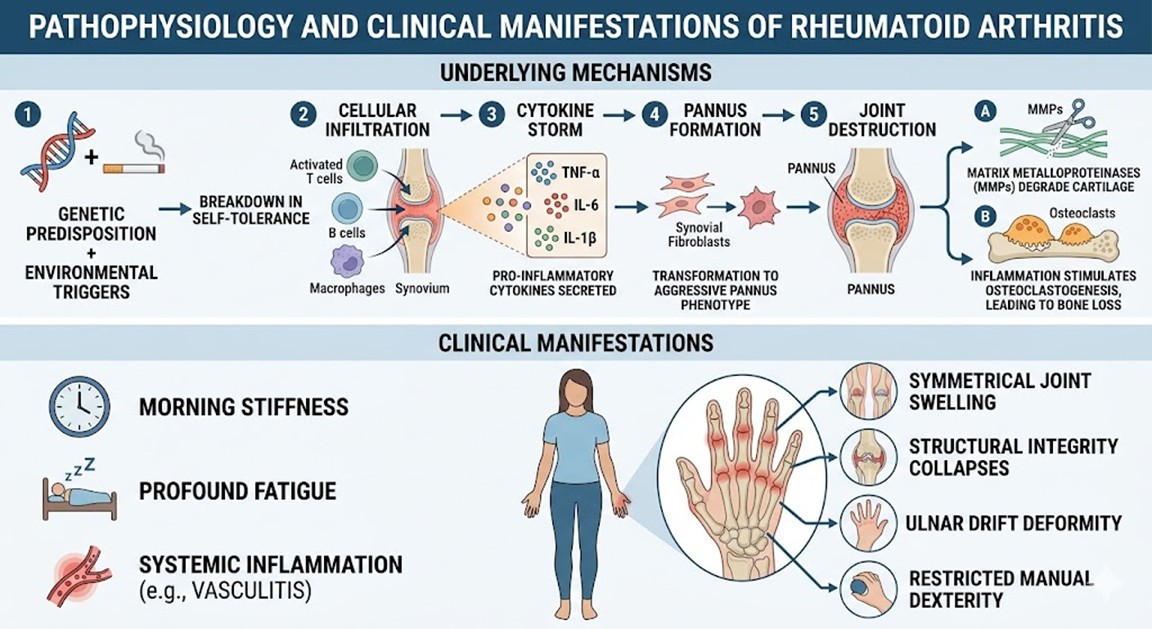
The pathogenesis of Rheumatoid Arthritis involves a complex interplay between genetic predisposition and environmental triggers, leading to a breakdown in self-tolerance. At the molecular level, the disease is driven by the infiltration of activated T cells, B cells, and macrophages into the synovium. These cells secrete a potent cocktail of pro-inflammatory cytokines, most notably Tumor Necrosis Factor-alpha (TNF-α), Interleukin-6 (IL-6), and Interleukin-1 (IL-1β). This cytokine storm induces the transformation of synovial fibroblasts into an aggressive, tumor-like phenotype known as the pannus.
As the pannus expands, it releases matrix metalloproteinases (MMPs) that actively degrade the cartilage matrix. Simultaneously, the inflammatory environment stimulates osteoclast genesis, leading to localized bone loss. Clinically, this manifests as symmetrical joint swelling, morning stiffness, and profound fatigue. If left unchecked, structural integrity collapses, leading to deformities such as ulnar drift, which severely restrict manual dexterity. The systemic nature of inflammation means patients often experience secondary issues like vasculitis, increasing the urgency for targeted intervention.
- Conventional Treatment Modalities and Limitations
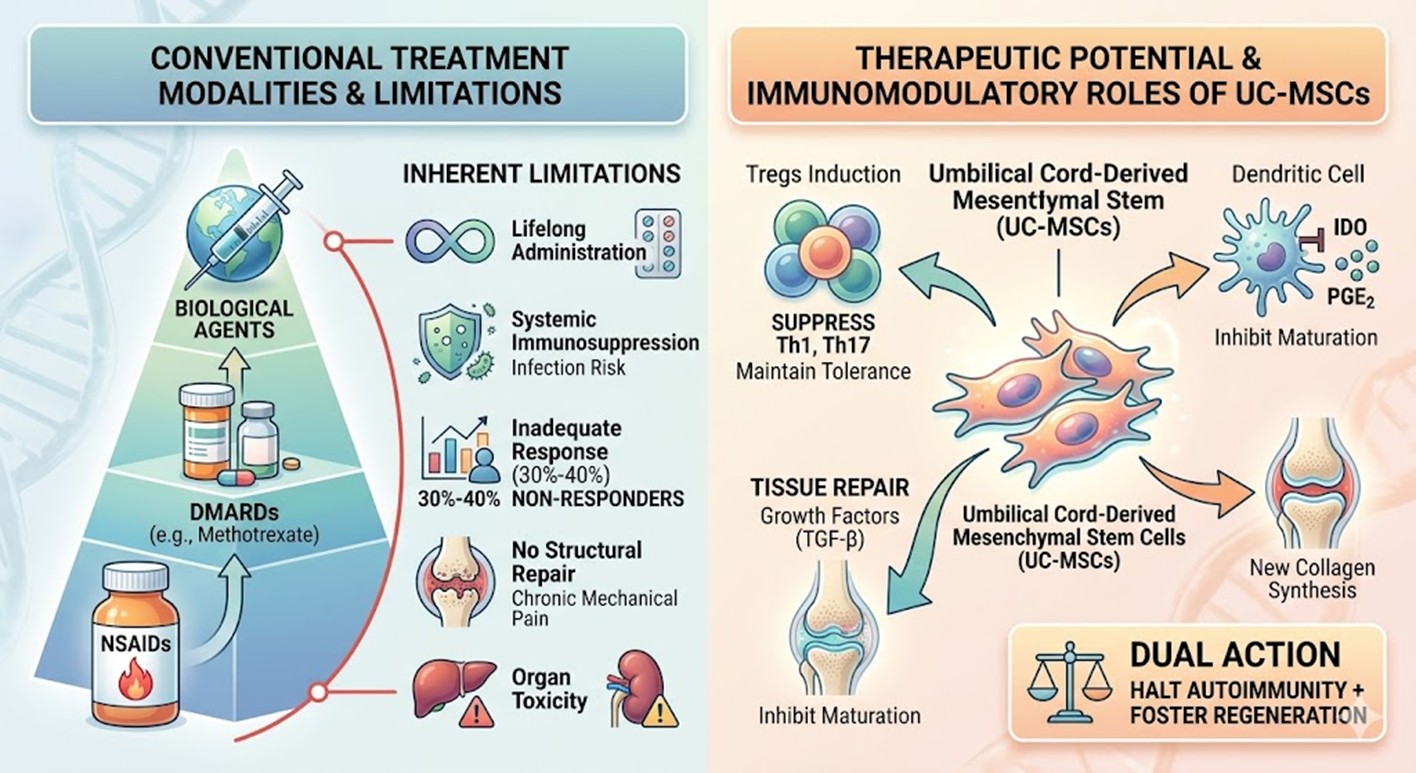
Currently, the clinical standard involves a tiered approach starting with Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), followed by Disease-Modifying Antirheumatic Drugs (DMARDs) like methotrexate. In refractory cases, biological agents targeting specific cytokines are utilized. While these treatments have improved outcomes, they are far from perfect.
The primary limitation of traditional DMARDs is the requirement for lifelong administration and the risk of systemic immunosuppression, leaving patients vulnerable to infections. Furthermore, approximately 30% to 40% of patients are classified as inadequate responders, failing to achieve low disease activity. Additionally, these drugs manage inflammation but do little to repair structural damage already inflicted upon the cartilage and bone, leaving patients with chronic mechanical pain. This creates a cycle of dependency on medications that may eventually lose efficacy or cause organ toxicity over prolonged periods.
- Therapeutic Potential and Immunomodulatory Roles of UC-MSCs
The shift toward using Umbilical Cord-Derived Mesenchymal Stem Cells, or UC-MSCs, is driven by their superior proliferative capacity and potent immunomodulatory profile compared to adult bone marrow sources. These cells possess a unique ability to sense the inflammatory temperature of their environment. When introduced into a high-inflammation setting, UC-MSCs exert influence through direct cell-to-cell contact and the secretion of bioactive paracrine factors.
One primary mechanism involves the induction of regulatory T cells (Tregs), essential for maintaining peripheral tolerance and suppressing overactive Th1 and Th17 immune responses. UC-MSCs also secrete Indoleamine 2,3-dioxygenase (IDO) and Prostaglandin E2 (PGE2), which inhibit the maturation of dendritic cells. Beyond immune modulation, these cells contribute to tissue repair by secreting growth factors like TGF-β that stimulate endogenous chondrocytes to synthesize new collagen. This dual action—halting the autoimmune assault while fostering a pro-regenerative microenvironment—is why stem cell Therapy Thailand has become a focal point for researchers seeking a holistic cure. These cells essentially act as biological pharmacists, dispensing signals to dampen inflammation exactly where needed.
- Future Trajectory of MSC Research within Thailand
Thailand has rapidly positioned itself as a global hub for regenerative medicine, specifically in the clinical application of mesenchymal lineages. The future of UC-MSCs in this region is supported by a robust infrastructure of specialized clinics and a regulatory environment encouraging biotechnological innovation. The reason why stem cell Therapy Thailand is gaining such momentum is the combination of world-class medical expertise and the increasing accessibility of advanced cell-based products.
Looking ahead, the integration of UC-MSCs into standard rheumatological care in Thailand is expected to move toward personalized protocols. Researchers are currently investigating the use of primed MSCs to enhance their homing capabilities to inflamed joints. Thailand’s strategic focus on medical tourism ensures the nation remains at the forefront of this field, providing a viable destination for patients seeking alternatives to lifetime pharmaceutical dependence. The focus is shifting from simple injection to a comprehensive therapeutic experience combining cellular biology with precision monitoring. As more clinical data emerges from local institutions, confidence in these therapies continues to rise, attracting international interest into the Thai biotech sector.
In summary, the autoimmune landscape is shifting significantly. While traditional therapies provide a foundation, drug toxicity and incomplete efficacy remain problematic. The utilization of UC-MSCs offers a sophisticated biological solution addressing root causes of synovial inflammation rather than just symptoms. By leveraging the natural reparative qualities of umbilical cord cells, clinicians provide a path toward joint restoration.
As evidence supporting stem cell Therapy Thailand grows, these cellular interventions represent the next frontier. For those living with Rheumatoid Arthritis, transitioning from chronic management to regenerative healing offers renewed possibility. Ongoing advancements in Thailand ensure access to high-tier biological tools, marking a pivotal moment against degenerative conditions. This evolution promises a future where long-term remission and physical restoration become primary goals, reducing the global disability burden. The synergy between cell biology and Thai clinical expertise sets a new benchmark for treating autoimmune pathologies.

